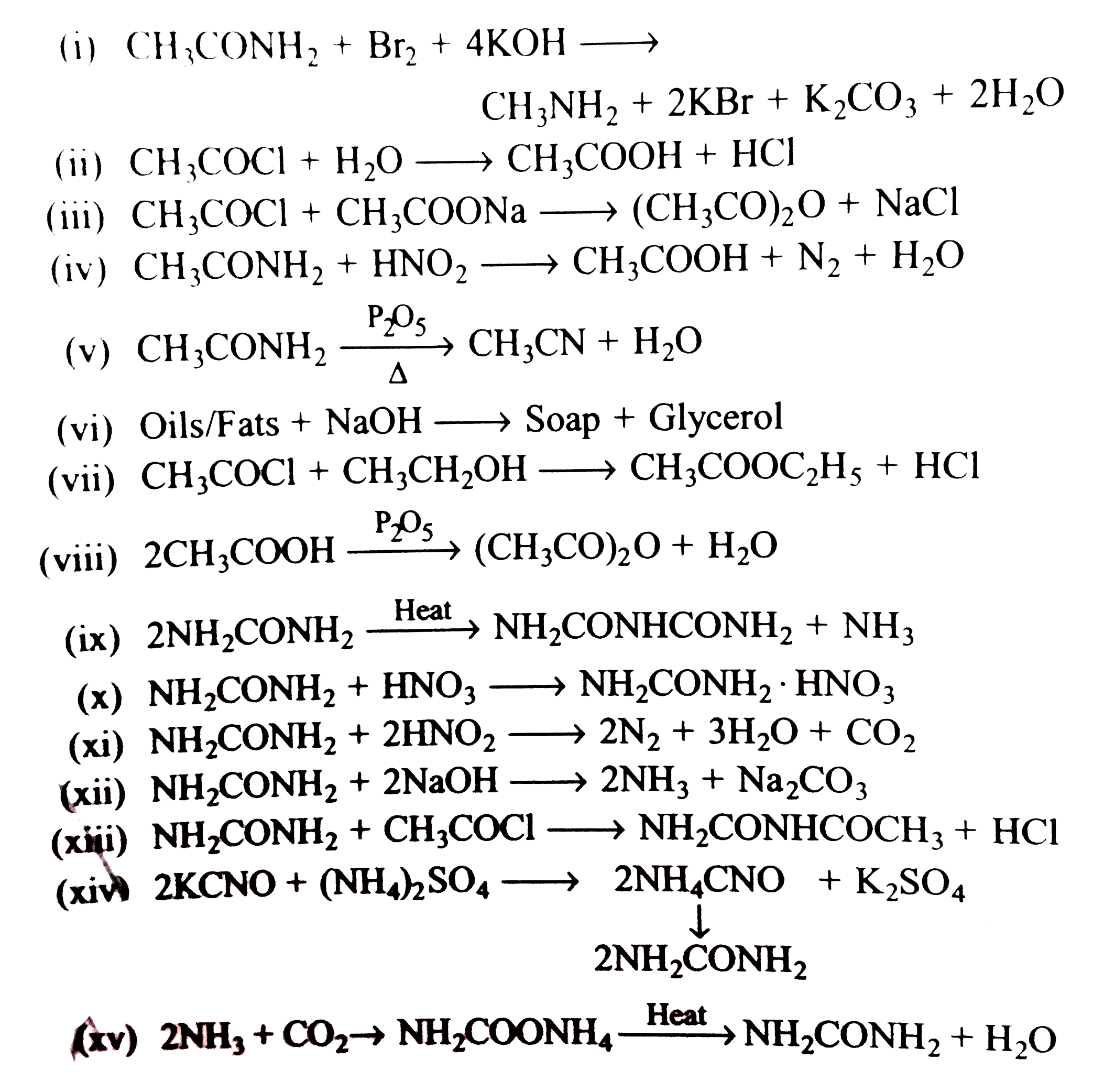acetamide reaction with naoh
What temperature is acetamide at?
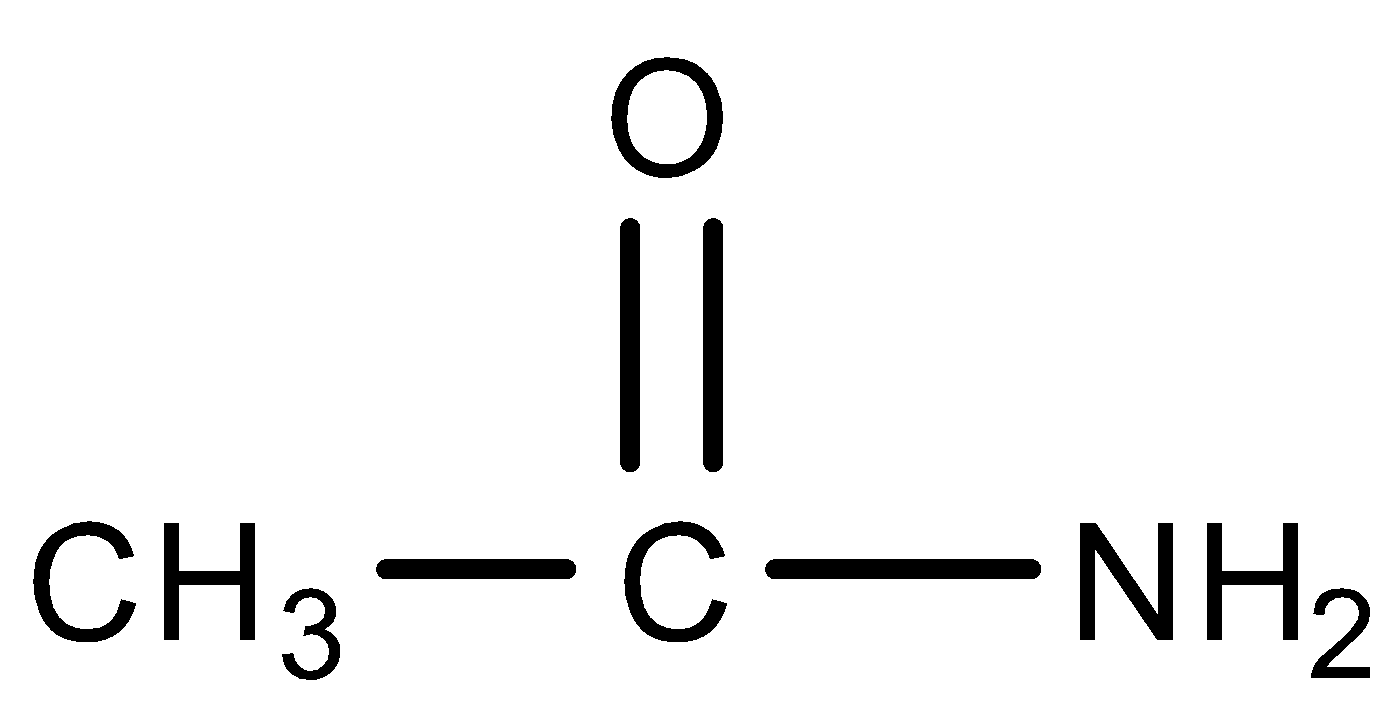
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). ?) Acetamide (systematic name: ethanamide) is an organic compound with the formula CH 3 CONH 2.
Is acetamide a compound?
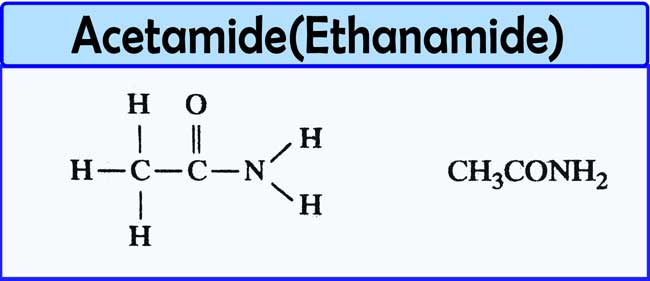
Acetamide (systematic name: ethanamide) is an organic compound with the formula CH 3 CONH 2. It is derived from acetic acid. It finds some use as a plasticizer and as an industrial solvent. The related compound N, N -dimethylacetamide (DMA) is more widely used, but it is not prepared from acetamide.
Is acetamide a hydrogen bonded dimer?
Acetamide can be considered an intermediate between acetone, which has two methyl (CH 3) groups either side of the carbonyl (CO), and urea which has two amide (NH 2) groups in those locations. Acetamide is also a naturally occurring mineral with the IMA symbol: Ace. Structure of acetamide hydrogen-bonded dimer from X-ray crystallography.
Can acetamide be heated with hydrochloric acid?
Taking acetamide (ethanamide) as a typical amide. If acetamide is heated with a dilute acid (such as dilute hydrochloric acid), acetic acid is formed together with ammonium ions. So, if you were using hydrochloric acid, the final solution would contain ammonium chloride and acetic acid.

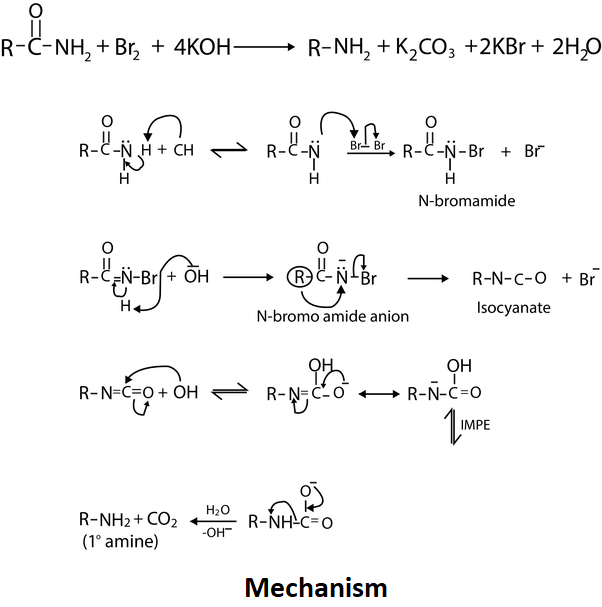
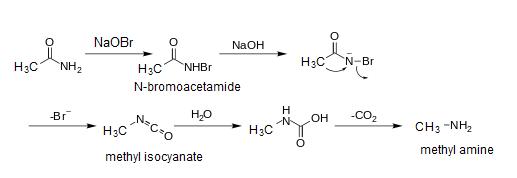
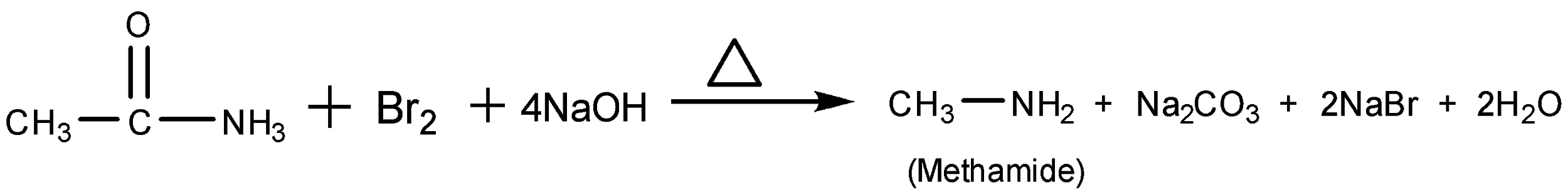
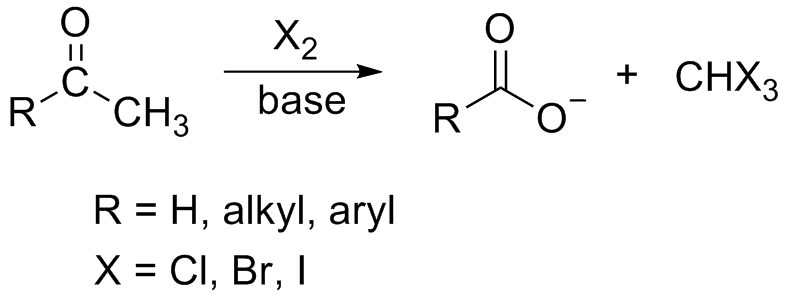
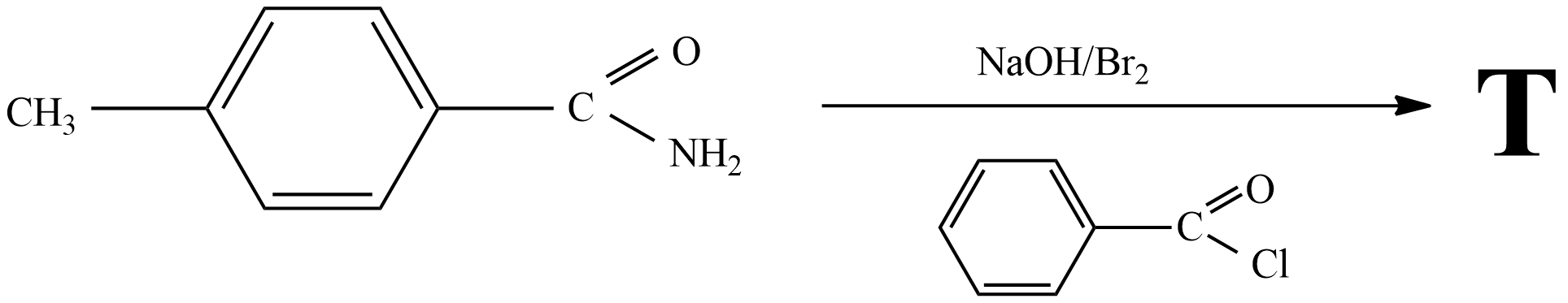
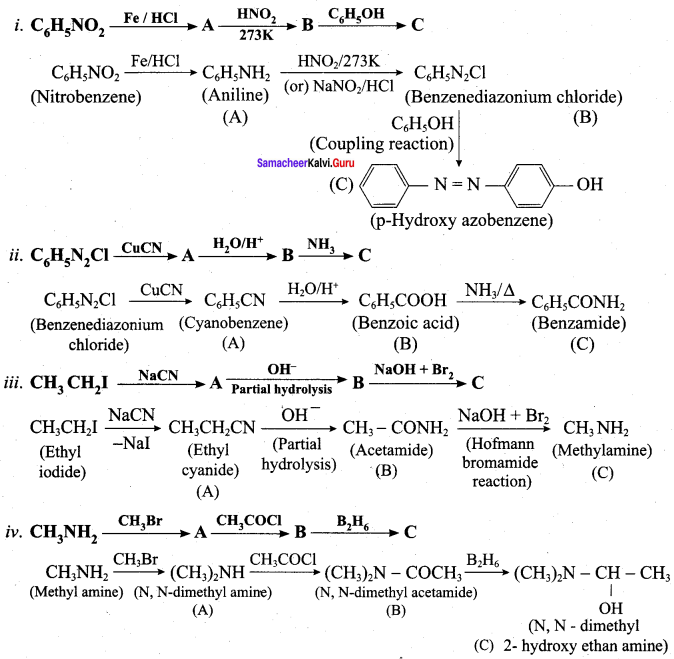
Assertion : When acetamide reacts with `NaOH and Br_(2)` methyl amine

Type of Reaction for CH3COOH + NaOH = NaCH3COO + H2O

Equation for NaOH + H2O (Sodium hydroxide + Water)
|
A Study of the Mechanism and Kinetics of the Thioacetamide
Thioacetic acid was detected as an intermediate in both reactions. The relative importance of thioacetic acid and acetamide as intermediates is not known. |
|
RÉACTIFS
22 oct. 2020 N[3-[(Diéthyl-amino)méthyl]-4-hydroxyphényl]acétamide. ... La solution donne la réaction de l'aluminium. (2.3.1). |
|
Page 1 of 4 Enzymatic Assay of AMIDASE (EC 3.5.1.4) PRINCIPLE
12 juil. 1994 Acetamide + Hydroxylamine Amidase > Acethydroxamate + NH3 ... Adjust to pH 7.2 at 37°C with 1 M NaOH.) ... Reaction Cocktail. 1.90 1.90. |
|
PRESSURE EFFECT AND MECHANISM IN ACID CATALYSIS: VIII
The hydrolysis of acetamide was followed by ailalyziilg for the acetic acid produced. About 5 in1 of reaction solutioil (initially either 0.2992 M in |
|
Determination of a Reactions Activated Energy Using Naoh as a
21 juin 2018 Chemical kinetics includes investigations into how different experimental conditions can influence the speed of a chemical reaction and yield ... |
|
Chapter 6 Amines and Amides
Learn the major chemical reactions of amines and amides and learn how to predict the Acetic acid. 60.0 g/mol 118°C ... + NaOH ---? R—NH2 + H2O + NaCl. |
|
Nitrosamines EMEA-H-A5(3)-1490 - Assessment Report
25 juin 2020 chloramination (by the reaction of monochloramine with ... acetic acid acetic anhydride |
|
Chpt 23Final7e
Step (1) Reaction with NaOH to produce the phenoxide followed by treatment with CH3 I. Step (2) Aromatic nitration reaction using HNO3 and H2 SO4 . |
|
Multiple Choice Questions Unit (9) Chem-100
When ______ reacts with NaOH the product is sodium benzoate. A. benzoic acid The missing chemical in the reaction (acetamide + sodium hydroxide ? |
|
Reactions of Amines
NaOH amine base ammonium salt (acidic) • Mechanism: Required Substitution for Aromatic Amines via the Diazonium Salts (“The Sandmeyer Reaction”) Of the three neutrals, acetic acid is most due to resonance, ammonia least due to |
|
THE HYDROLYSIS OF AMIDES IN THE ANIMAL BODY
Sencki,* many years earlier, concluded that acetamide is not hydrolyzed at all by dogs, since it did not evident that amide hydrolysis, being a relatively simple reaction, offered an the amide and HCI (or NaOH) were present in equivalent |
|
Determination of a Reactions Activated Energy Using Naoh as a
21 jui 2018 · titrated to determine the concentration of acetic acid In another study, Shatyski and Hanesian [2] determined the kinetics of the above reaction |