ethyl methanoate with conc h2so4
How does ethanoic acid react with sulphuric acid?
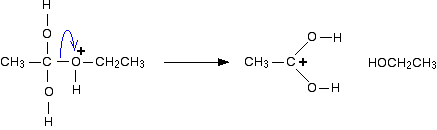
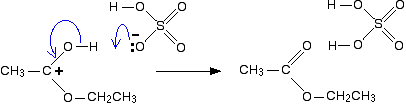
In the first step, the ethanoic acid takes a proton (a hydrogen ion) from the concentrated sulphuric acid. The proton becomes attached to one of the lone pairs on the oxygen which is double-bonded to the carbon.
How does ethyl ethanoate react with hydrochloric acid?
Ethyl ethanoate is heated under reflux with a dilute acid such as dilute hydrochloric acid or dilute sulphuric acid. The ester reacts with the water present to produce ethanoic acid and ethanol. Because the reaction is reversible, an equilibrium mixture is produced containing all four of the substances in the equation.
What happens when ethyl ethanoate is heated under reflux?
When ethyl ethanoate is heated under reflux with a dilute acid such as dilute hydrochloric acid or dilute sulfuric acid, the ester reacts with the water present to produce ethanoic acid and ethanol. Because the reaction is reversible, an equilibrium mixture is produced containing all four of the substances in the equation.
|
Topic 4.5 COMPOUNDS CONTAINING THE CARBONYL GROUP
reagents: any alcohol concentrated sulphuric acid catalyst this reason (eg ethyl methanoate gives raspberry flavour and ethyl butanoate gives pear. |
|
Untitled
ethyl methanoate: CH3CH2CHO heat with ethanol in conc. sulfuric acid/esterification methanoic acid: HCOOH. ? Figure 3 A reaction pathway showing how ethyl |
|
Chapter 5 Carboxylic Acids and Esters
Diethyl ether. 74 g/mol 35°C. Insoluble. Butanal sodium ethanoate. (sodium acetate) ... Benzyl diethyl ((26-xylylcarbamoyl)methyl) ammonium benzoate. |
|
Ester Synthesis Lab (Student Handout)
reacting ethanol with acetic acid to give ethyl acetate is shown below. Then add 2 drops of concentrated sulfuric acid to the tube. |
|
Chemistry 30 Assessment Exemplars 2020-2021
the leaching process concentrated aqueous sulfuric acid |
|
Chemistry
2018?1?11? 11 When concentrated sulfuric acid is added to ethanoic acid the reaction is ... C forms when butyl methanoate reacts with sulfuric acid. |
|
Alcohols Phenols and Ethers
For example C2H5OC2H5 is diethyl ether. with concentrated sulphuric acid which converts it to ... (c) Mononitration of phenyl methanoate. |
|
June 2010 (v1) QP - Paper 4 CIE Chemistry A-level.pdf
H2SO4. 55?C. KMnO4. + OH–. + heat then H+. C10H10O4. CH3OH + conc. H2SO4 The compound could be propanoic acid methyl ethanoate or ethyl methanoate. |
|
Subject: Chemistry Structure and Properties Reactions – as typical
HCOO- is called methanoate ion & concentrated sulfuric acid (reflux) to form esters. ... R1COOR2 for example HCOOCH2CH3 is called ethyl methanoate. |
|
CCEA GCE Specimen Assessment Material for - Chemistry
2021?11?16? of concentrated sulfuric acid and transfer the mixture to a flask ... (ii) Write the equation for the formation of ethyl methanoate by an. |
|
Worksheet 1
Next add 0 1 g or 60 drops of the carboxylic acid (as identified in your table) Then add 2 drops of concentrated sulfuric acid to the tube a Caution: Concentrated |
|
405 Compounds Containing the Carbonyl Group - Topic 13
this reason (eg ethyl methanoate gives raspberry flavour and ethyl butanoate gives reflux with concentrated H2SO4, a carboxylic acid and an alcohol will be |
|
As typical acids Esterification equation examples
HCOO- is called methanoate ion concentrated sulfuric acid (reflux) to form esters R1COOR2 for example HCOOCH2CH3 is called ethyl methanoate 2 |
|
Making esters from alcohols and carboxylic acids - Pearson Schools
Butan-1-ol is harmful and irritant • Conc sulfuric acid is corrosive Ethyl ethanoate is a solvent used in modelling paints and glue Procedure 1 Place 1 cm3 of |
|
UNIT 11 - A-Level Chemistry
the alcohol should be heated at 180oC with concentrated H2SO4 • The H which is lost (i) Ethyl ethanoate with water and concentrated H2SO4 (ii) Methyl |
|
Synthesis, Isolation, and Purification of an Ester
In this experiment the ester ethyl acetate (ethyl ethanoate) is prepared the addition of concentrated sulfuric acid, H2S04, and the reaction is reversible Adding |
|
Download File - Do Chemistry
Design a synthetic route to produce ethyl / of concentrated sulfuric acid, creating the sweet- chloromethane and the ester product, ethyl methanoate |
|
Esterification and Odors of Esters
pleasant odor Materials: Conc H2SO4 Plastic pipets Glacial acetic acid Thin- stemmed pipet (banana), ethyl acetate (fruity), methyl salicylate (wintergreen) |









![ESTER -Strudural formela a] Methyl methanoate (methyl formate] b ESTER -Strudural formela a] Methyl methanoate (methyl formate] b](https://www.coursehero.com/thumb/24/1b/241bac55290582e94ebbff3d083f5562d4f96cd4_180.jpg)




![ESTER -Strudural formela a] Methyl methanoate (methyl formate] b ESTER -Strudural formela a] Methyl methanoate (methyl formate] b](https://0.academia-photos.com/attachment_thumbnails/37241984/mini_magick20180815-15649-1ep8o36.png?1534397861)







![Text Book Answers - [PDF Document] Text Book Answers - [PDF Document]](https://d10lpgp6xz60nq.cloudfront.net/q-thumbnail/34510389.png)