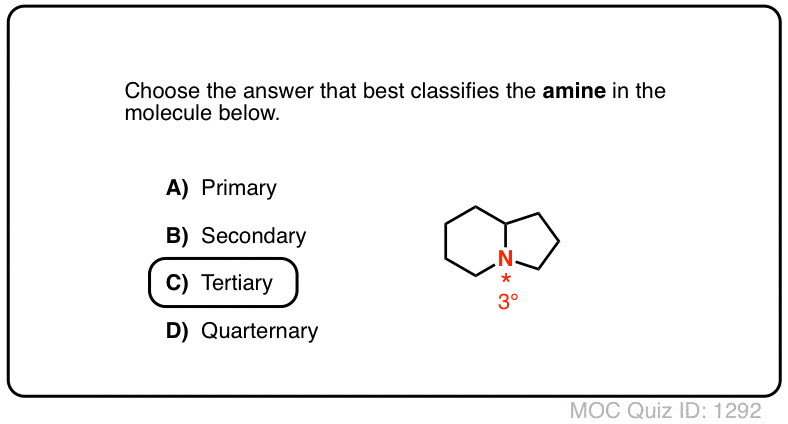
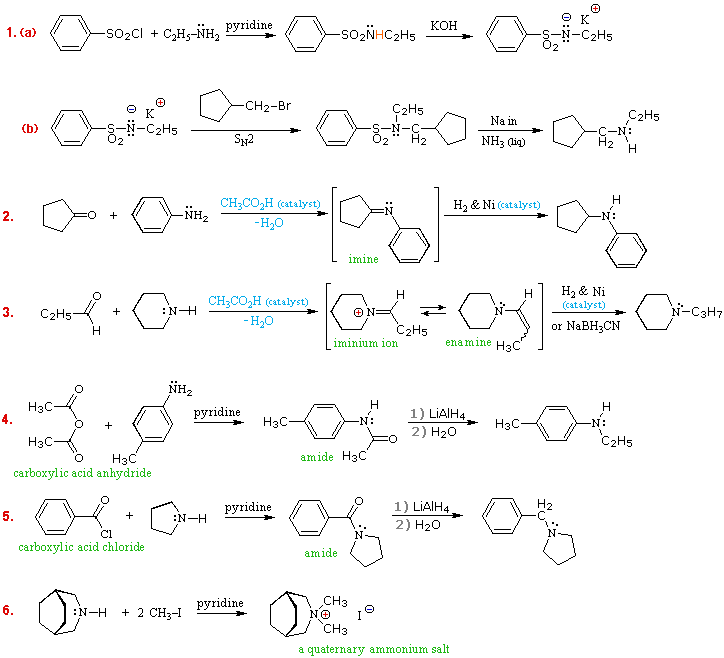
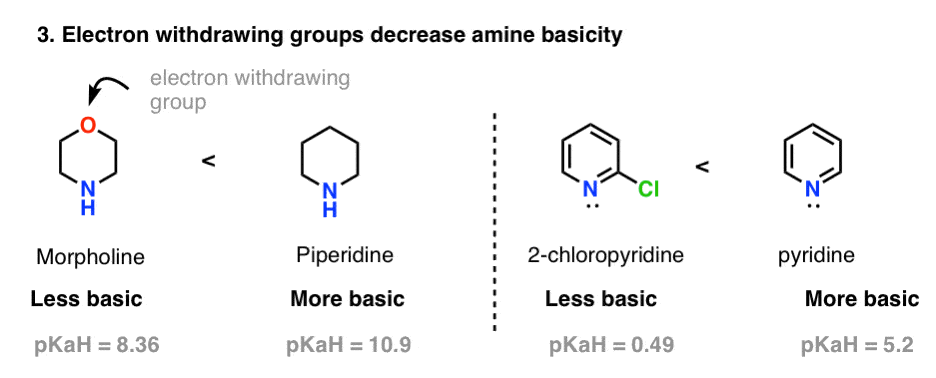
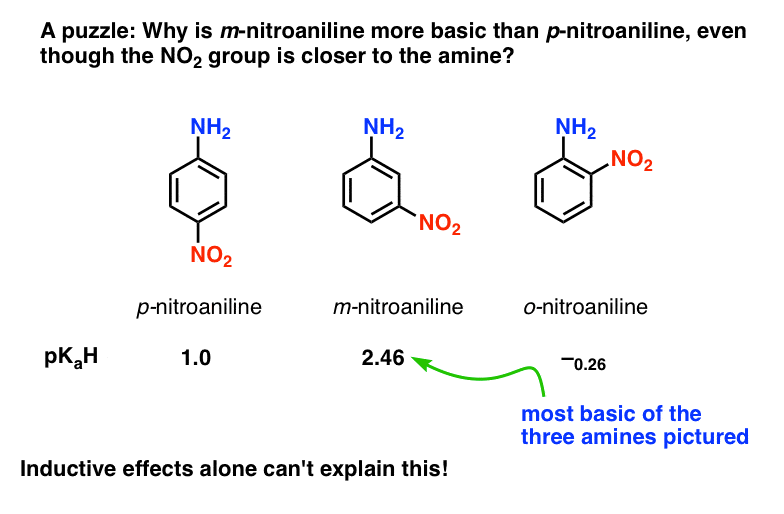
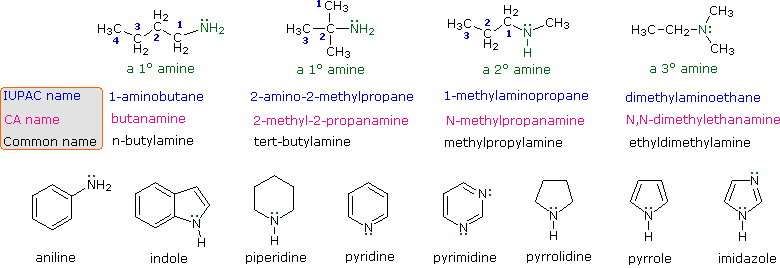
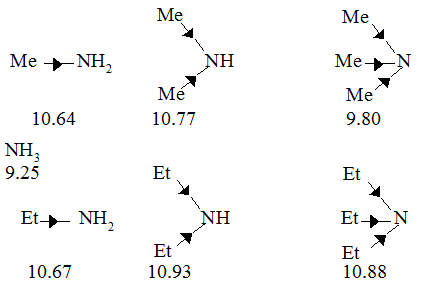
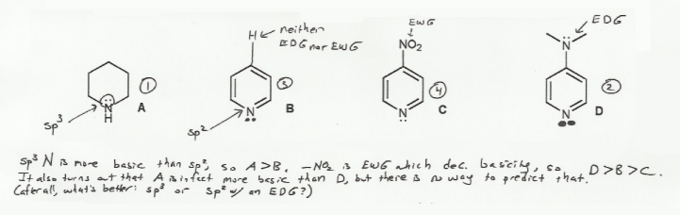
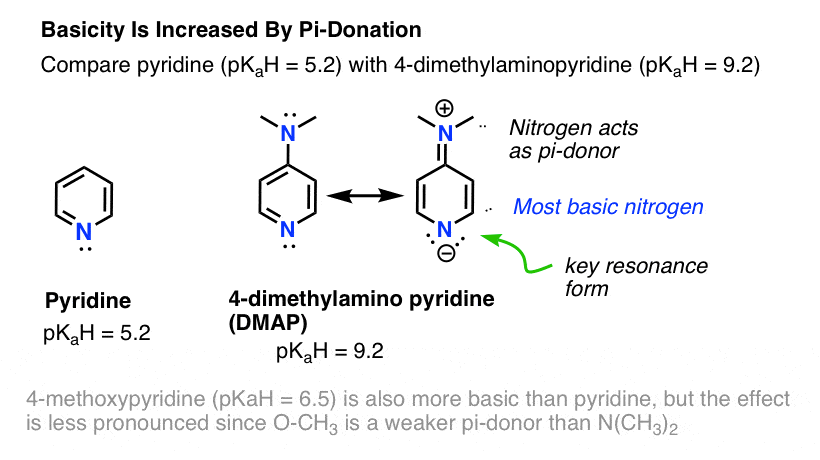
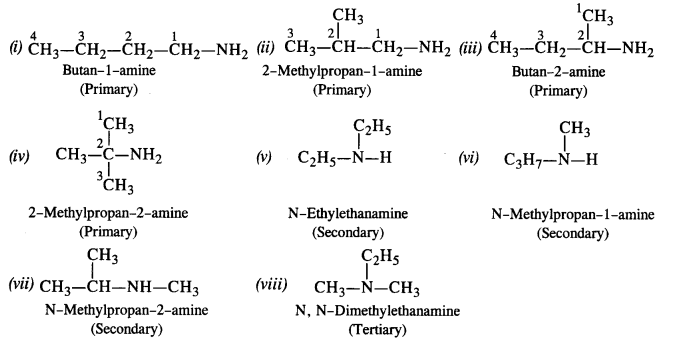
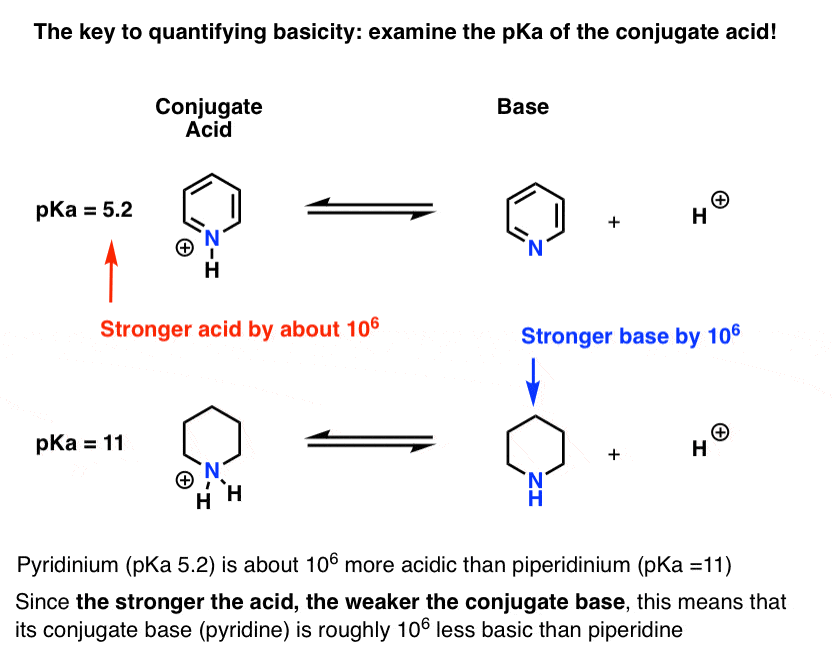
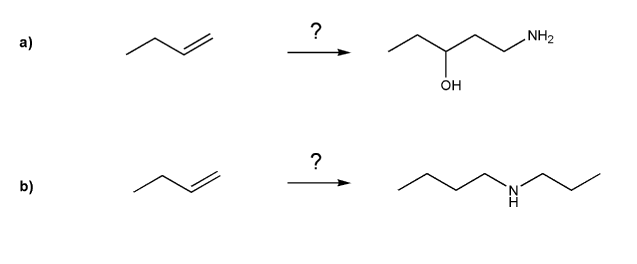
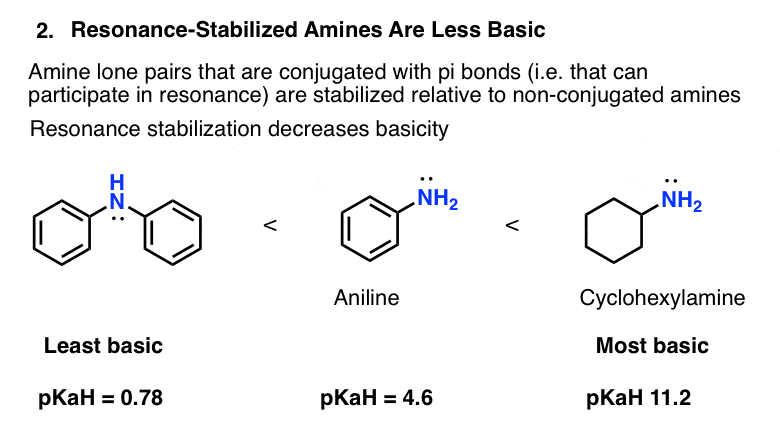basicity of amines practice problems
Why are amides weaker than amines?
In other words, the charge is delocalized on both nitrogen atoms of the ring. Amides are generally weaker bases than amines. The p K a of the conjugate acid of a typical amide is about zero and there are two factors contributing to this decreased basicity.

Amine Synthesis Reactions Organic Chemistry

22.2 Basicity of Amines Organic Chemistry

The Basicity of Amines (A2 Chemistry)
|
Organic Chemistry Jasperse Acid-Base Practice Problems
Acid-Base Practice Problems C. Rank the basicity of the following sets: Multiple Variable Problems. 12. CH3MgBr. CH3NHNa ... Amines/Ammoniums. |
|
Chapter 6 Amines and Amides
containing weakly basic organic compounds; thousands of these substances are known.) • Amines are organic derivatives of ammonia |
|
Reactions of Amines
That should help you solve relative basicity problems. If given ammoniums draw the related conjugate neutral amines |
|
Chapter 23 The Chemistry of Amines
The basicities of all these amines are reduced by resonance interaction of the nitrogen INSTRUCTOR SUPPLEMENTAL SOLUTIONS TO PROBLEMS • CHAPTER 23. |
|
Classification and Nomenclature of Amines
containing weakly basic organic compounds; thousands of these substances are known.) • Amines are organic derivatives of ammonia |
|
Practice-Sets-All-Organic-Chemistry-2.pdf
problem practice set which are also included in the class notes. C. Rank the basicity of the following sets: Multiple Variable Problems. 12. CH3MgBr. |
|
Objectives Organic Chemistry 360
alcohols aldehydes |
|
Medicinal Chemistry Self Assessment - Sample Chapter 3
Section 3 General Self Assessment Answers. 2.3 Solving pH/pK a. Problems pH = 1.8. Answer: Carboxylic acid. Acidic. Secondary amine. Basic. Aromatic. |
|
Test2 ch17a Acid-Base Practice Problems
Extra Practice Problems. General Types/Groups of problems: Conceptual Questions. Recognizing Strong versus Weak Acids; Recognizing Basic versus Nonbasic. |
|
1. For each of the species below identify the most acidic proton and
PRACTICE PROBLEMS FOR BRONSTED-LOWRY ACID-BASE CHEMISTRY Arrange the substances by order of acidity or basicity as indicated. You may use pKa. |
|
Reactions of Amines
Amines are completely converted to ammonium salts by acids • Ammonium Mechanism: Learned for last test (not tested this time) • Must have at Amines 12 Acidity/Basicity Table 19 2: With both Neutral and Cationic Acids and both o Example of resonance versus electronegativity: a C-H with carbonyl resonance |
|
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
Relative basicity of amines can be compared in terms of pK a values for Solubility of amines in dilute acid can be used as a chemical test • Amides are not |
|
Amines
*For each amine, pKa pKb 14 00 E X A M P L E 10 5 Predict the position of equilibrium for this acid–base reaction: CH3NH2 CH3COOH N CH3NH3 |
|
Amines
example of the electron-releasing nature of alkyl groups This increase in the basicity of an amine by alkylation is slightly enhanced by introduction of a second alkyl group Further A problem in synthesizing 1 o amines by the direct alkylation |
|
145 Chapter 24: Amines Amines: Nitrogen containing organic
make amines both basic and nucleophilic There are many The nomenclature for primary amines is similar to that of alcohols, practice problem 24 1 (p 908) |
|
Chapter 19 – Amines
Pyrrole is even less basic, with a pKb around 16, because its lone pair is involved in diethyl ether o Here's an example with draining in between o The problem with this reaction is that it's difficult to control how many alkyl groups add |
|
ORGANIC CHEMISTRY TOPICAL: Amines Test 1 - From the makers
you are using this test for the sole purpose of content reinforcement Which of the following amines is the most basic in as a product is stabilized, for example |
|
AMINES
Amines are organic derivatives of ammonia in which one or more of Aqueous solutions of amines are basic because For example, aniline is less basic than |
|
Chapter 17: Amines and Amides
drugs used for the treatment of mental illness, hay fever, hear problems, and other physical In the example above water is acts as an acid The resulting it is also common to discuss the basicity of amines by reference to the acid ionization |
|
Practice Problems - Amines
For each of the molecules below, circle the most basic nitrogen atom HN N H2N N N H NH NH H2N N N N N N H N NO2 N H O NH2 H2N NH2 O2N |