alkene reaction with naoh
|
Chapter 7: Alkenes: Reactions and Synthesis
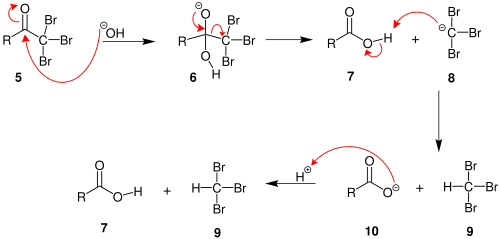
alkene halohydrin Br2 H2O HBr OH anti stereochemistry Organic molecules are sparingly soluble in water as solvent The reaction is often done in a mix of organic solvent and water using N-bromosuccinimide (NBS) as the electrophilic bromine source O OH O + N Br DMSO H2O Br + N H Note that the aryl ring does not react!!! |
|
Reactions of Alkenes
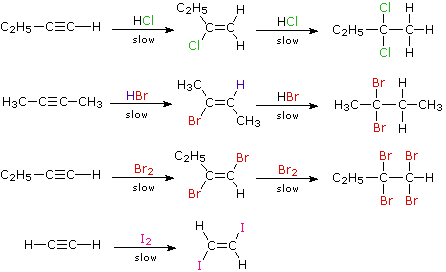
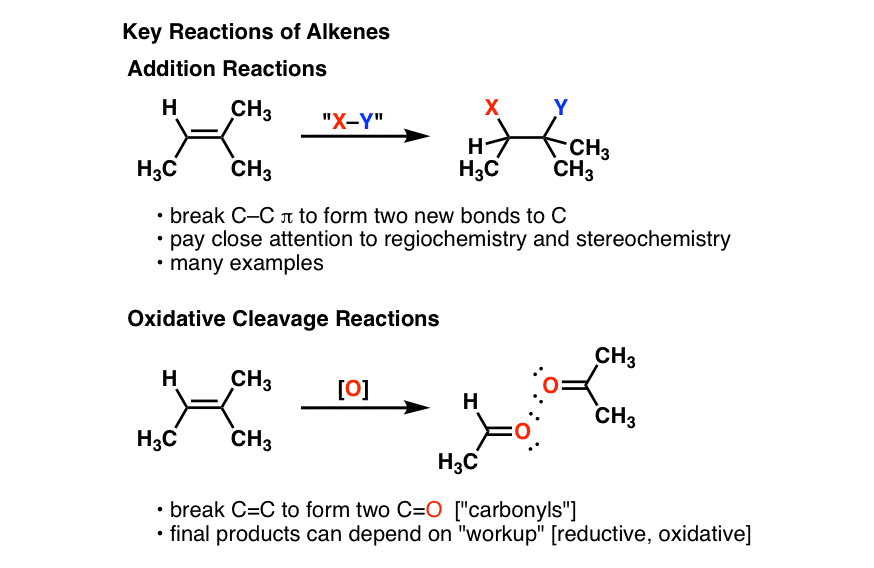
Electrophilic addition is probably the most common reaction of alkenes Consider the electrophilic addition of H-Br to but-2-ene: The alkene abstracts a proton from the HBr and a carbocation and bromide ion are generated The bromide ion quickly attacks the cationic center and yields the final product |
Does alkene react with water?
This action is not available. An alkene does not react with pure water, since water is not acidic enough to allow the hydrogen to act as an electrophile to start a reaction. However, with the presence of small amount of an acid, the reaction does occur with a water molecule added to the double bond of alkene, and the product is an alcohol.
What is the mechanism for acid-catalyzed hydration of alkene?
The mechanism for acid-catalyzed hydration of alkene is essentially the same as the mechanism for the addition of hydrogen halide, HX, to alkenes, and the reaction therefore follows Markovnikov’s rule as well in terms of regioselectivity. The hydration of 1-methylcyclohexene and the reaction mechanism are shown below.
How do alkenes react with bromide ion?
Electrophilic addition is probably the most common reaction of alkenes. The alkene abstracts a proton from the HBr, and a carbocation and bromide ion are generated. The bromide ion quickly attacks the cationic center and yields the final product. In the final product, H-Br has been added across the double bond.
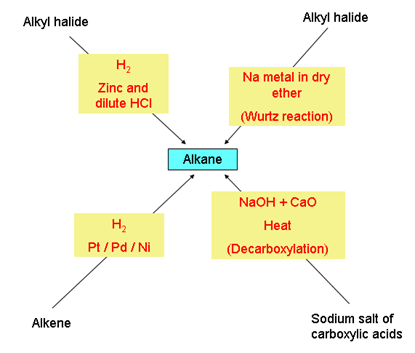
What happens when H2 is added to an alkene?
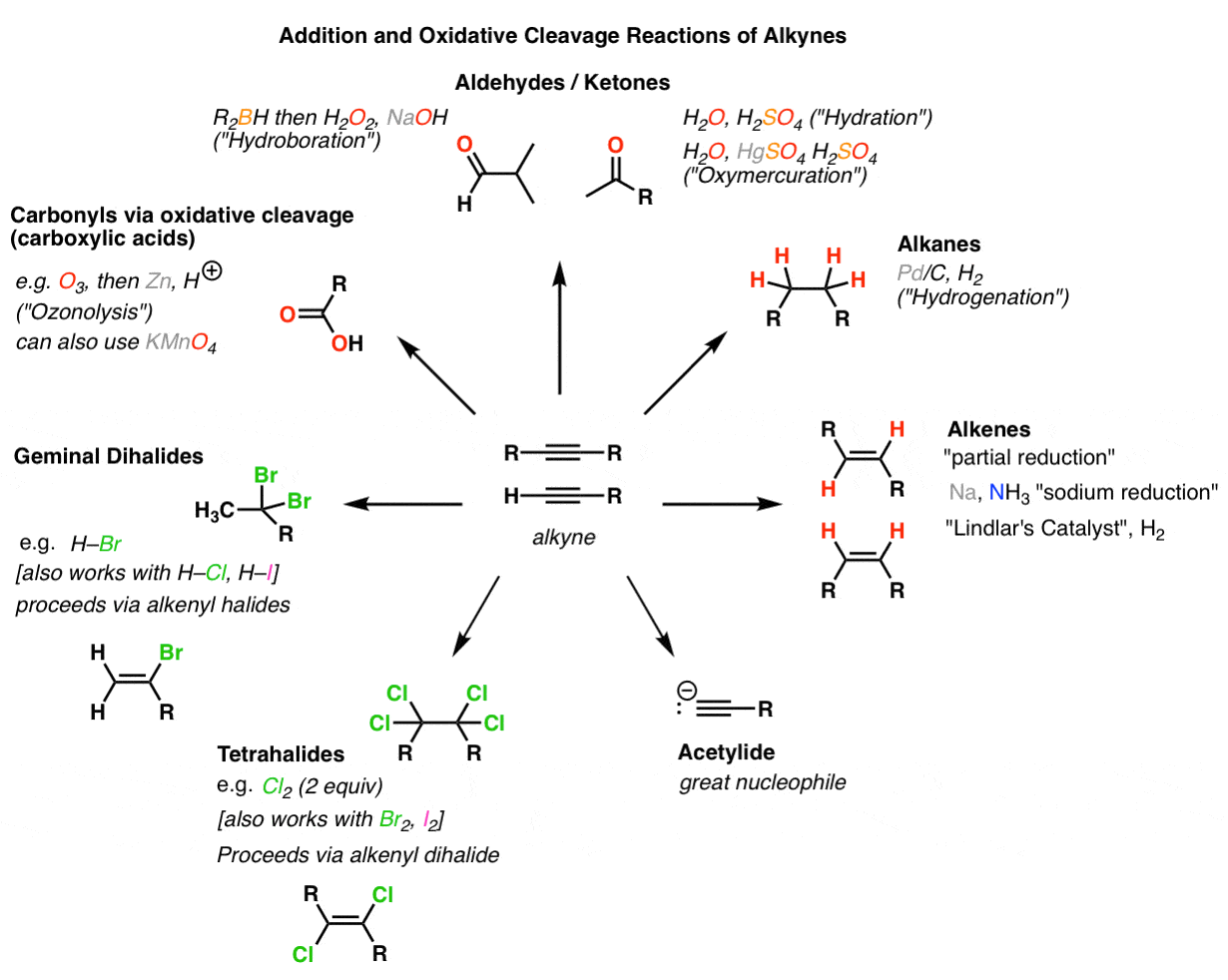
Hydrogenation: Addition of H2 across the p-bond of an alkene to give an alkane. This is a reduction. The reaction uses H2 and a precious metal catalyst. The catalysts is not soluble in the reaction media, thus this process is referred to as a heterogenous catalysis. The catalyst assists in breaking the p-bond of the alkene and the H-H s-bond.

Alkene Reactions

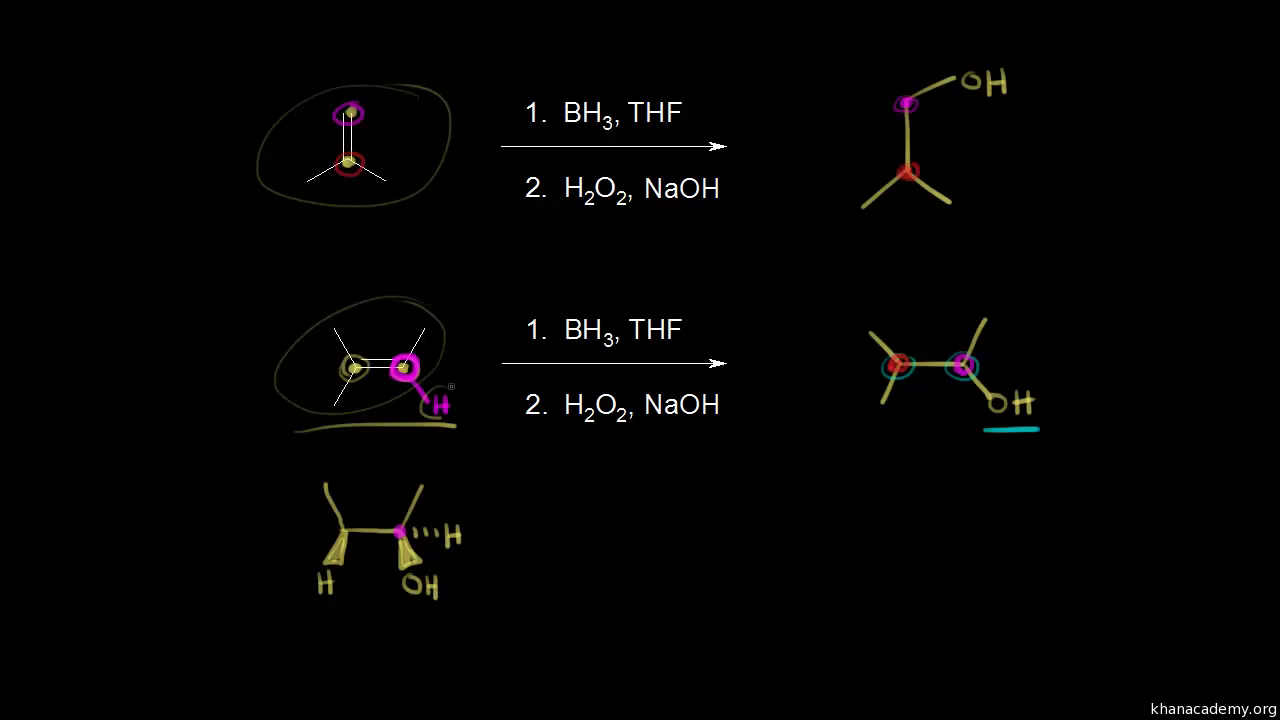
Hydroboration-oxidation Alkenes and Alkynes Organic chemistry Khan Academy

Hydroboration Oxidation Mechanism of Alkenes
|
Reactions of Alkenes
Reactions of Alkenes. Product. Type of Reaction (name). Reaction Conditions. Regiochemistry H2O2 NaOH |
|
Summary of Alkene Reactions Ch. 8. Memorize Reaction
H. CH3. OH. H. BH2. H. CH3. BH2. H2O2 NaOH. Notes a. concerted addition of B-H across C=C. -explains the cis stereochemistry b. the B-H addition is Markovnikov |
|
1 Chapter 7: Alkenes: Reactions and Synthesis Electrophilic
Addition of Halogens (X2) to Alkenes: 12-dihalides 2) H2O2 |
|
ORGANIC CHEMISTRY I – PRACTICE EXERCISE Elimination
ORGANIC CHEMISTRY I – PRACTICE EXERCISE. Elimination Reactions and Alkene Synthesis NaOH. 20) Based on Saytzeff's rule select the most stable alkene. |
|
Reactions of Alkenes and Alkynes
We discuss this alkene reaction in Chapter 16. 5.2 What Is a Reaction Mechanism? 5.1 What Are the Characteristic Reactions of Alkenes? ... 2) NaOH H2O2 ... |
|
Practice Set Answer Keys Organic Chemistry I Table of Contents
Test 3 PS3: Test 3 Alkene Reactions Practice NaOH. H2O. 8. 9. 10. 11. CH3CH2OH + NaOH. CH3CH2ONa + H2O. CH3CH2NHLi + CH3OH. CH3CH2NH2 + CH3OLi. |
|
Reactions of Alkenes
Electrophilic addition is probably the most common reaction of alkenes. The alkene reacts with conc. sulfuric acid to give an alkyl hydrogen sulfate ... |
|
1 Only typical first year organic reactions are listed below following
You will find alkene and alkyne reactions at the very end. 1C starting compound = CH4. Br2 h?. CH4. H3C. Br. H3C. Br. NaOH. |
|
Many of the reactions below have competing reactions. We mostly
We mostly focus on the dominant reaction in a synthesis. Organic Reaction Review. 4. NaOH. NaOH. Br. Br. OH. SN2 > E2 only E2 alcohol alkene. NaOH. NaOH. |
|
Important Reactions from Chem2310 I. Reactions of Alkenes and
I. Reactions of Alkenes and Alkynes. A. Electrophilic addition reactions: 1. Hydro-halo-addition: ionic mechanism 2) NaBH4 NaOH. |
|
Reactions of Alkenes
Reactions of Alkenes Product Type of Reaction (name) Reaction Conditions Regiochemistry H2O2, NaOH, H2O Anti-Markovnikov Syn Stereochemistry |
|
Reactions of Alkenes
Reactions of Alkenes Product Type of Reaction (name) Reaction Conditions Regiochemistry H2O2, NaOH, H2O Anti-Markovnikov Syn Stereochemistry |
|
Summary of Alkene Reactions, Ch 8 Memorize Reaction
H CH3 OH H BH2 H CH3 BH2 H2O2, NaOH Notes a concerted addition of B-H across C=C -explains the cis stereochemistry b the B-H addition is |
|
Reactions of Alkenes
addition product Electrophilic addition is probably the most common reaction of alkenes The alkene reacts with conc sulfuric acid to give an alkyl hydrogen sulfate, which then in turn is hydrolyzed to give the CH(Hal)3, NaOH N/A Syn |
|
1 Only typical first year organic reactions are listed below, following
You will find alkene and alkyne reactions at the very end 1C starting NaOH 2 CH3Br S CH3 SH 1 NaN3 2 LiAlH4 3 workup H3C Br H3C NH2 7 8 9 |
|
Reactions of Alkenes - CUTM Courseware
of alkene addition reactions that you have not seen before You will also find it easier to of the following reactions (a) cyclohexene + CHCl3, 50 NaOH>H2O |
|
Chem 343 – Organic Reactions Chapter 5 & 7 Prepared by José
Chem 343 – Organic Reactions Chapter 5 7 Prepared Alkene Reactions #8: Hydroboration-‐Oxidation Mechanism 2) H2O2 / NaOH OH H BH2 BH2 |
|
Lecture 23 - Reactions of Alkenes - Addition Reactions
Mechanism of Hydroboration - Concerted Syn Addition Larger B adds to less substituted end of the alkene Step 2 Oxidation H O O H + NaOH H O O Na |
|
Reactions of Alkenes and Alkynes
We discuss this alkene reaction in Chapter 16 The most characteristic reaction of alkenes is addition to the carbon–carbon double bond 2) NaOH, H2O2 |








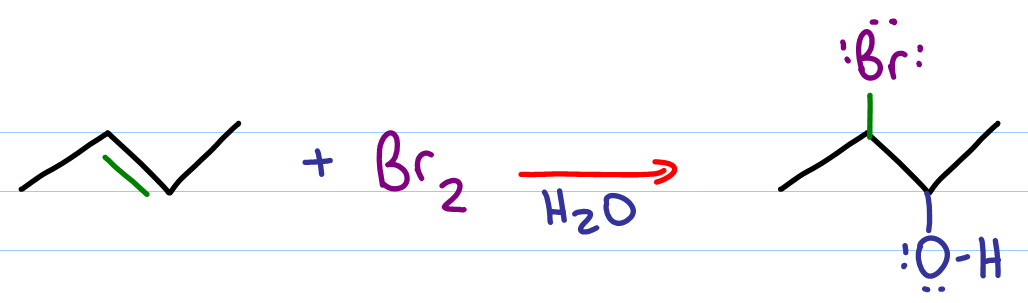

![Organic Reagents - [PDF Document] Organic Reagents - [PDF Document]](https://online.fliphtml5.com/nroaj/eaxq/files/large/252.jpg?1588695985)




![Alkenes PDF - [PDF Document] Alkenes PDF - [PDF Document]](https://toppr-doubts-media.s3.amazonaws.com/images/1546862/36d7314f-c645-4b16-9701-8ea346794789.jpg)