pka benzene ring
|
Approximate pKa chart of the functional groups: values to know
Carboxylic acid pKa = 4-5 4 Ammonium ion pKa = 9-10 5 Phenol pKa = 10 6 Thiol pKa = 10 7 Alcohol pKa = 16-18 8 Water pKa = 15 7 9 Amide pKa = 18 10 |
|
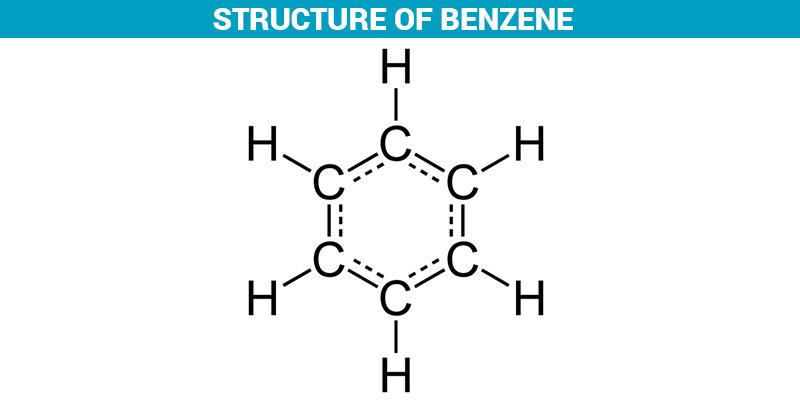
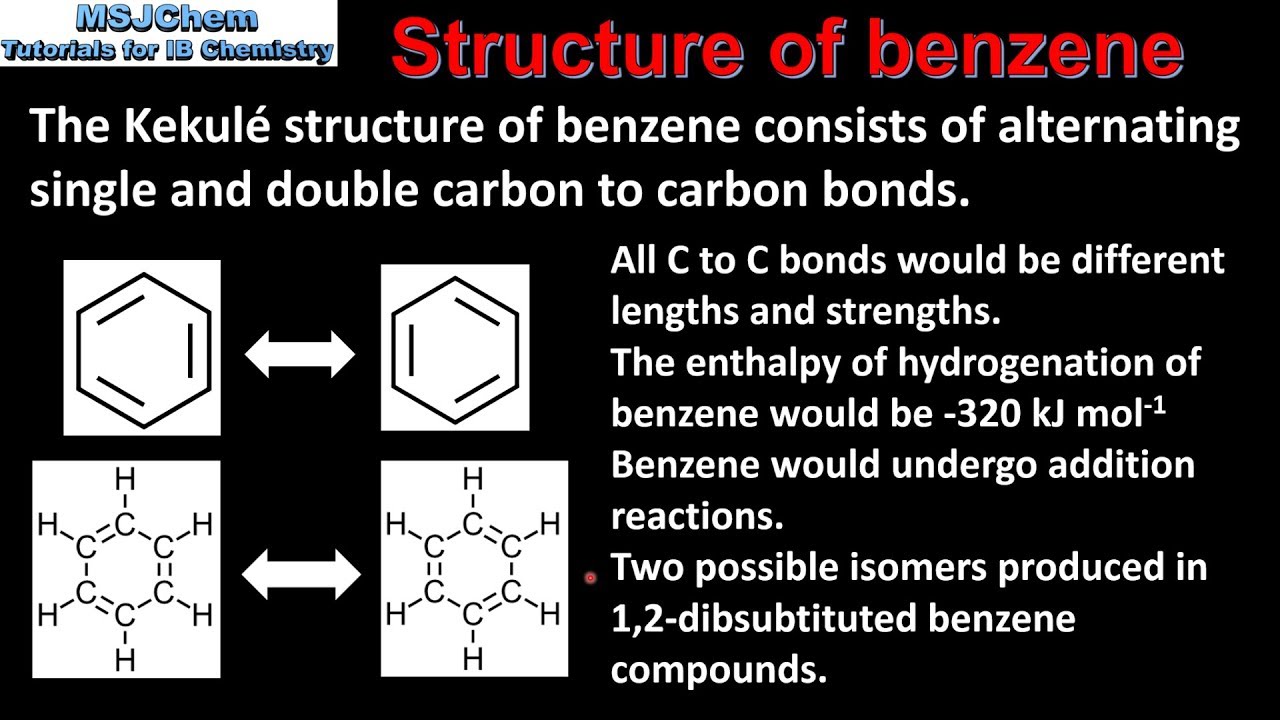
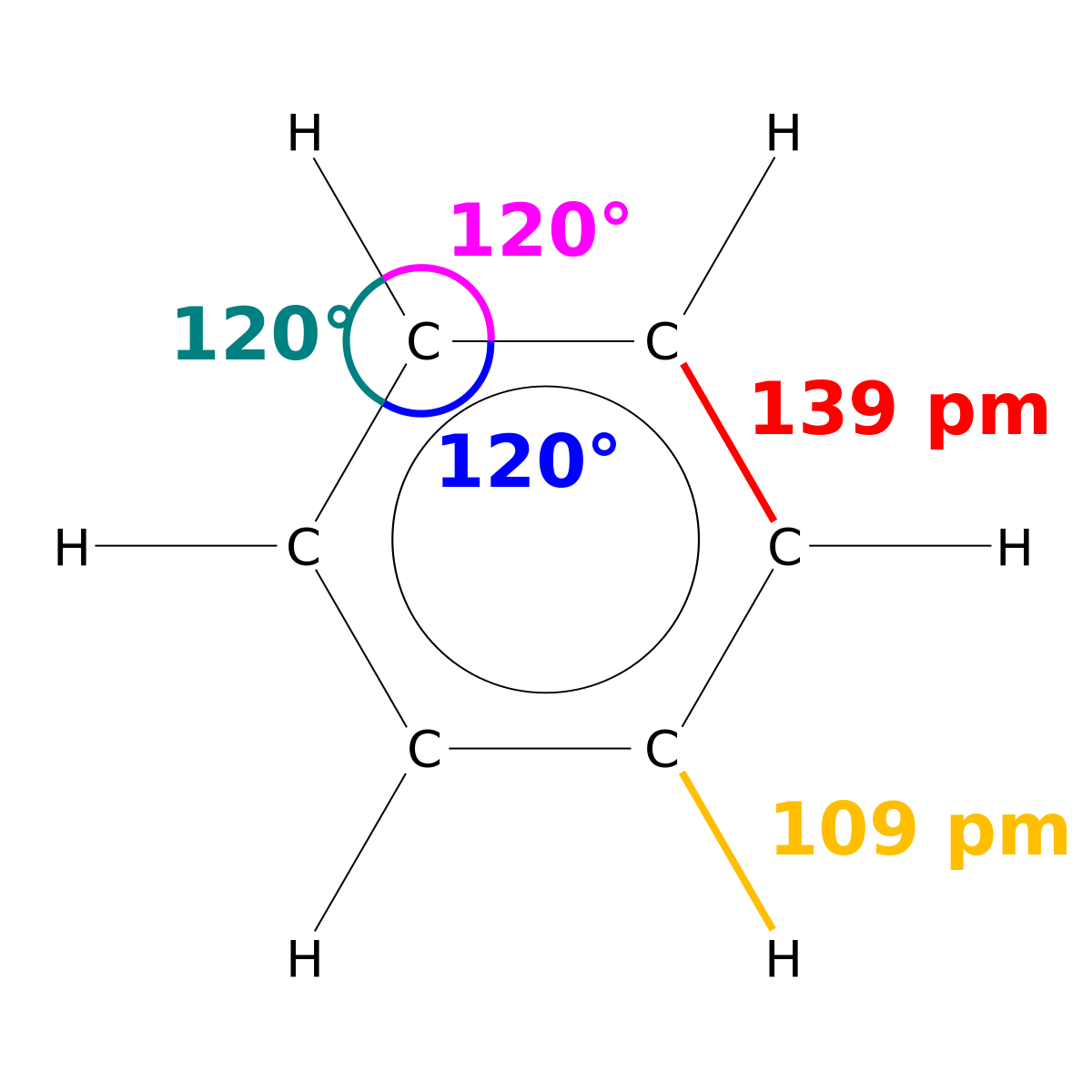
Benzene & Aromaticity
group bonded to a benzene ring 12-Benzenediol (Catechol) 14-Benzenediol pKa 10 01 p-Cresol pKa 10 17 OH OH OH OH OH CH3 CH3 Cl Cl Acidity of |
|
PKa values
The pKa value is typically between -6 and 0 O OH R 5 carboxylic acid (OH attached to carbonyl) |
|
Chapter 17: Alcohols and Phenols
Chapter 17: Alcohols and Phenols phenol. (aromatic alcohol). pKa~ 10 alcohol Phenols contain an OH group connected to a carbon of a benzene ring. |
|
Approximate pKa chart of the functional groups: values to know 1
Protonated carbonyl pKa = -7 Other important pKa's. 2. Protonated alcohol or ether pKa = -2 to -3. H2 = 35. 3. Carboxylic acid pKa = 4-5. |
|
120 Chapter 24: Phenols. Alcohols contain an OH group bonded to
bonded to an sp2-hybridized carbon of a benzene ring. 24.1: Nomenclature (please read) pKa ~ 10. 235. Factors that influence acidity: Inductive effect:. |
|
Structural basis for the acidity of sulfonamides. Crystal structures of
pronounced acidity of dibenzenesulfonamide (pKa = the benzene ring causes the. pKa ... atom” role in dominating the largest ?'s. The benzene rings were. |
|
The Effects of Chemical Substitution and Polymerization on the pKa
25 sept. 2009 with -NO2 groups which form an extended conjugated ?-system with the benzene ring exhibits the strongest enhancement of the acidity. |
|
On the basicity of conjugated nitrogen heterocycles in different media
additional benzene ring does not always lead to basicity increase in solution: aqueous pKa values of many different types of heterocycles are available. |
|
Benzene & Aromaticity
ring. 3. be planar or nearly planar so that there is group bonded to a benzene ring ... pKa. 7.15 m-Nitrophenol. pKa. 8.28. Acidity of Phenols ... |
|
PKa Data Compiled by R. Williams pKa Values INDEX Inorganic 2
pKa Data Compiled by R. Williams. pKa Values Benzene Polycarboxylic acids ... Ring amines and imines (in 80% methyl cellosolve) (ref. 2). Pentamethylene. |
|
Chapter 21: Amines. Organic derivatives of ammonia NH3. Nitrogen
amines are considerably more acidic than alkyl amines (pKa < 5). Nucleophilic ring opening of epoxides with NH3 (Ch. 16.11). |
|
Synthesis and Chemistry of Indole
Indole is a benzo[b]pyrrole formed by the fusion of benzene ring 3.1.1 Protonation: Indole is a very weak base pKa -3.5. The nitrogen atom of indole ... |
|
Benzene & Aromaticity
ring 3 be planar or nearly planar so that there is continuous or nearly continuous 9 95 p-Nitrophenol pKa 7 15 m-Nitrophenol pKa 8 28 Acidity of Phenols |
|
Chapter 17: Alcohols and Phenols
76 Chapter 17: Alcohols and Phenols phenol (aromatic alcohol) pKa~ 10 alcohol Phenols contain an OH group connected to a carbon of a benzene ring 77 |
|
120 Chapter 24: Phenols Alcohols contain an OH group bonded to
bonded to an sp2-hybridized carbon of a benzene ring pKa ~ 16 pKa ~ 10 235 Factors that influence acidity: Inductive effect: CH3CH2OH FCH2CH2OH |
|
PKa Table1
pKa (DMSO) (DMSO) pKa pKa (DMSO) pKa's of Inorganic and Oxo-Acids 8 2 benzene-CO3H Ring amines and imines (in 80 methyl cellosolve) (ref |
|
Approximate pKa chart of the functional groups: values to know 1
Approximate pKa chart of the functional groups: values to know 1 Protonated carbonyl pKa = -7 Other important pKa's 2 Protonated alcohol or ether pKa = -2 to |
|
On the basicity of conjugated nitrogen heterocycles in - FULIR
additional benzene ring does not always lead to basicity increase in solution: its aqueous pKa values of many different types of heterocycles are available |
|
8 - MSU chemistry
and is not soluble in water at any pH p-‐Toluic acid is a weak acid (pKa about 4 5) carbon in the benzene ring and one very low field carbon (at large δ) for |