entropy change in non ideal solution
|
Chapter 5: The Thermodynamic Description of Mixtures
PHYSICAL CHEMISTRY: QUANTA MATTER |
|
Equilibrium and Non-equilibrium Thermodynamics: Concept of
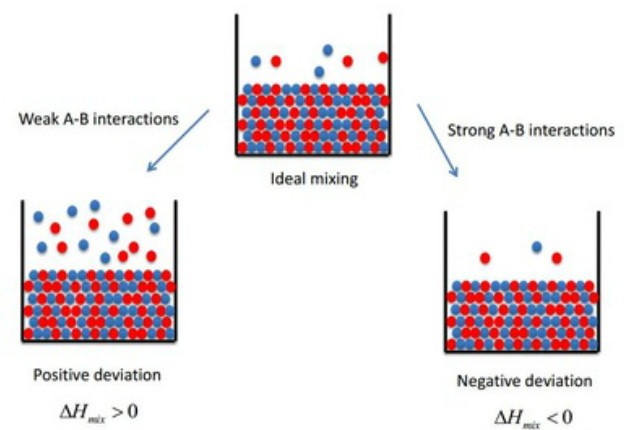
A non-ideal solution is a solution that does not abide to the rules of an entropy change ?S for a chemical or physical transformation approaches zero. |
|
Chemical Engineering Thermodynamics II
Example 3.5-4: A refrigeration cycle with nonideal compressor 3-35 thermodynamics we can predict the amount of energy needed to change a system from an. |
|
Effect of Chemical Composition on Enthalpy of Evaporation and
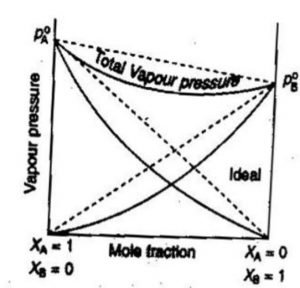
Keyword: organic liquid mixtures ideal and non-ideal solutions temperature dependence of the vapor pressure to the change of enthalpy of the phase. |
|
Entropy analyses of four familiar processes
from the enthalpy change of the thermal reservoir.2 AStot is often designated as ASanv also illustrate the use of non ideal solution theory. For the. |
|
Thermodynamic modelling of solid solutions
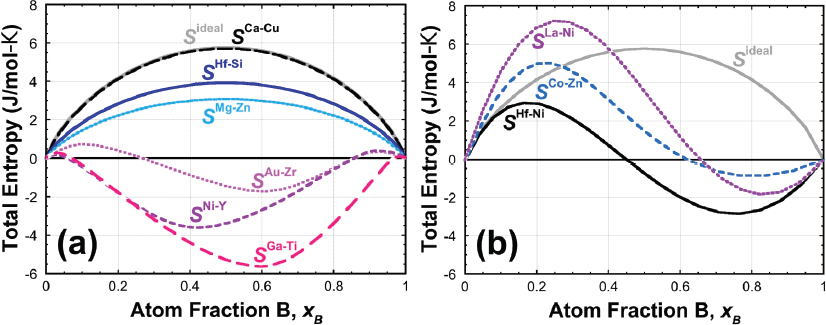
entropy of mixing namely that of an ideal solution as given by Equation 14 which is due to non-linear change of vibrational properties as a function of ... |
|
1st Year Thermodynamic Lectures Dr Mark R. Wormald
Behavoiur of non-ideal solutions (and solutes). Figures include the loss of entropy associated with the gas as well as the entropy change of the. |
|
Mole fraction of component A = xA Mass Fraction of component A
So the chemical potential must drop in the solution for a solution to exist. Ideal gasses only have entropy so entropy drives mixing in this case. |
|
Download Free Thermodynamics Ideal Solution 7 Edition Copy
discusses the problems of non-ideal solutions and the concept of activity and how is entropy defined in thermodynamics |
|
Intracellular spatial organisation: The effects of non-ideal solutions
C.2 Form of the reaction rates in the non-ideal solution theory . According to the second law of thermodynamics the entropy of the universe always ... |
|
(Lec 3 Solution Models)
energy on mixing even when there are no enthalpy changes The free Ideal Solution An ideal solution is one in which the atoms are, at equilibrium, dis- |
|
Chapter 5: The Thermodynamic Description of Mixtures
For a non-ideal solution, all partial molar quantities differ from the corresponding molar quantities of a solution or mixture varies with changes in molar composition at constant T, P Thermodynamics of mixing, Entropy and Enthalpy |
|
Thermodynamic modelling of solid solutions - Geosciences
entropy of mixing, namely that of an ideal solution as given by Equation 14, we which is due to non-linear change of vibrational properties as a function of |
|
LECTURE NOTES - VSSUT
Efficiency of Heat Engine, Entropy Changes in Reversible and Irreversible The solution which deviate from ideal behaviour are called non ideal solution or |
|
54 Liquid Mixtures
the enthalpy of mixing is non-zero Page 2 (a) Ideal solutions • The Gibbs energy of mixing of two liquids to form an ideal solution is calculated in the same If the enthalpy change is large and positive or if the entropy change is adverse, the |
|
Binary Solutions
Entropy of formation and Gibbs free energy of an ideal solution ➢ Regular solutions: Heat of formation of a solution energy change upon mixing is only due to the change in For a nonideal solution we have to take into account heat of |
|
Regular solutions - Faculty Washington - University of Washington
6 août 2014 · The simplest non-ideal solution model that works beyond the Henry's Law model is the regular If mixing is not random then the entropy will deviate from this expression • Ideal solution not change much upon mixing |
|
SOLUTIONS AND SOLUTION MODELS
A few non ideal solution models of general interest for multicomponent which is the result found in l 4d for the entropy change due to isothermal expansion |
|
Solution Behavior
Non-ideal solutions Auming Raoultian behavior, what are the heat and entropy changes in What is the free energy change in the system due to mixing ? |