aman dhattarwal chemistry notes aldehydes ketones and carboxylic acid
How will you distinguish between aldehyde and ketone and carboxylic acid?
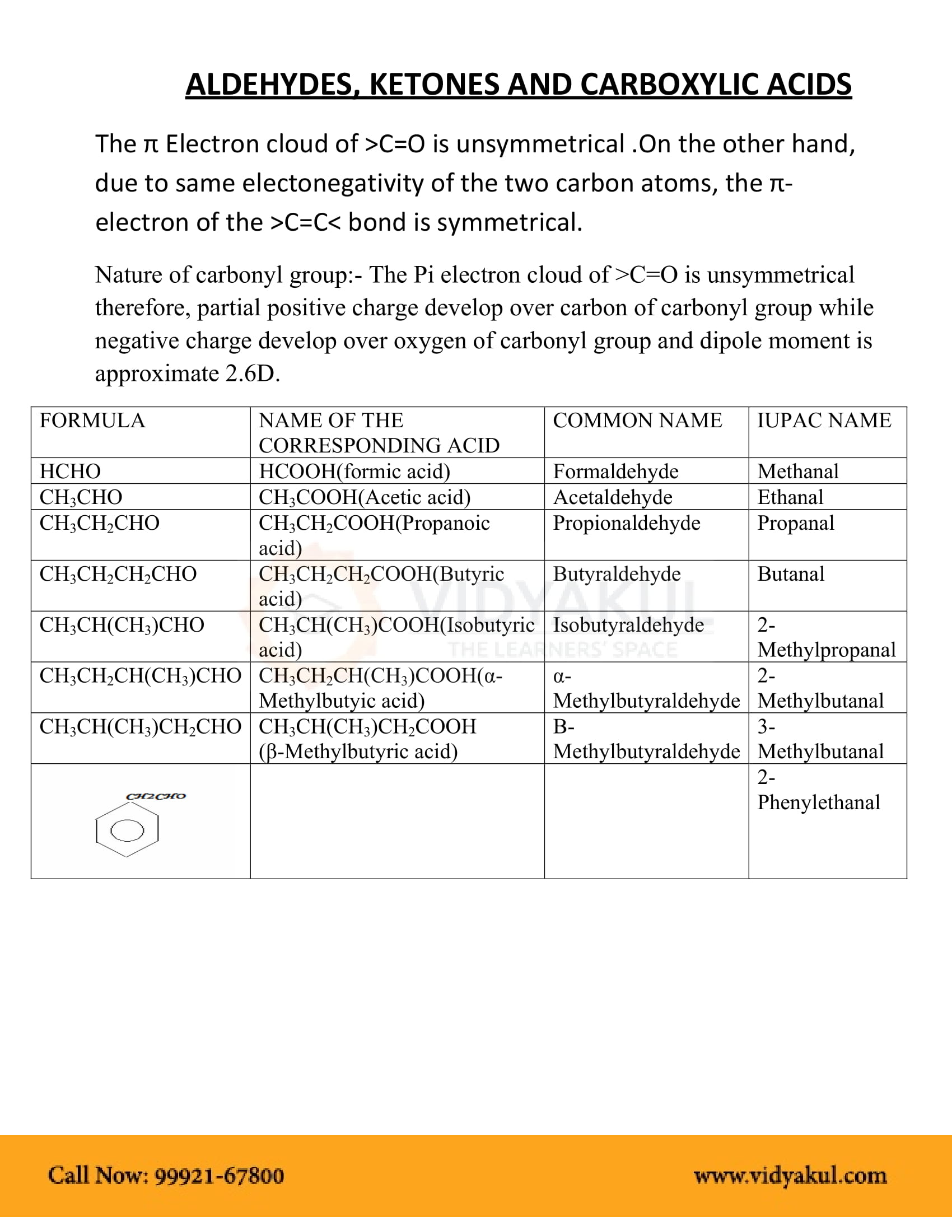
The carbonyl group, a carbon-oxygen double bond, is the key structure in these classes of organic molecules: Aldehydes contain at least one hydrogen atom attached to the carbonyl carbon atom, ketones contain two carbon groups attached to the carbonyl carbon atom, carboxylic acids contain a hydroxyl group attached to
What is the structure of aldehydes and ketones?
In an aldehyde, the carbonyl group is bonded to at least one hydrogen atom.
In a ketone, the carbonyl group is bonded to two carbon atoms: As text, an aldehyde group is represented as –CHO; a ketone is represented as –C(O)– or –CO–.
|
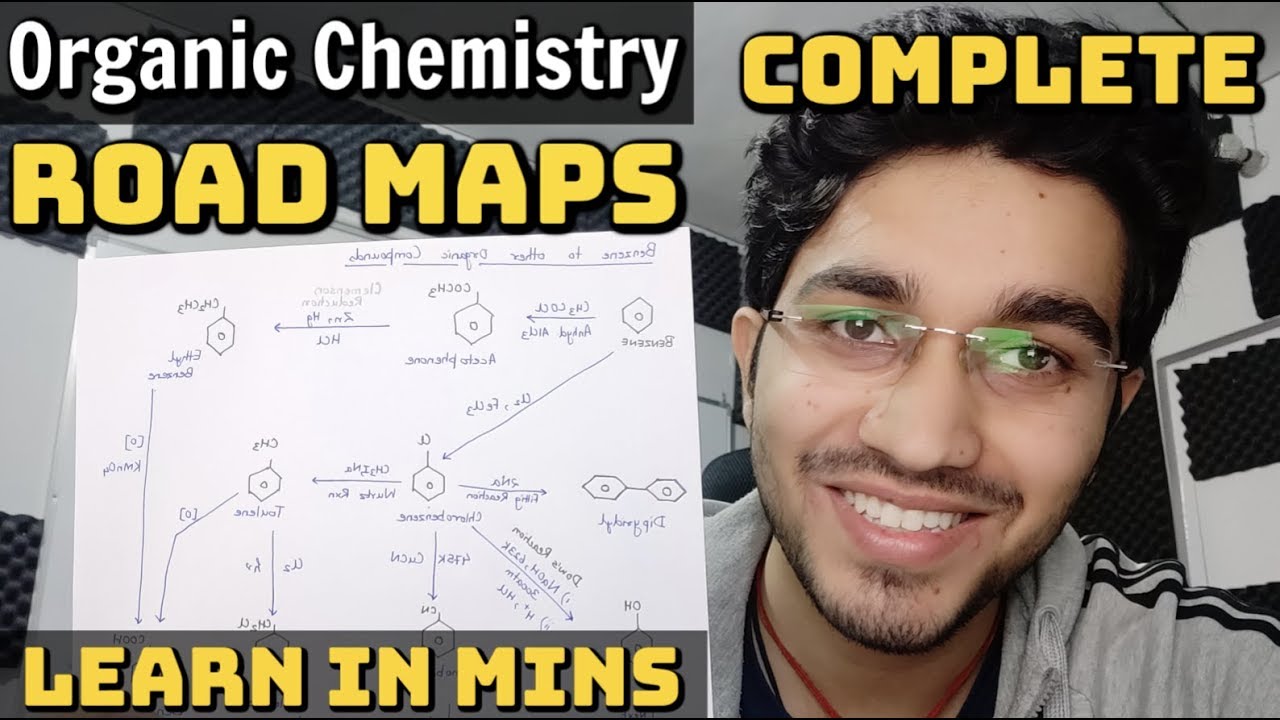
Class 12 Organic Name Reactions
Aldehydes without ?-hydrogen atom undergo self-oxidation and reduction reaction when prepared with concentrated alkali. Aman Dhattarwal. Page 6. Kolbe |
|
Aldehydes, Ketones and Carboxylic Acids - Ncert Help
Chemistry Notes for class 12 Chapter 12 Aldehydes, Ketones and Carboxylic Acids In aldehydes, the carbonyl group ( )C=O) is bonded to carbon and |
|
Class 12 Organic Name Reactions
Aldehydes without α-hydrogen atom undergo self-oxidation and reduction reaction when prepared with concentrated alkali Aman Dhattarwal Page 6 Kolbe |


















![PDF] Download MTG 30 years Chapterwise Solutions of NEET Chemistry PDF] Download MTG 30 years Chapterwise Solutions of NEET Chemistry](https://i.ytimg.com/vi/gKp1aM42VvI/maxresdefault.jpg)