example of colloid
|
Muddy water animation with different details
22 apr. 2015 An example of colloid is milk. Mud is composed of various sized particles and they produce different effects “depending on their sizes” when. |
|
Chemical Mixtures
An example of a colloid is milk. Milk is a mixture of liquid butterfat globules dispersed and suspended in water. Colloids are generally considered |
|
Some Remarks on Colloid Stability: Selected Examples Taken from
10 dec. 2020 Colloids move as a whole kinetic entity in a fluid: diffusion in water-based media is a typical example. The colloid state thus |
|
0412MOLE - FEATURE.indd
1 iul. 2012 This is an example of a gas-liquid colloid where gas is trapped within a liquid to form a stable mixture. 2. Liquid-liquid colloid (an ... |
|
Solutions Suspensions and Colloids.pdf
Colloids contain some particles that are intermediate between those in a solution and those in a suspension. Examples include peanut butter pudding |
|
Iesc102.pdf
But actually a colloidal solution is a heterogeneous mixture |
|
Introduction
For this reason colloid and interface chemistry are usually treated as a Examples of lyophobic colloidal dispersions are emulsions foams and particle ... |
|
WATER TREATMENT BY MEANS OF COLLOIDS
examples of colloidal solutions are gelatine starch |
|
Monodispersed Colloidal Spheres: Old Materials with New
persed colloidal samples in relatively large quantities.[8] For example the availability of colloidal particles that are uni- form in size and shape plays |
|
Separating live and dead fine roots using colloidal silica: an
dead roots from root samples obtained from forests of Ceriops tagal and Rhizophora sty losa respectively. Live roots of both plant species were separated |
|
Colloids and Surface Chemistry - University of Sydney
Example:blood Solution homogeneous particlesare molecules of one phase in (“solute-like”) cellin entity >> water Colloid size1–1000nm particlesremain suspended id? another ordinarymolecules Suspension heterogeneous particlessettle out Examp Continuousphaseisoftenwater Widerangeofsizes: –bovineserumalbumin:3nm –cells:tensofmicrons |
|
Colloids - University of Cincinnati
Colloids are materials that display phase structures larger than those that display quantum effects (atomic and molecular) but small enough that the structures are sensitive to thermal fluctuations (smaller than 50 µm or the thickness of a hair) that is colloids are affected by Brownian motion and are subject to diffusion |
|
What is an example of a colloid - Lisbdnetcom
Network colloid : consists of two inter-penetrating networks and hard to specify which is dispersed and which is continuous phase (porous glass opal glass gels) Practical examples of colloidal system 10 2 2 Colloid Size The size of dispersed phase falls in the range of 1~1000nm |
|
Colloid - Tishk International University
A colloid is a solution in which the particle size ranges between 10-7cm and 10-5cm For example milk blood honey smoke ink gum starch solution etc Every colloid consists of two parts: colloidal particles or (Dispersed Phase) and the dispersing medium The dispersing medium is the substance in which the colloidal particles are |
|
8 COLLOIDS - The National Institute of Open Schooling (NIOS)
For example colloidal solutions ofstarch gelatin gum etc are prepared by simply dissolving these substances inhot water Similarly a colloidal sol of cellulose nitrate is obtained by dissolving itin alcohol The resulting solution is called collodion However lyophobic colloids cannot be prepared by direct method |
|
Searches related to example of colloid filetype:pdf
Common examples of colloids: The colloidal particles are not necessarily corpuscular in shape In fact these may be rod-like disc-like thin films or long filaments For matter in the form of corpuscles the diameter gives a measure of the particle size |
|
A colloid is one of the three primary types of mixtures with the other
Example of colloids Colloidal AgCl AgI Ag proteinate (effective germicide) colloidal sulphur Many natural and synthetic polymers are important in |
|
8 COLLOIDS - NIOS
For example in a colloidal solution of sulphur in water sulphur particles constitute the 'dispersed phase' and water is the 'dispersion medium' Each of the |
|
Solutions and Colloids
Table 11 1 gives examples of several different solutions and the phases of the solutes and solvents Different Types of Solutions Solution Solute Solvent |
|
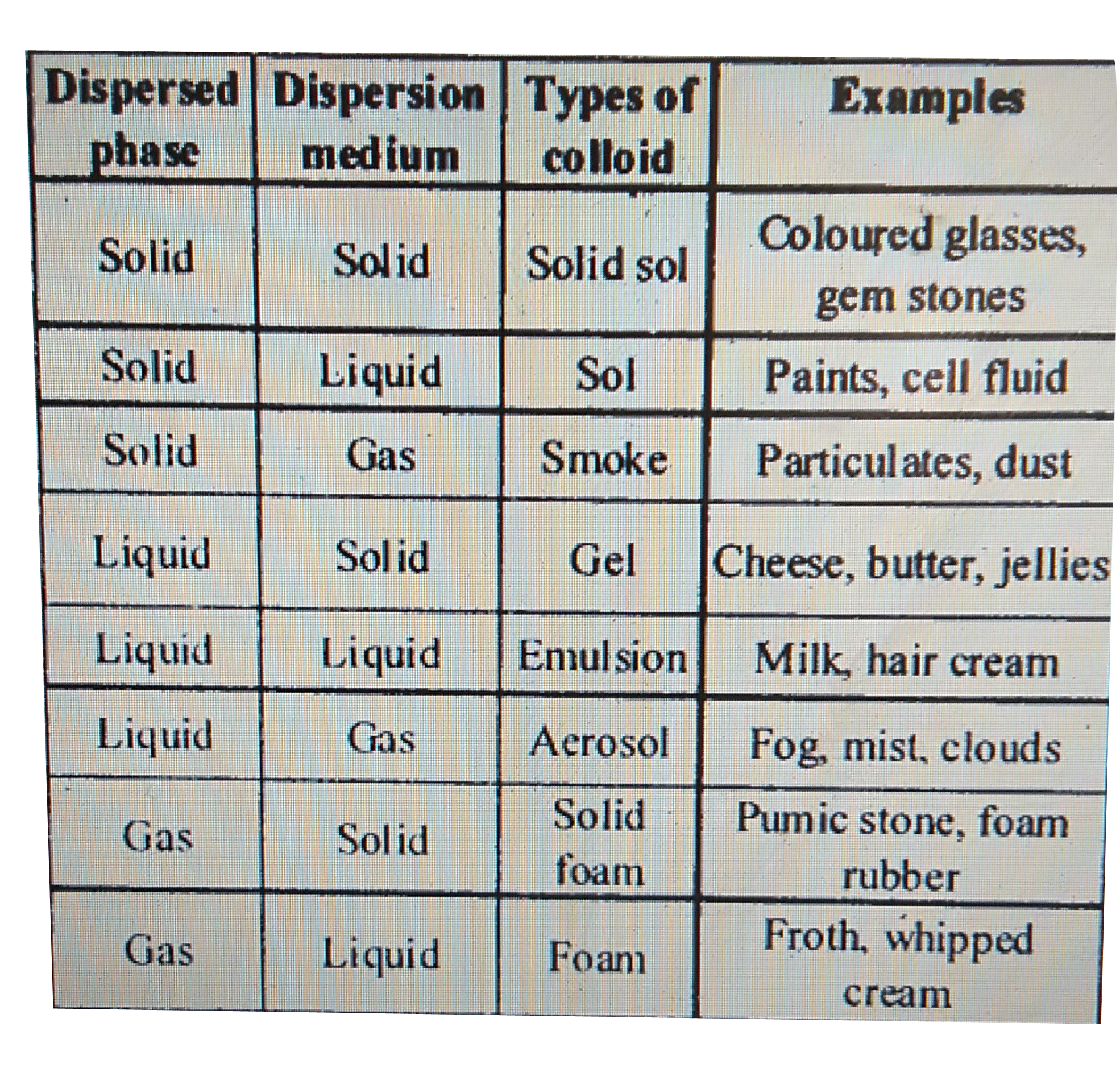
Colloids
Common examples of colloids: Soap bubbles aerosols fog whipped cream butter ice-cream etc Dispersed phase Dispersion medium: A colloidal system is |
|
COLLOID CHEMISTRY
15 avr 2016 · The examples of lyophilic sols are dispersions of starch gum and protein in water ? Lyophobic sols are those in which the dispersed phase |
|
Colloid
For example consider the hydrosol of gold with particles about 4 nm in size This was one of the first sols studied extensively and has interesting properties |
|
Colloidal particles - NCERT
Egg albumin starch and gum are lyophilic sols Freshly prepared ferric hydroxide aluminium hydroxide and arsenic sulphide sols are examples of lyophobic sols |
|
B Sc II-Sem Colloidal state (1) The foundation of colloidal chemistry
For example in the colloidal solution of silver in water (silver acts as a dispersed phase) (ii) External phase or Dispersion medium (continuous phase) |
|
Introduction to the colloid chemistry and surface phenomena
Examples of colloidal systems and surface phenomena from daily life Why the leaves of lotus possess self- cleaning property? |
|
UNIT 7: COLLOIDAL CHEMISTRY
Gelatin dispersed in water is an example of a lyophilic colloidal system Other examples of hydrophilic colloids are biopolymers such as seaweed gums pectic |
What is a colloid and what is an example?
- What is a colloid write 3 examples? Colloids are mixtures whose particles are larger than the size of a molecule but smaller than particles that can be seen with the naked eye. Colloids are common in everyday life. Some examples include whipped cream, mayonnaise, milk, butter, gelatin, jelly, muddy water, plaster, coloured glass, and paper.
What does the term "colloid" mean?
- colloid noun. An intimate mixture of two substances one of which, called the dispersed phase (or colloid), is uniformly distributed in a finely divided state throughout the second substance, called the dispersion medium (or dispersing medium).
Can you describe a colloid?
- In simple terms, we can define colloids as a mixture where one of the substances is split into very minute particles which are dispersed throughout a second substance. The minute particles are known as colloidal particles. Alternatively, we can also say that colloids are basically solutions in which solute particle size ranges from 1nm – 1000 nm.
|
Colloids and Colloidal Systems in Human Health and - MedCrave
27 mai 2016 · solid, liquid or gas colloids dispersed in the medium Essentially, sols in water are examples of lyophobic colloids, that is, they lack the affinity |
|
CHAP 2pmd - NCERT
But actually, a colloidal solution is a heterogeneous mixture, for example, milk Because of the small size of colloidal particles, we cannot see them with naked |
|
Types Of Colloid Solutions - 50000+ Free eBooks in the Genres you
Access Free Types Of Colloid Solutions Types of Colloidal Systems and Examples The species formed is known as the multimolecular colloids For example, |
|
1 Introduction - Wiley-VCH
Examples of lyophilic colloidal dispersions are surfactant micelles, For example, a clay dispersion in water could be classified as a lyophobic colloid with |
|
COLLOIDAL SOLUTIONS
This is light scattering by colloidal solution (for example by dust, fog, milk,etc ) When light beam passes through the colloidal dispersion it is scattered and |
|
Colloidal Solution Types
Solution?: Introduction, Colloid Colloids - Definition, Example and TypesExamples of Colloids - Definition, Types, Examples in Solutions, Suspensions, Colloids |