spectrophotometric determination of the pka of bromothymol blue discussion
|
Spectrophotometric Determination Of The Pka Of Bromothymol Blue
In the experiment the pKa of bromothymol blue (3'3"- dibromo-thymolsulfonephthalein) is determined by the two methods which have been discussed. At pH less |
|
Spectrophotometric Determination of the pKa of Bromothymol Blue
causing the solution color to change depending on the concentration of each form present. For example in strongly acidic solution |
|
Spectrophotometric Determination of the pKa Isosbestic Point and
7 déc. 2014 Some colorimetric pH indicators such as bromothymol blue in acidic solutions |
|
Determination of acid dissociation constants of Alizarin Red S
19 mars 2020 The pKa values of the diprotic Alizarin Red S (ARS) and the monoprotic. Bromophenol Blue (BPB) |
|
SPECTROPHOTOMETRIC DETERMINATION OF THE pKa OF AN
First a solution of bromothymol blue is prepared in acidic solution where essentially the entire indicator remains in the HIn form. The absorption spectrum is |
|
RGB-LED-PHOTOMETER AND THE DIGITAL IMAGE-BASED
calculation for determining the equilibrium constant and pKa of bromothymol blue which was used as proof of concept. The advantages are the use of low-cost ... |
|
Bromothymol Blue and Bromocresol Purple as Indicators of pH
We have assumed an approximate extinction coefficient of 20 mil-l cm-1 calculated from the data of Kuntz et al. [13] in our estimation of electron flow. Unless |
|
EXPT. 5 DETERMINATION OF pKa OF AN INDICATOR USING
These can then be used to compute the pKa value of methyl red using Henderson-Hasselbach equation. This forms the basis of the spectrophotometric determination |
|
Interactions between charged dye indicators and micelles to
determination of surfactants at various pH values. Methods: UV spectra of neutral red (NR) and bromothymol blue (BTB) were recorded and the absorbance at ... |
|
Spectrophotometric Determination of an Equilibrium Constant
3 déc. 2017 ... pKa of bromothymol blue (3'3"- dibromo- thymolsulfonephthalein) by the two methods which have been discussed. At a pH which is less than 6 ... |
|
Spectrophotometric Determination Of The Pka Of Bromothymol Blue
Spectrophotometric Determination of the pKa of Bromothymol Blue. INTRODUCTION. Acid-base indicators are compounds that are simply weak acids (or bases) that |
|
Physical Chemistry
Determination of the acidity con- stant of bromothymol blue To determine the acidity constant and the pKa value of bromothymol blue. |
|
Determination of acid dissociation constants of Alizarin Red S
2020?3?19? Alizarin Red S Methyl Orange |
|
Spectrophotometric Determination of the pKa Isosbestic Point and
2014?12?7? Some colorimetric pH indicators such as bromothymol blue in acidic solutions |
|
EXPT. 5 DETERMINATION OF pKa OF AN INDICATOR USING
5.3 Spectrophotometric Determination of pKa Value of Indicator 5.1: Schematic spectra of ionised (basic) form (blue curve) and unionised (acidic). |
|
Lab.10. Equilibrium. Determination of dissociation constant for a
This is the point at which the pH of the solution equals pKA of the indicator. The acid dissociation constant of bromothymol blue can thus be obtained by |
|
A High-Throughput Amenable Colorimetric Assay for
(Bromothymol blue 0.01%) were selected in such a way that both have the same Key words: nitrilase |
|
Extractive-spectrophotometric methods for determination of anti
2010?11?13? rapid and accurate determination of amantadine HCl (AMD) using bromocressol green (BCG) bromophenol blue (BPB) and bromothymol blue (BTB). |
|
Experiment # 11: Spectroscopic determination of indicator pKa
pH indicators may be defined as highly colored Bronsted-Lowry acid-base conjugate pairs. Used in low concentrations these compounds signal pH changes |
|
Interactions between charged dye indicators and micelles to
red (NR) and bromothymol blue (BTB) were recorded and the absorbance at the 2.3 CMC determination using UV spectrophotometry ... Results and discussion. |
|
Spectrophotometric Determination Of The Pka Of Bromothymol Blue
In the experiment the pKa of bromothymol blue (3'3"- dibromo-thymolsulfonephthalein) is determined by the two methods which have been discussed At pH less |
|
Spectrophotometric Determination of the pKa of Bromothymol Blue
Two spectrophotometric graphical methods were used to estimate the dissociation constant (pK b) with resulting average value of 10 12±0 41 Download Free PDF |
|
PKa OF AN ACID-BASE INDICATOR - chem321labspring11
In this experiment spectrophotometry is employed to measure the pKa of bromothymol blue an acid-base indicator The indicator (HIn) is a monoprotic acid |
|
Spectrophotometric Determination - Scientific Research Publishing
7 déc 2014 · The pKa can be determined from the spectrophotometric data using Some colorimetric pH indicators such as bromothymol blue in acidic |
|
Bromothymol Blue Spectrophotometry - Report & Experiment - Odinity
10 déc 2017 · Experiment and Report - Determining the pKa of Bromothymol Blue by spectrophotometry To determine the pKa of an acid-base indicator |
|
Spectrophotometric Determination of pKa of Bromothymol Blue
Spectrophotometric Determination of the pKa of Bromothymol Blue Purpose • To review the concepts of pH calculations and measurements |
|
Physical Chemistry - LD Didactic
To investigate the pH dependency of the colour of bromothymol blue To determine the acidity constant and the pKa value of bromothymol blue To use and |
|
Spectrophotometric Determination of the pka of Bromothymol Blue
Spectrophotometric Determination of the pka of Bromothymol Blue INRODUCION cidbase indicators are compounds that are simply weak acids (or bases) that |
|
Spectrophotometric Determination of the pKa Isosbestic Point and
18 fév 2023 · PDF The pKa and the isosbestic point of the universal pH indicator Carlo Ebra 1-11 (catalog number 45712) were determined using UV-Vis |
|
Determination of acid dissociation constants of Alizarin Red S
19 mar 2020 · The pKa values of the diprotic Alizarin Red S (ARS) and the monoprotic Bromophenol Blue (BPB) Bromothymol Blue (BTB) and Methyl Orange (MO) |
What is the pKa value of bromothymol blue indicator?
Acidity and basicity measurement
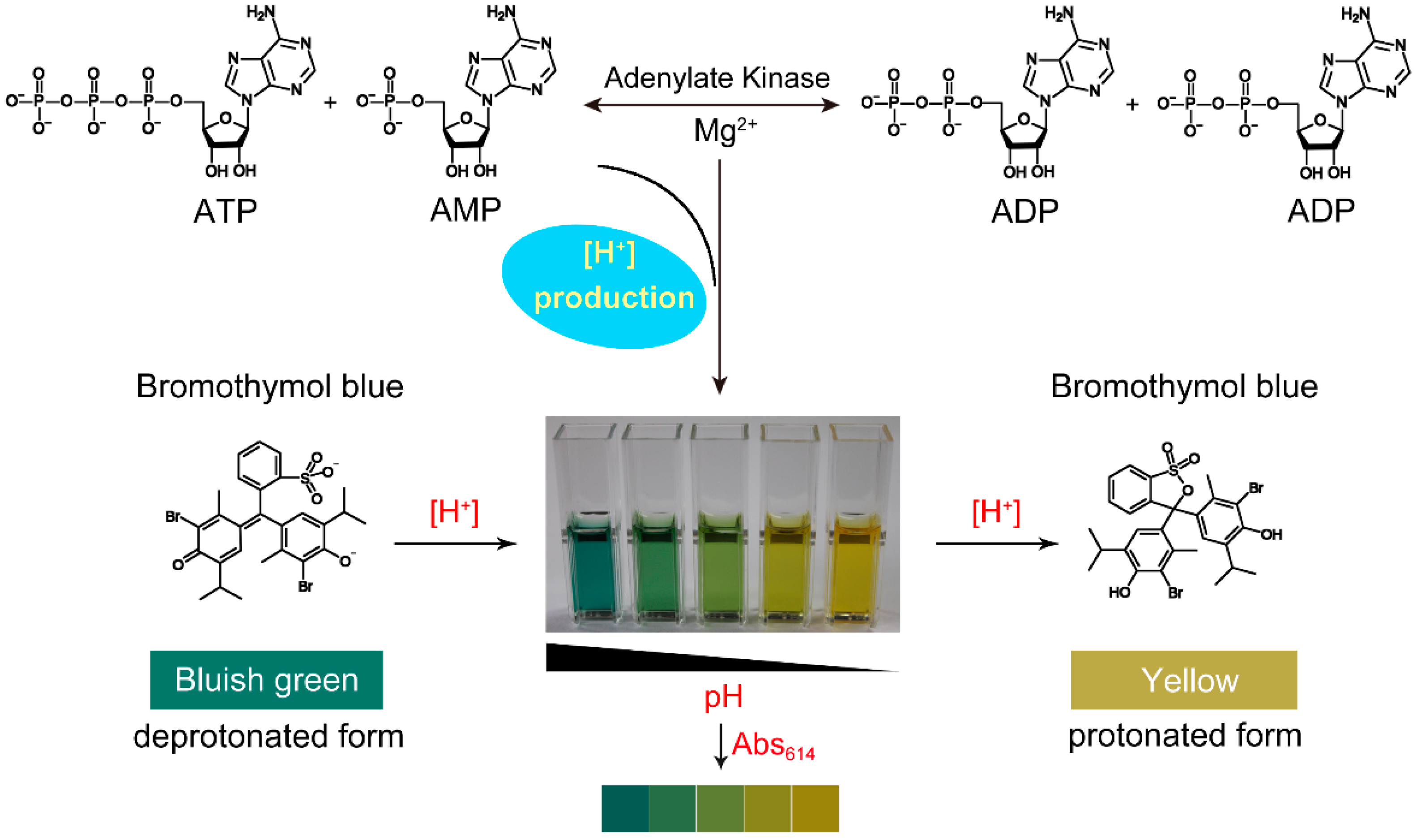
The employed indicators included dimethyl yellow (pKa = 3.3), methyl red (pKa = 4.8), neutral red ( pKa = 6.8 ), bromothymol blue ( pKa = 7.2 ), and thymol blue (pKa = 8.9).How can you determine pKa using spectrophotometry?
By using UV-VIS spectroscopy, we can determine the pKa of an indicator, which are weak acids or bases. Our indicator has a protonated and deprotonated form, which absorbs differently at different wavelengths. Since they absorb differently, the absorbance spectra can be used to determine the value of the pKa.What is the approximate pKa of bromophenol blue?
The actual pKa of bromothymol blue is 7.1 and the actual pKa of phenol red is 7.8.
|
Spectrophotometric Determination Of The Pka Of Bromothymol Blue
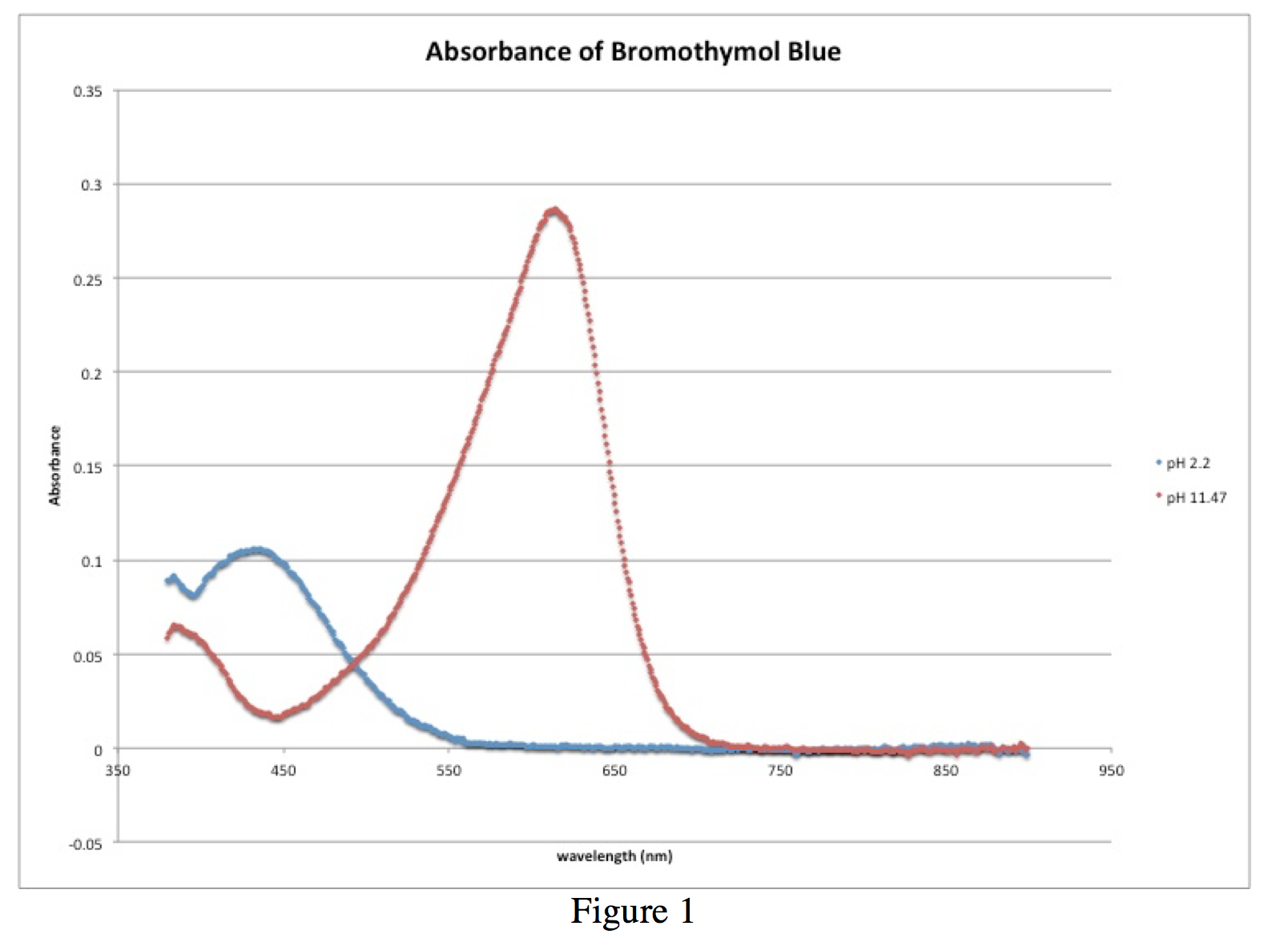
In the experiment the pKa of bromothymol blue (3',3"- dibromo-thymolsulfonephthalein) is determined by the two methods which have been discussed At pH less than 6, the indicator is yellow and at pH greater than 7 6, the indicator is blue At an intermediate pH, the blue and yellow combine to yield a green solution |
|
Physical Chemistry - LD Didactic
protonated forms are known in the equilibrium, the pKa value can be determined The indicator bromothymol blue used here is blue in alkaline form and yellow |
|
SPECTROPHOTOMETRIC DETERMINATION OF THE pKa OF AN
SPECTROPHOTOMETRIC DETERMINATION OF THE pKa OF AN of bromothymol blue is prepared in acidic solution where essentially the entire indicator remains in the HIn discuss the effect of ionic strength on the pKa value Discuss all |
|
Experiment &# 11: Spectroscopic determination of indicator pKa - ULM
This color change range depends upon the relative acid strength (or pKa ) of the conjugate the Henderson-Hasselbalch equation will be used to determine pKa values of various indicators As in the earlier blue (610 nm) Chlorophenol red |
|
Spectrophotometric Determination of Conditional Acidity Constant of
Spectrophotometric studies of bromothymol blue, bromophenol blue and pKa values of the reagents due to combination of electrostatic and micro Theory As it is known [30, 31], the acid-base equilibrium can be described by the following |
|
Pka of Indicators
Spectrometric Determination of the Acid Dissociation Constant of an Acid-base Indicator Learning Details of these measurements are discussed below You will be assigned to study one indicator, either bromophenol blue or phenol red |
|
A Simplified Method for Finding the pKa of an Acid–Base Indicator
3 mar 1999 · chemistry laboratory courses is the spectrophotometric The solution for determining the pKa of bromophenol blue Discussion of Results |
|
Investigation 7: Spectrophotometric Measurement of an - AWS
Investigation 7: Spectrophotometric measurement of an Equilibrium Bromothymol blue is an acid-base indicator that is yellow at pH values less than 6 You will measure three of these values for a solution that is green in color, meaning that |
|
Spectrophotometric Determination of the pKa, Isosbestic Point and
7 déc 2014 · Design of Buffers, Spectrophotometric Titration, Determination of pKa, Isosbestic Point, Results and Discussion Some colorimetric pH indicators, such as bromothymol blue in acidic solutions, are unstable over long |






















![SPECTROPHOTOMETRIC DETERMINATION OF NICOTINE IN - [PDF Document] SPECTROPHOTOMETRIC DETERMINATION OF NICOTINE IN - [PDF Document]](https://scielo.conicyt.cl/fbpe/img/jcchems/v56n4/fig22-01.jpg)



