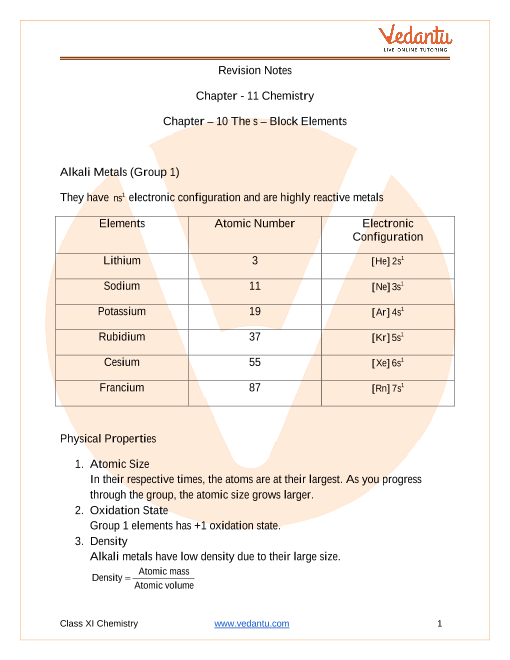
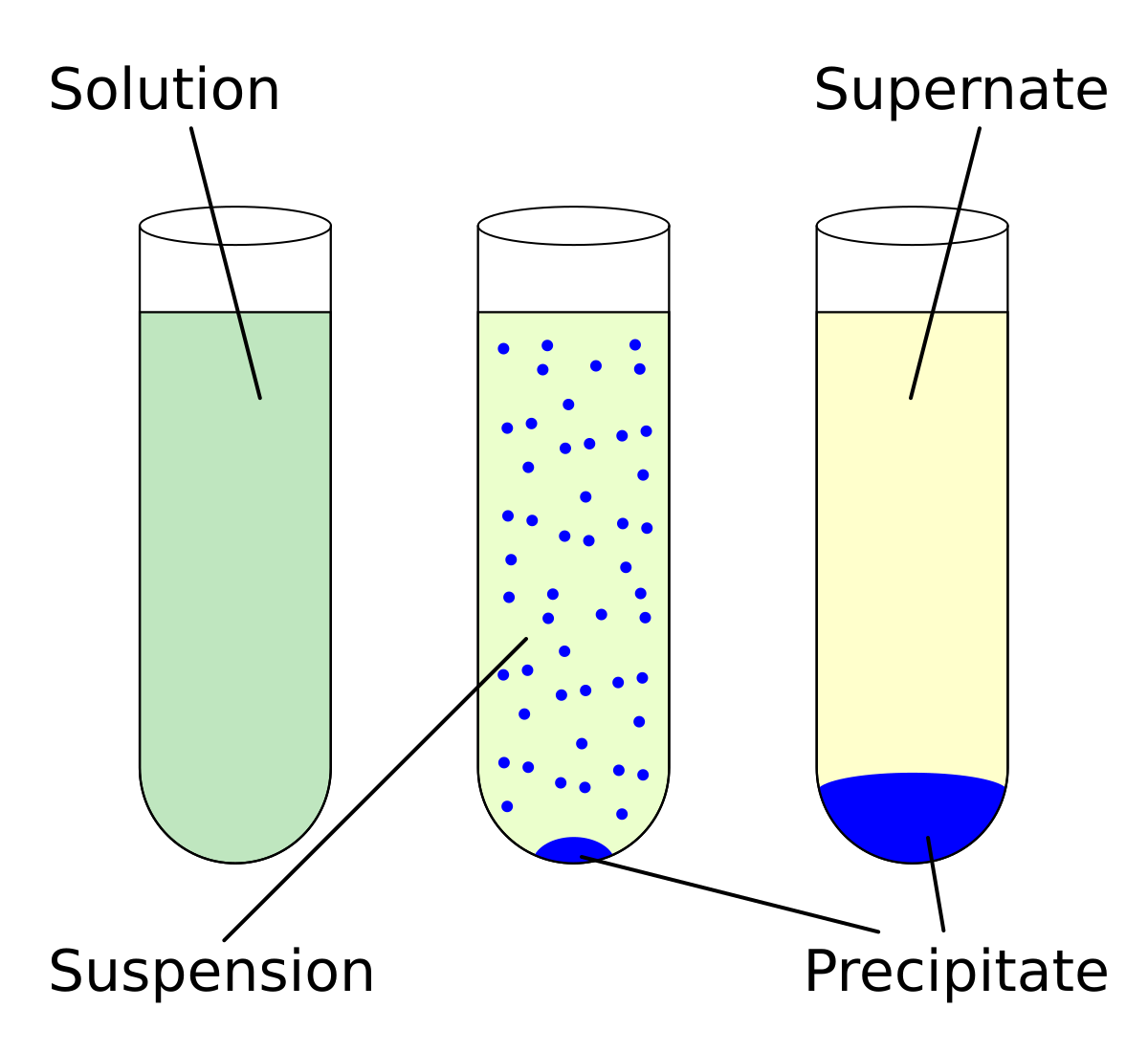
ap chemistry properties of solutions notes
|
AP* Chemistry PROPERTIES OF SOLUTIONS
➢ molar solubility—the number of moles of solute that dissolves in exactly 1 0 L of solvent, expressed in units of molarity, M or the use of square brackets ➢ |
|
Chapter 11 – Properties of Solutions
componentA of fraction Mole + = = Chapter 11 – Properties of Solutions 11 1 Solution Composition A Molarity 1 solution of liters solute moles M Molarity = |
|
AP Chemistry: Properties of Solutions Lecture Outline 131 The
13 1 The Solution Process A solution is a homogeneous mixture of solute and solvent Solutions may be gases, liquids, or solids Each substance present is a component of the solution The solvent is the component present in the largest amount |
|
Chapter 13 Properties of Solutions
There will be no further increase in the amount of dissolved solute 11 “The Use of Dots in Chemical Formulas” from Further Readings 12 “Hydrated Magnesium |
|
Chapter 13 Properties of Solutions
Solutions Chapter 13 Properties of Solutions Chemistry, The Central Science, 10th Solutions The intermolecular forces between solute and solvent particles |
|
AP Chemistry Unit 3 Solutions and Mixtures Notes • A - Effingham
: water is the dissolving medium, or ______ • One of most important properties of water is its ______ to many different substances • Water is Polar • |
|
Chapter 12 Physical Properties of Solutions
SUMMARY The Solution Process (Sections 12 1 - 12 2) Some Definitions The main subject of this c ~ter concerns the formati n ag~ a_&~ properties of liquid |
|
Chapter 7 lecture notes: Solutions
by ______ methods • The properties of mixtures depend on the Chemistry 108 Lecture Notes Chapter 7: Solutions 3 Solutions • The primary ingredient in |
|
UNIT 4- BONDING (Ch - Denton ISD
STUDENT NOTES Pre-AP Chemistry Evaluate colligative properties - boiling point elevation and freezing point Leggett PreAP Chem Solutions 1 (7:56) |
|
Chapter 4 Aqueous Reactions and Solution Stoichiometry
AP Chemistry Chapter 4 Molarity is moles of solute per liter of solution not per liter of solvent Lecture Outline 4 1 General Properties of Aqueous Solutions |