crystal field theory pdf
|
US05CCHE22 UNIT-2 Crystal Field Theory-I
Crystal field theory is a powerful and simple method of understanding and correlating properties of ionic crystals that arise primarily from the presence of |
|
Outline of crystal field theory
2 1 Introduction Crystal field theory describes the origins and consequences of interactions of the surroundings on the orbital energy levels of a |
|
Crystal field theory (cFT)
CRYSTAL FIELD THEORY (CFT) This theory advanced by Bethe and Van Vleck_was originally applto mainly to ionic crystals and is therefore called crystal field |
|
CRYSTAL FIELD THEORY (CFT)
This model was based on a purely interaction between the ligands and the metal ion in the complexes with various geometries like octahedral tetrahedral square |
|
Crystal Field Theory (CFT)
19 déc 2018 · As originally developed crystal field theory was used to describe the electronic structure of metal ions in crystals where they are |
|
Crystal field Theory (CFT)
Crystal field Theory (CFT): Developed by H Bethe and van Vleck Originally crystal field theory was applied to transition metal ions in ionic crystals |
|
Crystal Field Theory
Advanced by Bethe and Van Velck and originally applied to explain the colour and magnetic properties of some ionic crystal |
|
Crystal Field Theory
Crystal field theory (CFT) is a bonding model that explains many important properties of transition-metal complexes including their colors magnetism |
|
Crystal Field Theory
Crystal Field Theory (Text : JD Lee; pp 204-222) •This theory (CFT) largely replaced VB Theory for interpreting the chemistry of coordination compounds •It |
|
6 Crystal Field Theory (CFT)
Brethe and Van Vlick have been proposed a theory to explain the bonding in the ionic crystals which is called as Crystal Field Theory (CFT) Initially this |
Who introduced crystal field theory?
The crystal field theory (CFT) was developed for crystalline solids by the physicist Hans Bethe in 1929.
What is the crystal field group theory?
In molecular physics, crystal field theory (CFT) describes the breaking of degeneracies of electron orbital states, usually d or f orbitals, due to a static electric field produced by a surrounding charge distribution (anion neighbors).
What is the crystal field theory explained?
Crystal field theory (CFT) is a bonding model that explains many important properties of transition-metal complexes, including their colors, magnetism, structures, stability, and reactivity.
The central assumption of CFT is that metal–ligand interactions are purely electrostatic in nature.30 jui. 2023Crystal field theory says nothing about the orbitals of ligands.
It only focuses on the metal orbital.
It does not account for why some ligands split the d orbitals greatly and some ligands split the d orbitals shortly.
|
Crystal Field Theory
The splitting of d orbital energies and its consequences are at the heart of crystal field theory. Page 5. 5. CFT-Octahedral Complexes. •For the Oh |
|
Crystal Field Theory (CFT)
19-Dec-2018 As originally developed crystal field theory was used to describe the electronic structure of metal ions in crystals |
|
Untitled
Crystal field Theory (CFT):. Developed by H. Bethe and van. Vleck. Originally crystal field theory was applied to transition metal ions in ionic crystals. |
|
MSCCH-17/18/19 Course Code-CHE-501 Unit- 6 Crystal Field
CRYSTAL FIELD THEORY (CFT). In view of the weaknesses of Valence Bond Theory (VBT) an alternative bonding model was applied to transition metal complexes. |
|
Crystal Field Theory
❖ Electric field generated by the ligands influences the distribution of electrons in the metal ions i.e. d-orbital splitting. ❖ The bonding between the |
|
CRYSTAL FIELD THEORY (CFT)
Crystal Field Theory was proposed by the physicist Hans Bethe in 1929 to describe the bonding in coordination complexes and to rationalize and predict some |
|
Coordination Compounds
(vi) It does not distinguish between weak and strong ligands. The crystal field theory (CFT) is an electrostatic model which considers the metal-ligand bond to |
|
CRYSTAL FIELD THEORY (CFT) This theory advanced by Bethe
7) Different crystal fields will have different effects on the relative energies of the five d orbitals. Crystal field splitting of d-orbitals: The out come of |
|
Crystal Field Theory (CFT)
Crystal Field Theory (CFT). The bonding of transition metal complexes can be explained by two approaches: crystal field theory and molecular orbital theory. |
|
Crystal Field Theory
Crystal field theory. (CFT) is a bonding model that explains many important properties of transition-metal complexes including their colors |
|
Crystal Field Theory
The splitting of d orbital energies and its consequences are at the heart of crystal field theory. Page 5. 5. CFT-Octahedral Complexes. •For the Oh |
|
Crystal Field Theory (CFT)
Dec 19 2018 As originally developed |
|
CRYSTAL FIELD THEORY (CFT)
Crystal Field Theory was proposed by the physicist Hans Bethe in 1929 to describe the bonding in coordination complexes and to rationalize and predict some |
|
Topic: Crystal Field Theory (CFT)
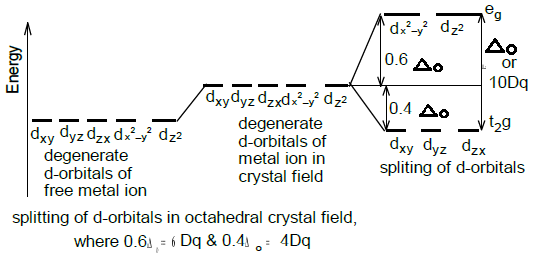
The crystal field splitting is measured in terms of energy difference between t2g and eg orbital and is denoted by a symbol ?o . It is generally measured |
|
Chemistry Notes for class 12 Chapter 9 Coordination Compounds .pdf
By using spectroscopic data for a number of coordination compounds having the same metal ions but different ligand |
|
Coordination Compounds
(vi) It does not distinguish between weak and strong ligands. The crystal field theory (CFT) is an electrostatic model which considers the metal-ligand bond to |
|
ATOICV1-7-1-Limitation-of-Crystal-Field-Theory.pdf
The main drawback of the crystal field theory is that it does not consider the covalent character in metal-ligand bonding at all. It treats the metal-ligand |
|
Crystal Field Theory
Crystal field theory. (CFT) is a bonding model that explains many important properties of transition-metal complexes including their colors |
|
Crystal Field Splitting in an Octahedral Field
as e. The crystal field splitting in the tetrahedral field is intrinsically smaller than in the octahedral field For most purposes the relationship may be |
|
B.Sc. III YEAR INORGANIC CHEMISTRY-III
2.4 AN ELEMENTARY IDEA OF CRYSTAL FIELD THEORY. In view of the above weaknesses an alternative bonding model was applied to transition metal complexes. |
|
CRYSTAL FIELD THEORY (CFT)
Too much stress has been given on metal ion while the importants of ligands is not properly addressed Page 3 Crystal Field Theory Sem-IV Gen (1st Part) Crystal |
|
Crystal Field Theory
The splitting of d orbital energies and its consequences are at the heart of crystal field theory Page 5 5 CFT-Octahedral Complexes •For the Oh |
|
Crystal Field Theory (CFT) In Detail
Crystal Field Theory (CFT) In Detail CFT provides a simple model for d orbital splitting: in an 'electrical field' created by the ligand (donor) electrons: |
|
CRYSTAL FIELD THEORY Hans Bethe (1929) and Van Vleck (1935
CRYSTAL FIELD THEORY Hans Bethe (1929) and Van Vleck (1935) Failures of VBT 1 Tetrahedral or Square planar ?? 2 Distortions in complexes – No 3 |
|
Crystal Field Theory - Banwarilal Bhalotia College
Splitting of five d-orbitals in Octahedral Ligand Field: 6 For weak ligand field: crystal field splitting low; Δ(oct) < Pairing Energy (P); results high spin metal |
|
Chapter 21 d-block metal chemistry: coordination complexes
coordination complexes Bonding: valence bond, crystal field theory, MO Spectrochemical series Crystal field stabilization energy (CFSE) Electronic Spectra |
|
Outline of crystal field theory - HIGP
Crystal field theory describes the origins and consequences of interactions of the surroundings on the orbital energy levels of a transition metal ion These |
|
Crystal (ligand) field theory
1 Title page Crystal (ligand) field theory Literature Crystal field Schrödinger Splitting of states for electron configuration d2 – sphere symmetry 1 Hund 2 |
|
Limitation of Crystal Field Theory - Dalal Institute
Therefore, the main limitations of crystal field theory can be concluded only after benefits like downloading any PDF document for your personal preview |
|
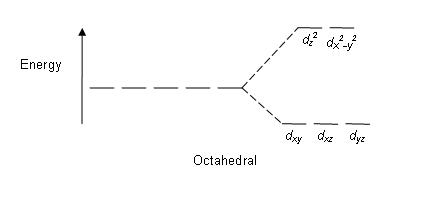
Crystal Field Splitting in an Octahedral Field
Crystal Field Splitting in an Octahedral Field eg Energy 3/5 o o t2g 2/5 o e g - The higher energy set of orbitals (d z2 and d x2-y2 ) t 2g - The lower energy set |