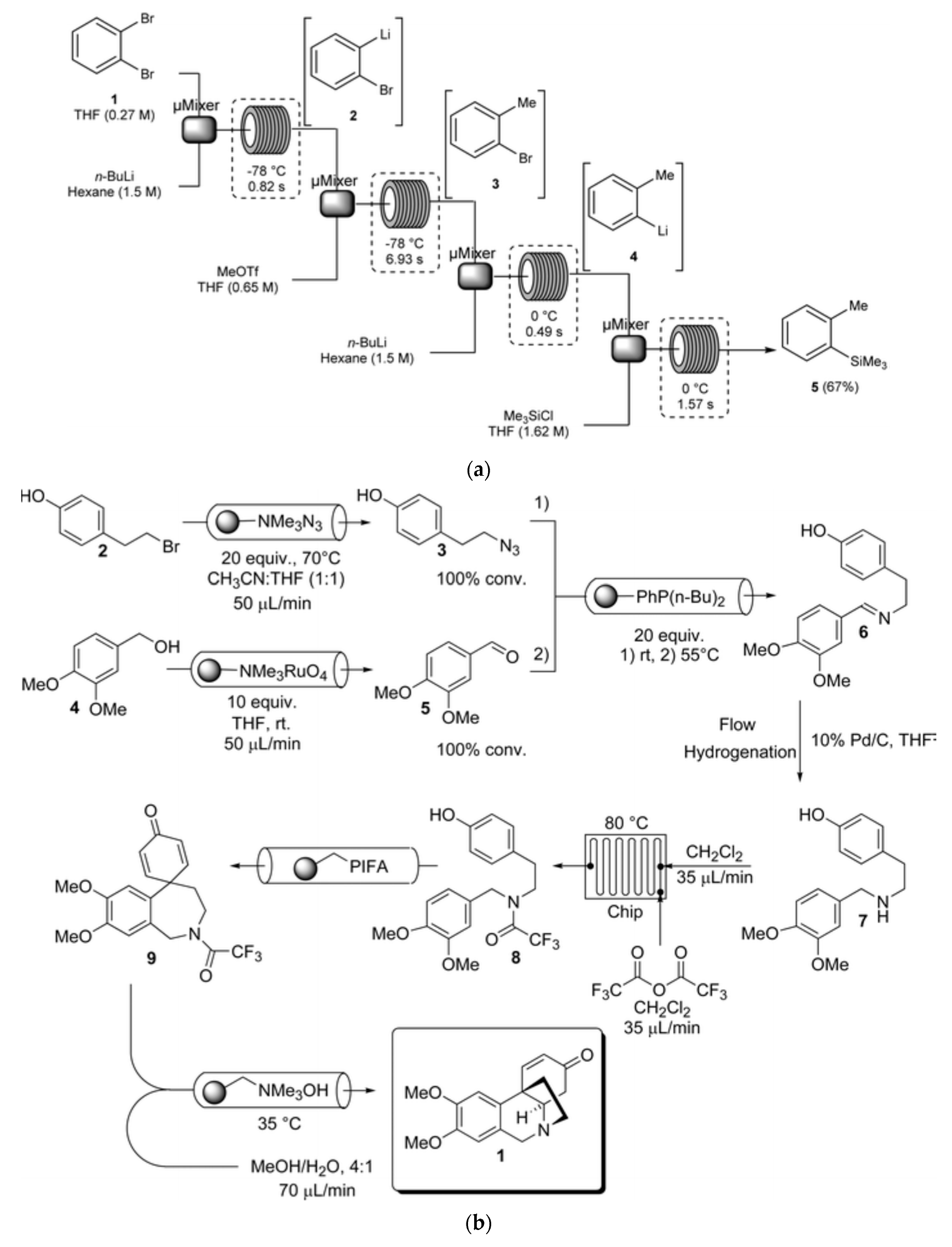
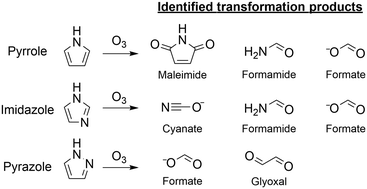
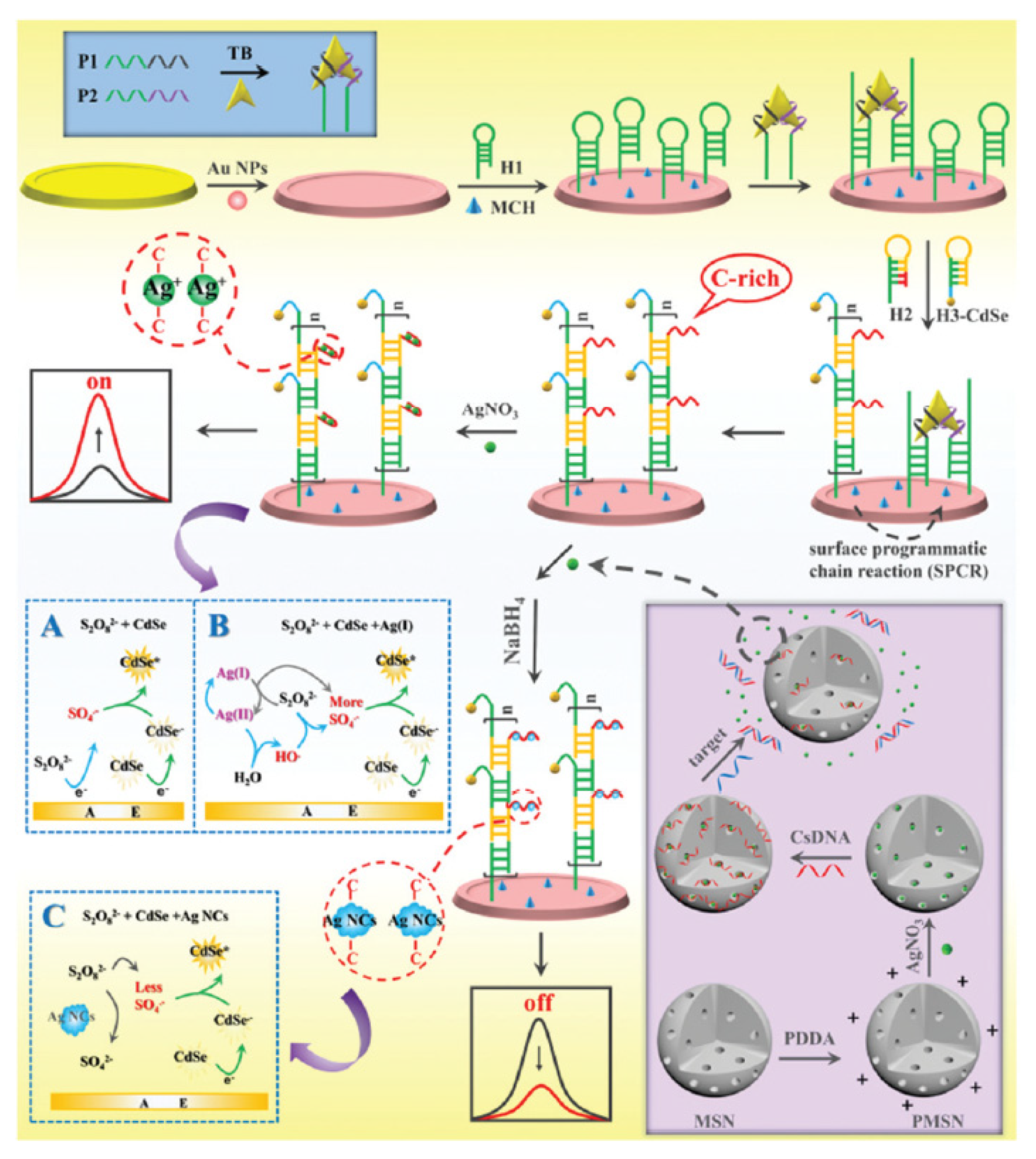
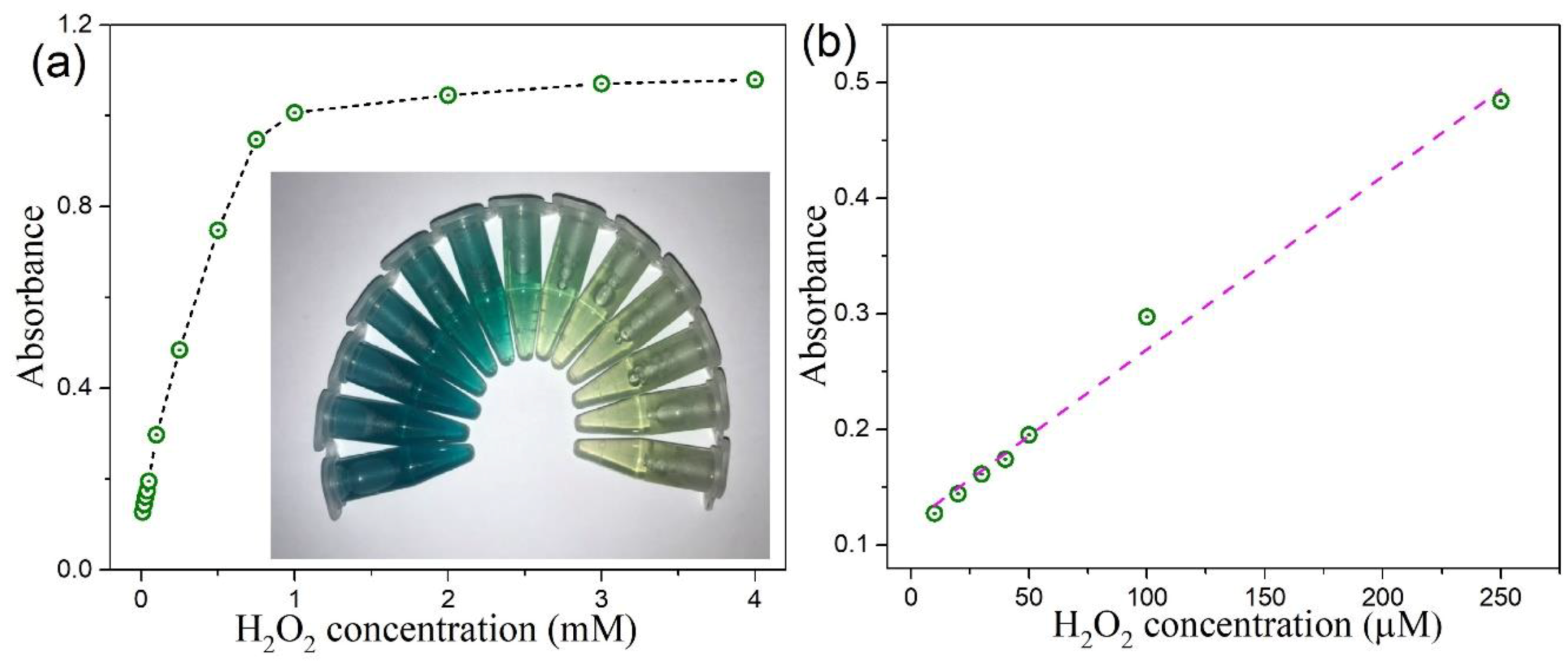
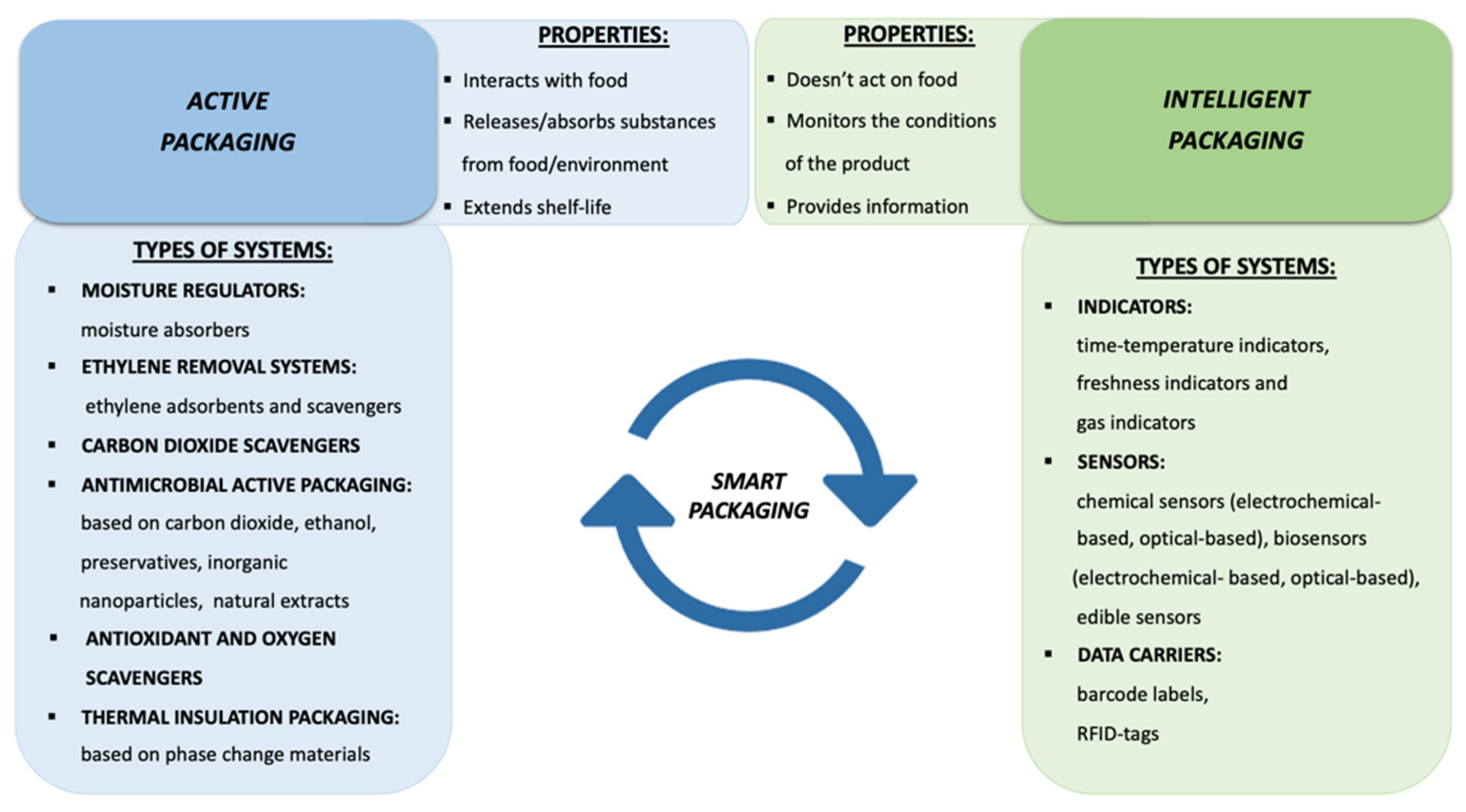
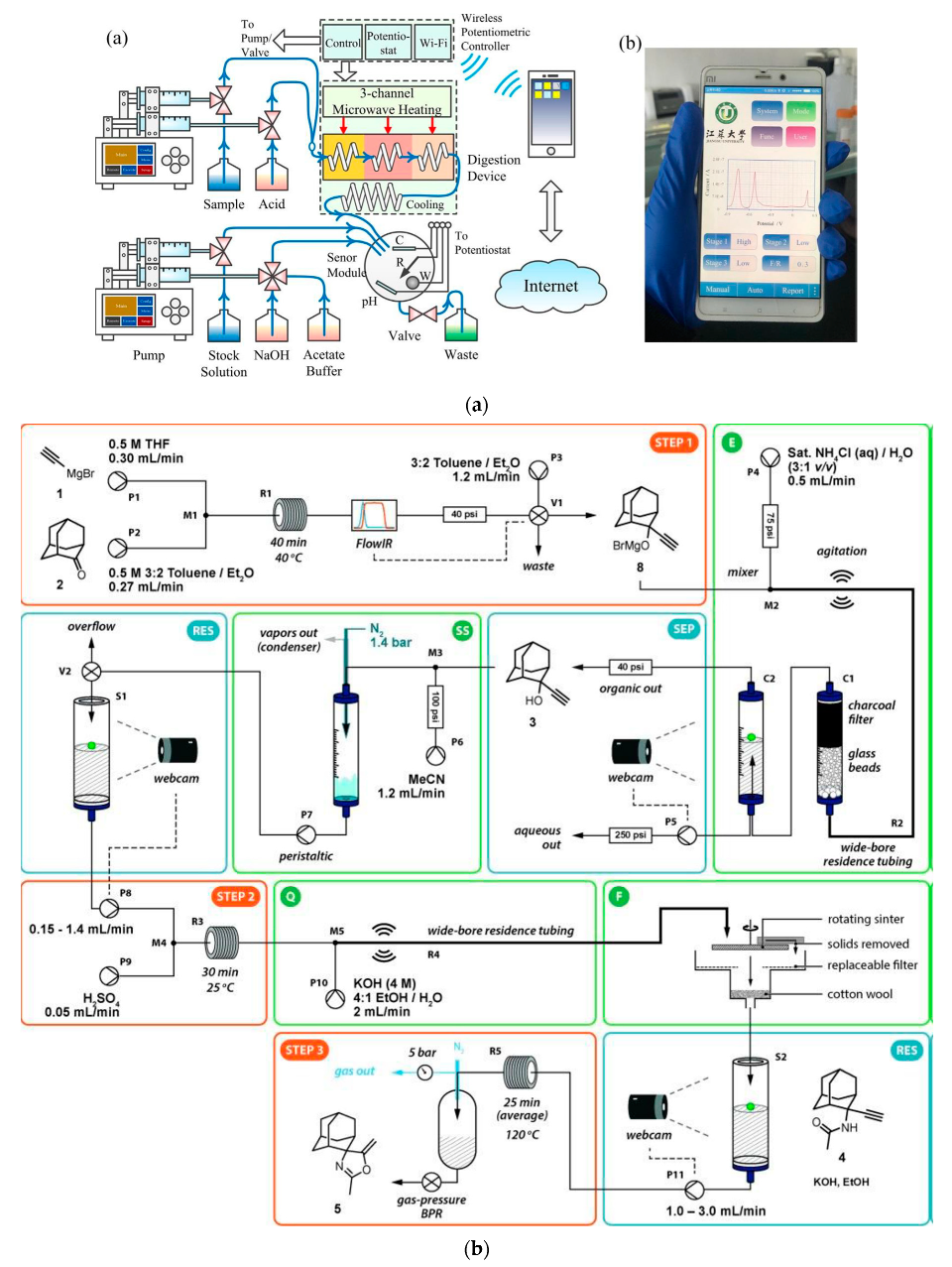
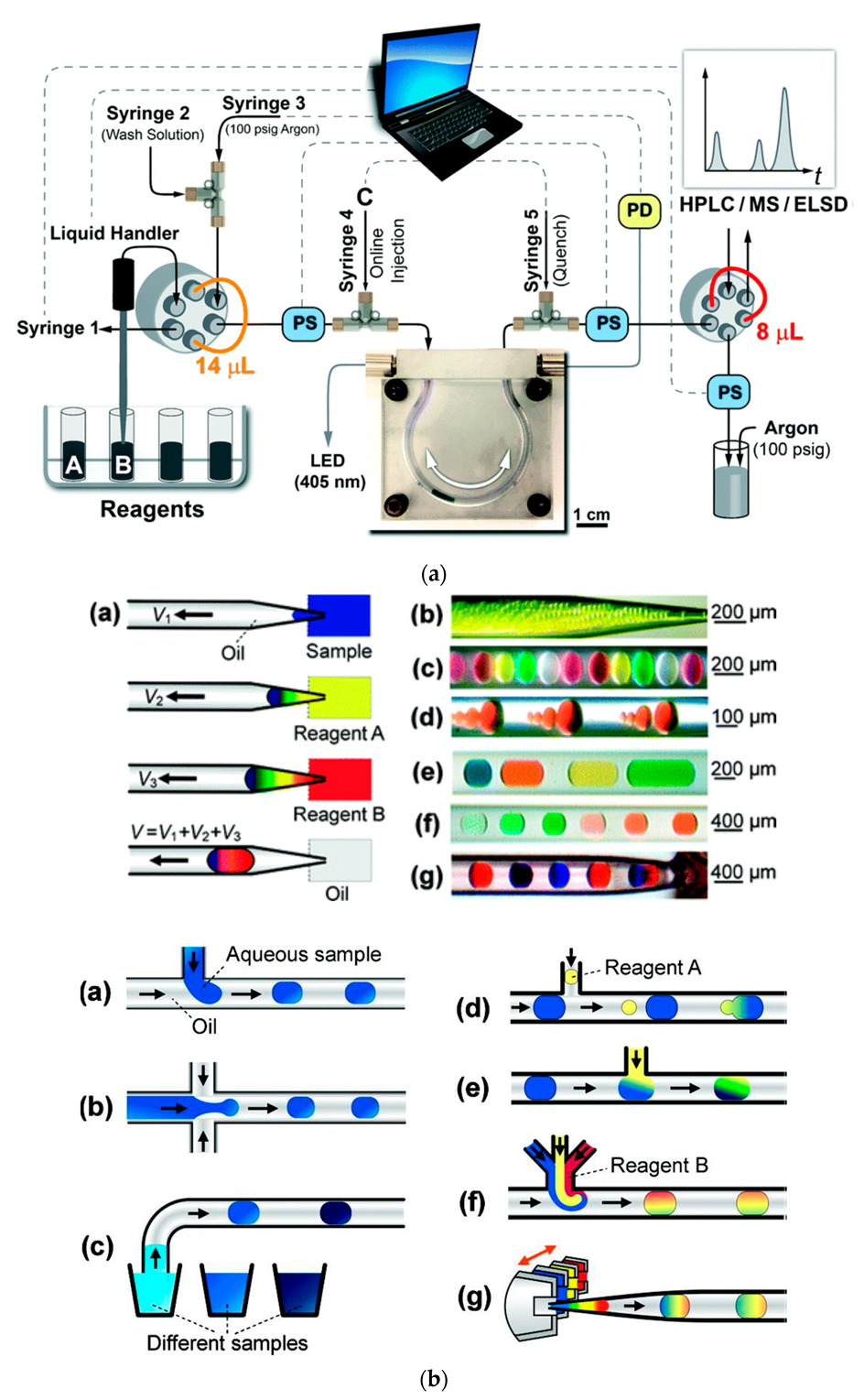
the chemiluminescence reactions in experiment 9 belong to which reaction type?
What type of reaction is chemiluminescence?
Chemiluminescence is the chemical production of light through exothermic oxidation reactions.
Several compounds exhibit chemoluminescence, although luminol is widely applied for sensor applications.Chemiluminescence in aqueous system is mainly caused by redox reactions.
Is chemiluminescence a combustion reaction?
Chemiluminescence imaging is one of the most widely used optical diagnostic techniques for combustion.
Chemical reactions in the flame front create regions of high local heat release, species are excited and emit light.
Is chemiluminescence a spontaneous reaction?
Chemiluminescence (CL) is the spontaneous emission of light from an electronically excited state of a species produced by a chemical reaction [1].17 déc. 2021
|
Chemiluminescence Demonstration Illustrating Principles of Ester
of the relevant chemical reactions have also been described. (4–6). In this experiment |
|
Chemiluminescence Demonstration Illustrating Principles of Ester
luminescence reaction to demonstrate how the pKa of the leaving group and nature of the catalyst affect the rate of ester hydrolysis reactions. Experimental |
|
INTRODUCTION
reactions performed in undergraduate labs are chemiluminescent In this sample experiment a three-step chemiluminescent reaction. ORGANIC. |
|
John C. Polanyi - Nobel Lecture
methods belong to the modern period of reaction dynamics both have clear antecedents in earlier times. Infrared chemiluminescence derives from the presence |
|
Chemiluminescence involving acidic and ambident ion light emitters
These reactions generate light emitters that are acids or ambident ions; most however |
|
An introduction to chemiluminescence methods for lubricant
Chemiluminescence (CL) lightproduced as a result of a chemical reaction Hydrocarbon oxidation is covered in references [9] and. |
|
Electrogenerated chemiluminescence: from materials to sensing
31 mai 2017 Les réactions ECL se classent principalement en 2 grandes ... Electrogenerated chemiluminescence (ECL) is a type of chemiluminescence ... |
|
Efficiency of Electron Transfer Initiated Chemiluminescence
16 mai 2013 nylimidazole) (9) and the peroxyoxalate reaction first described ... oxidized or reduced producing intermediates that eventually react. |
|
P) + CH3OH reaction as an efficient loss process for gas-phase
no earlier experimental studies exist of the gas-phase reactions of aliphatic reaction using the MESMER (Master Equation Solver for Multi Energy Well ... |
|
Chemistry_lab_activity_9-student_sheetspdf - PS21
luminescence reaction Generally the reactions use hydrogen peroxide as an oxidant; luminol (Equation 1), oxalates (Equation 2), or oxalyl chloride as |
|
An introduction to chemiluminescence methods for - GovInfo
Chemiluminescence (CL), lightproduced as a result of a chemical reaction is a valuable Hydrocarbon oxidation is covered in references [9] and [16] 2 rate constants refer to the reactions in the original sequence (1 to 7) General from such temperature variation experiments were used to obtain activation energies |
|
Chemiluminescence with Luminol - NC State University
reactions with luminol Demonstration A this reaction is performed on a large scale 1 Make fresh 0 5 NaClO (9 mL of commercial bleach in 91 mL water) 2 Chill both chemiluminescent reactions of luminol are oxidations occurring under basic conditions as shown in the general equation below: NH NH O O NH2 |
|
Illuminating Latent Blood - Abacus Diagnostics
4 7 Morphoanalytical Study- Fixative/Shear Thinning Agent from different chemiluminescent reactions depending on the type of reaction and reaction conditions (4) Luminol plant and bacterial proteins and pigments and lastly, enzymes belonging to the oxidoreductases of hydrogen peroxide to the blood stain (8,9) |
|
John C Polanyi - Nobel Lecture - Nobel Prize
reactions, and some examples of the application of these concepts We are, methods belong to the modern period of reaction dynamics, both have clear antecedents in (“Proposal for an Infrared Laser Dependent on Vibrational Excitation”) [9] we pointed to Infrared chemiluminescence experiments in flow tubes have |
|
Chemical Reactions - Smithsonian Science Education Center
All of the chemicals in this module belong to the green category, which is the These activities will help them understand the signs that a chemical reaction has taken place type of atom is conserved, and thus the mass does not change Science Starts With a Question: Chemical Reactions 9 Reaction of Baking Soda |
|
Light Stick Chemiluminescence - Science Made Fun
For this experiment, you will find out how temperature affects the chemical You will observe how the light sticks react when placed in three different 9 Let them sit in the water, and watch the light sticks 10 Notice how quickly or slowly the chemical reaction releases a type of energy that causes a colorful luminescent |