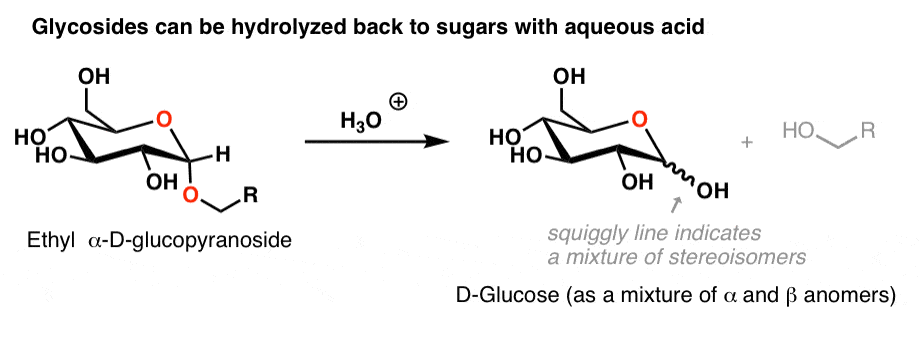acid catalyzed hydrolysis of sucrose
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
experiment stops short of this goal It is often given or a bimolecular process where a molecule of water is as an example of a pseudo-first order reaction |
What is the catalyst for the hydrolysis of sucrose?
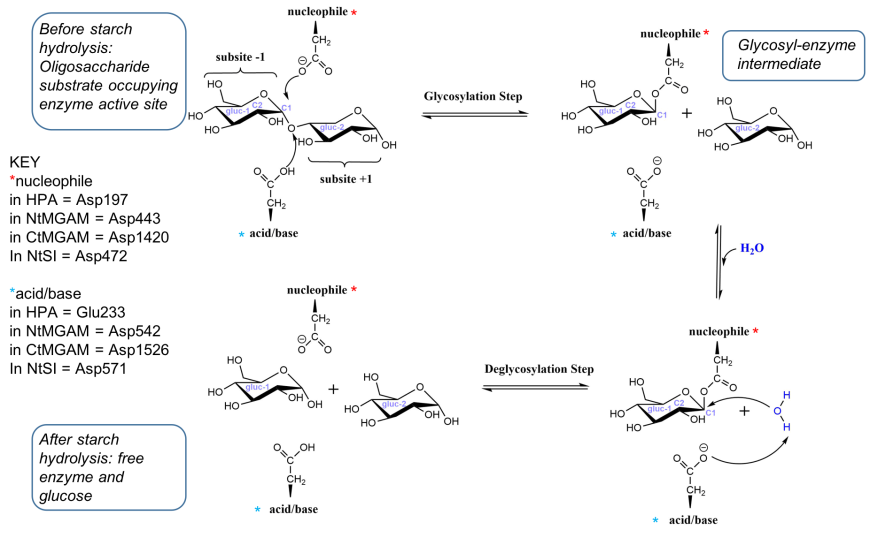
The hydrolysis of sucrose to glucose and fructose is catalyzed by invertase.
Sugarcane is sucrose (C12H2O11), which is dextrorotatory with a specified rotation angle of +62.5 degrees.
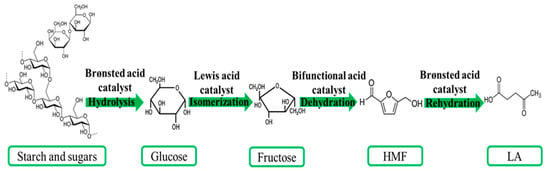
Sucrose is hydrolyzed by the enzyme invertase, yielding – D(+) glucose and – D(-) fructose as products.Acid-catalyzed hydrothermal hydrolysis was performed using 0.5–2.
0) N of HCl, H3PO4, HNO3, and H2SO4 at reaction temperatures of 170–210 °C and a reaction time of 3 min, which was optimized for glucose production (data are not shown), and the concentrations of glucose and decomposed products were compared between the
What is the mechanism of hydrolysis of sucrose?
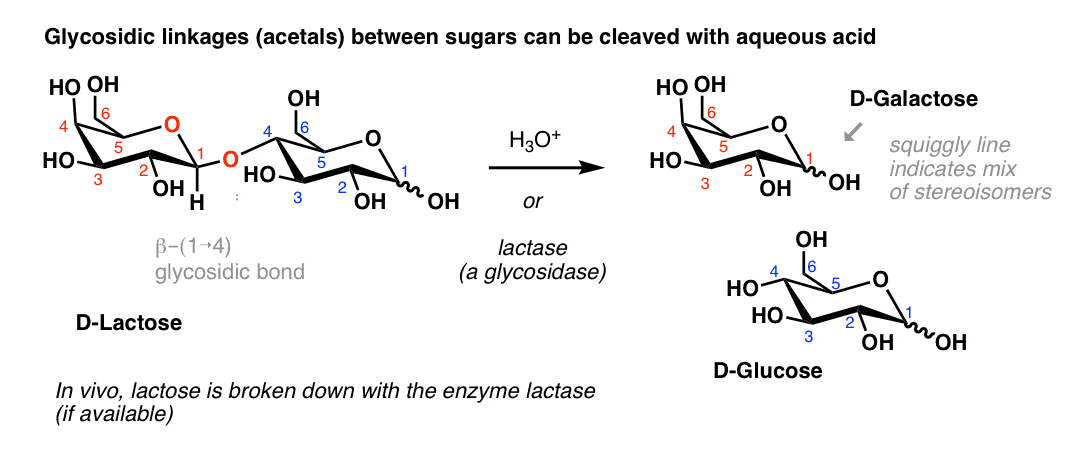
Hydrolysis of sucrose to give glucose and fructose is called inversion. hydrolysis of sucrose brings about a change in the sign of rotation, from dextro (+) to laevo (–) and the product is named as invert sugar.
What is acid hydrolysis of sucrose?
Therefore, sucrose upon acidic hydrolysis i.e. reacting with water in presence of hydrochloric acid forms glucose and fructose.
The sign of total optical rotation of products changes which means there is inversion of sign.
The optical orientation changes from dextrorotatory to laevorotatory.
|
Kinetic solvent effects on acid-catalyzed hydrolysis of sucrose in
Rate constants of acid-catalyzed hydrolysis of sucrose (S) to D-glucose and L-fructose have been determined at 25OC by optical rotation measurements in |
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
power of acid solutions as measured by the Hammett acidity function (1-5). Acid-Catalyzed Hydrolysis of Sucrose. A student study of a reaction mechanism. H++~ |
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
example of a pseudo-first order reaction and the student merely determines a single valueof the rate constant at a fixed concentration of catalyzing acid. |
|
Chem 11 Lab Manual
Hydrolysis of Sucrose (Acid versus Base Catalysis). Sample. Condition of Acid-Catalyzed Hydrolysis of Starch. Sample. Heating Time. (min). Color Observation. |
|
Base-catalyzed sucrose degradation studies
In fruit juices sucrose contributes to nonenzymic browning only after acid-catalyzed hydrolysis to the more reac- tive reducing sugars d-fructose and |
|
The oxygen-18 isotope shift in carbon-13 nuclear magnetic
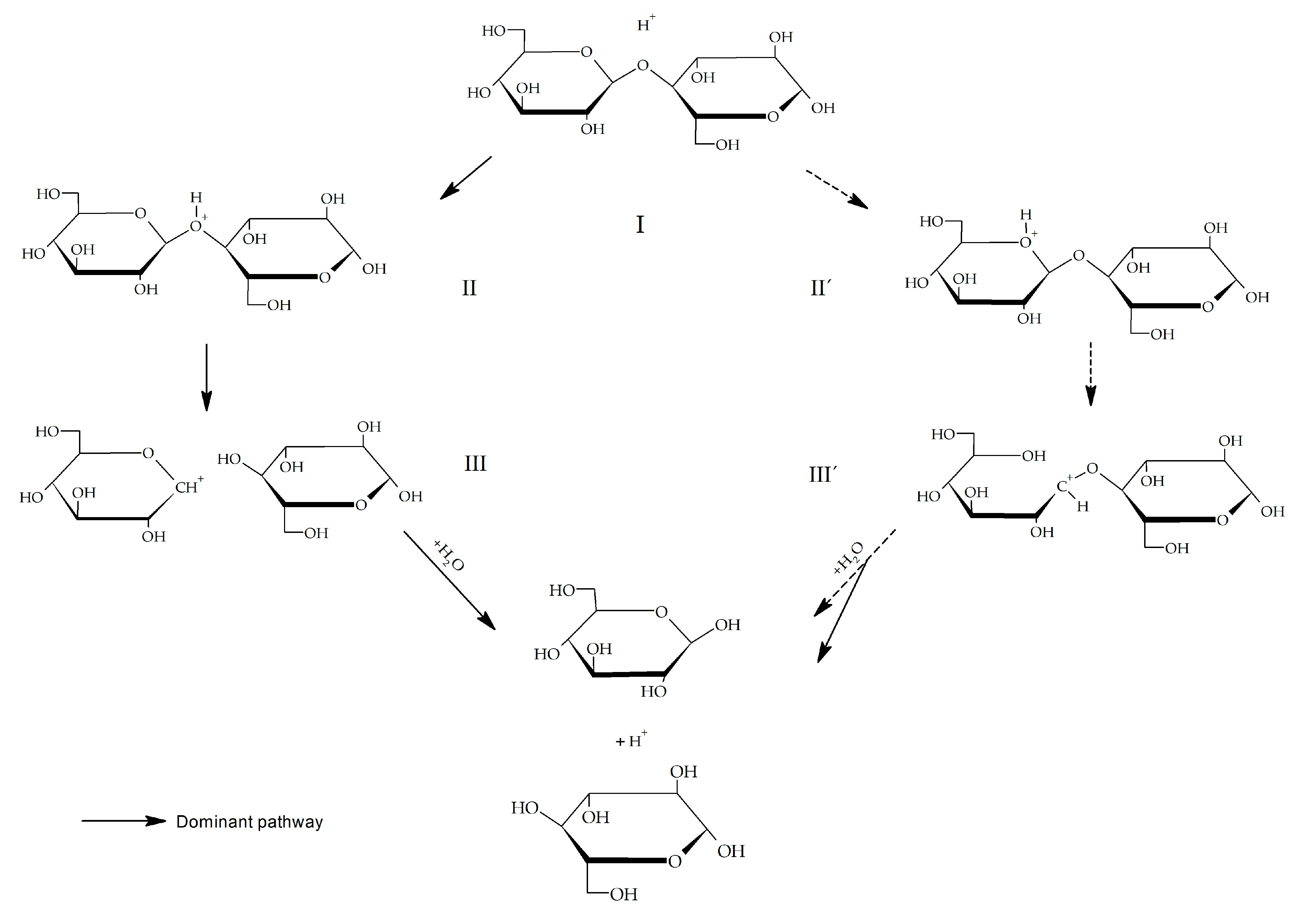
Abstract: The 180 isotope-induced shift in 13C NMR spectroscopy was used to elucidate the point of bond cleavage in the acid-catalyzed hydrolysis of sucrose |
|
Salt effects on the hydrolysis of sucrose
SO. 1.0. ''sail. 2 0. 3 0. The acid-catalyzed hydrolysis of sucrose has long been regarded as a third-order reaction. XN°3 involving sucrose water |
|
THE HYDROLYSIS OF SUCROSE BY HYDROCHLORIC ACID IN
C. [Contribution from the Department of Chemistry of the University of. Pittsburgh]. THE HYDROLYSIS OF SUCROSE BYHYDROCHLORIC ACID IN on the catalysis by ... |
|
The Relation between the Rates of Some Acid Catalyzed Reactions
and the trichloroacetic acid hydrolysis of sucrose can indeed be properly classified with them. Summary. The rates of the acid catalyzed hydrolysis of. |
|
Hydrolysis of Sucrose over Sulfonic Acid Resins
09-Jul-2021 catalysis which is 107 kJmolÀ 1 KÀ 1.[1213 |
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
Acid-Catalyzed Hydrolysis of Sucrose. A student study of a reaction mechanism. H++~GLUCOSE. = a GLUCOSE. Figure 1. For dilute solutions of acids the most |
|
Kinetic solvent effects on acid-catalyzed hydrolysis of sucrose in
Rate constants of acid-catalyzed hydrolysis of sucrose (S) to D-glucose and L-fructose have been determined at 25OC by optical. |
|
Enzymic and Acid Hydrolysis of Sucrose as Influenced by Freezing
Sucrose hydrolysis catalyzed by acid (HCI) was studied over the temperature range 12 to -16.5”C and an Arrhenius plot of the data was linear between 12 and. |
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
the hydrolysis of sucrose the beginning student in chemistry can gather some constant at a fixed concentration of catalyzing acid. If. |
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
the hydrolysis of sucrose the beginning student in chemistry can gather some constant at a fixed concentration of catalyzing acid. If. |
|
Salt effects on the hydrolysis of sucrose
The acid-catalyzed hydrolysis of sucrose has long been regarded as a third-order reaction. XN°3 involving sucrose water |
|
The oxygen-18 isotope shift in carbon-13 nuclear magnetic
acid-catalyzed hydrolysis of sucrose. Sucrose was hydrolyzed in the presence of H2180 and the incorporation of 180 into the. |
|
Sucrose hydrolysis catalyzed by auto-immobilized invertase into
Acid and enzymatic hydrolysis have been identified as chemical and biochemical ways to sucrose inversion. (disaccharide) into glucose and fructose (soluble. |
|
Chem 11 Lab Manual
Hydrolyzed sucrose (a mixture of D–glucose and D–fructose) will give a positive test Hydrolysis of Sucrose (Acid versus Base Catalysis). |
|
Acid-catalyzed hydrolysis of sucrose: A student study of a reaction
Acid-Catalyzed Hydrolysis of Sucrose A student study of a reaction mechanism H++~GLUCOSE = a GLUCOSE Figure 1 For dilute solutions of acids the most |
|
Application of the acid hydrolysis of sucrose as a temperature
The hydrolysis of sucrose in an acid medium was used as a temperature indicator to measure holding temperatures in a con- tinuous thermal-processing unit |
|
Acid-Catalyzed Inversion of Sucrose in the Amorphous State at Very
Reaction at 0:1 5 87 0 0 limited water concentration 1 Sucrose hydrolysis J Food Sci 1:10 2 43 0 76 0 09 0 176 0 014 |
|
Sucrose Inversion: An Experiment on Heterogeneous Catalysis* - IJEE
The experiment deals with the acid-catalyzed sucrose inversion, sucrose concentration, within the catalyst goes acid hydrolysis into glucose and fructose |
|
Inversion of Sugar : The hydrolysis of sucrose by boiling with a
The hydrolysis of sucrose by boiling with a mineral acid, or by the enzyme invertase, produce a mixture of equal molecules of D-glucose and D-fructose H+ |



![PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a](https://imgv2-1-f.scribdassets.com/img/document/345162169/298x396/473380c12c/1605197801?v\u003d1)

![PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a](https://www.frontiersin.org/files/Articles/357117/fchem-06-00146-HTML-r1/image_m/fchem-06-00146-g001.jpg)
![PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a](https://ars.els-cdn.com/content/image/1-s2.0-S0920586118301068-fx1.jpg)




![PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a PDF] Acid-Catalyzed Hydrolysis of Sucrose A student study o f a](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs00706-020-02636-z/MediaObjects/706_2020_2636_Figa_HTML.png)