determining pka of an indicator using spectrophotometry
|
PKa of a dye: UV-VIS Spectroscopy
By using UV-VIS spectroscopy we can determine the pKa of an indicator which are weak acids or bases Our indicator has a protonated and deprotonated form |
|
Experiment &# 11: Spectroscopic determination of indicator pKa
Used in low concentrations these compounds signal pH changes within a specific range determined by the particular indicator in use This color change range |
|
Spectrophotometric Determination of pKa of Phenol Red
In this experiment the pKa of phenol red (an acid-base indicator) is determined through a combination of wet chemistry and spectrophotometric analysis In |
|
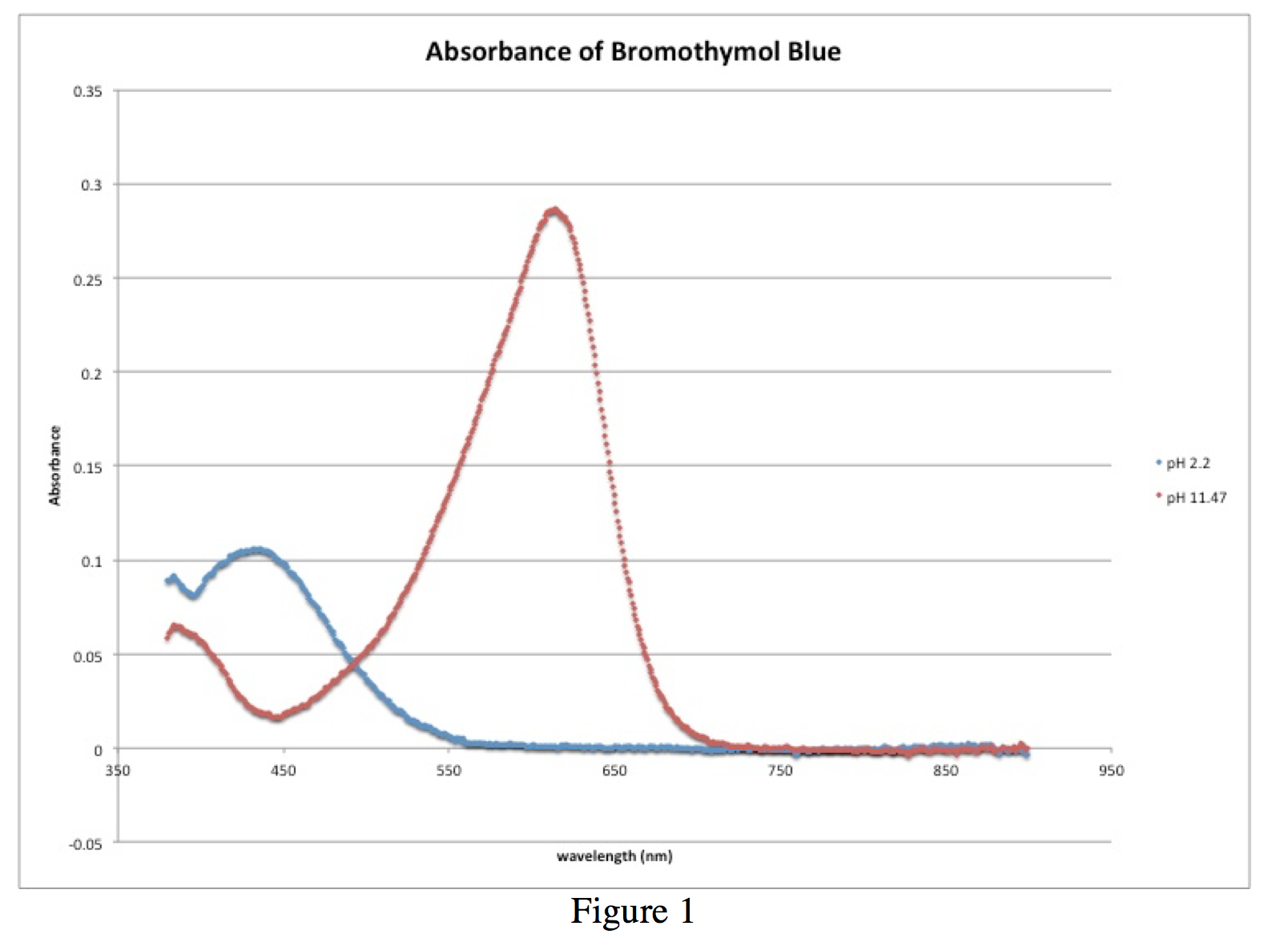
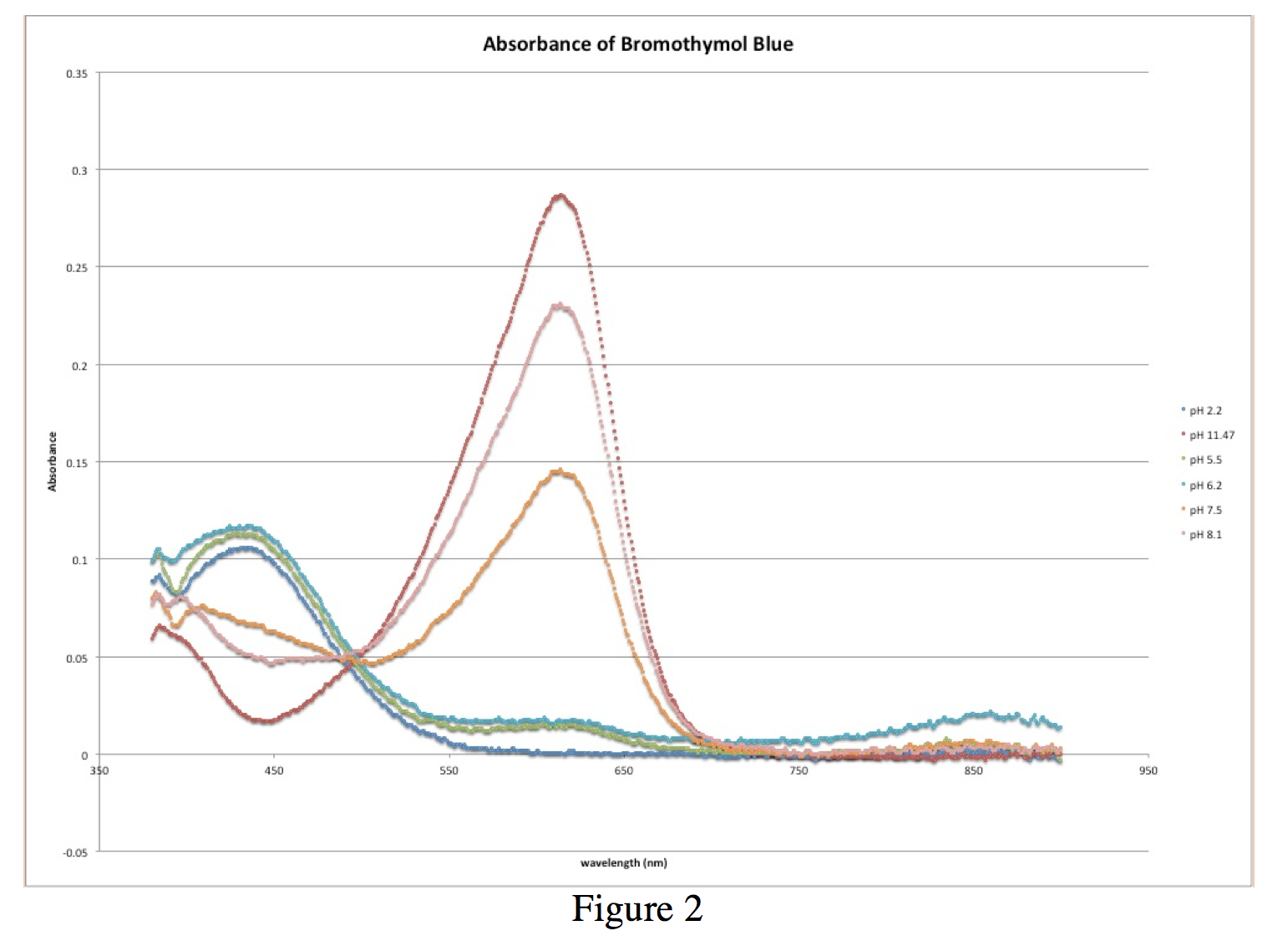
Spectrophotometric Determination of the pKa of Bromothymol Blue
The pKa of the indicator corresponds to the intersection of the line with the pH axis Truman State University CHEM 222 Lab Manual Revised 12/14/04 Page 4 |
|
Spectrophotometric Determination Of The Pka
Connect the points with a smooth line and determine a value of the pKa of the indicator from the inflection point in each plot 2 From each of the two |
|
Spectrophotometric Determination
7 déc 2014 · The pKa and the isosbestic point of the universal pH indicator Carlo Ebra 1-11 (catalog number 45712) were determined using UV-Vis |
In pH-metric methods, pKa is measured by titrating a solution of the sample in water or solvent with acid and base, and calculating the pKa from the shape of the titration. pH-metric methods work for any ionizable compound, but require more sample than UV-metric methods.
What is the technique used for the determination of pKa value?
Potentiometry.
The simplicity and low cost of potentiometric titration has made it one of the most commonly used methods for pKa determination.
In a potentiometric titration, a known volume of reagent is added stepwise to a solution of analyte.
How is UV vis used to determine pKa?
By using UV-VIS spectroscopy, we can determine the pKa of an indicator, which are weak acids or bases.
Our indicator has a protonated and deprotonated form, which absorbs differently at different wavelengths.
Since they absorb differently, the absorbance spectra can be used to determine the value of the pKa.
How do you find pKa from spectrophotometry?
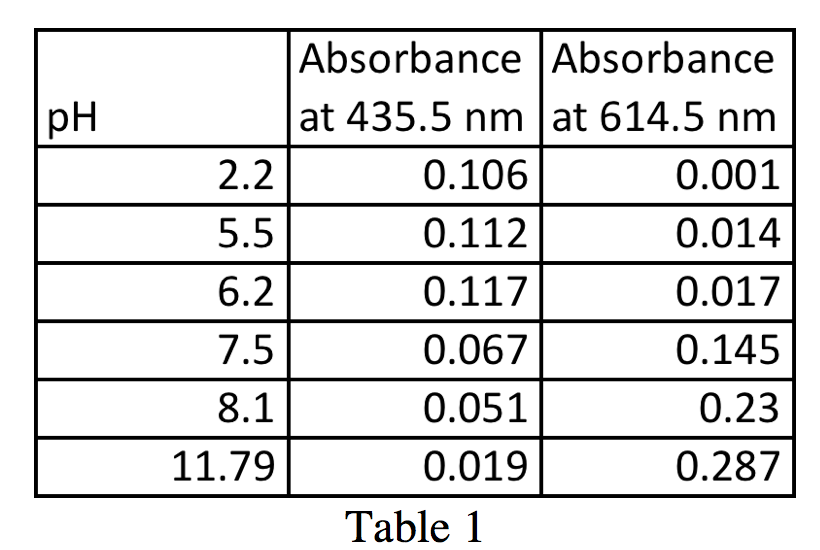
Prepare a plot of absorbance (y axis) as a function of pH (x axis) for the 12 solutions at each of the two wavelengths.
Connect the points with a smooth line and determine a value of the pKa of the indicator from the inflection point in each plot.
|
EXPT. 5 DETERMINATION OF pKa OF AN INDICATOR USING
INDICATOR USING. SPECTROPHOTOMETRY. Structure. 5.1 Introduction. Objectives. 5.2 Principle. 5.3 Spectrophotometric Determination of pKa Value of Indicator. 5.4 |
|
Experiment # 11: Spectroscopic determination of indicator pKa
This time however |
|
Spectrophotometric Determination Of The Pka Of Bromothymol Blue
causing the solution color to change depending on the concentration of each form present. For example in strongly acidic solution |
|
Physical Biochemistry Lab - pKa of a dye: UV-VIS Spectroscopy
By using UV-VIS spectroscopy we can determine the pKa of an indicator |
|
Spectrophotometric Determination of the pKa Isosbestic Point and
07-Dec-2014 The pKa and the isosbestic point of the universal pH indicator Carlo Ebra 1-11 (catalog number. 45712) were determined using UV-Vis ... |
|
Www.rsc.org/methods
Determination of acidity constant of azo indicators by spectrophotometric method introduced for determining the acidity constant pKa |
|
Spectrophotometric Determination of pKa of Phenol Red
In this experiment the pKa of phenol red (an acid-base indicator) is determined through a combination of wet chemistry and spectrophotometric analysis. In |
|
Spectrophotometric Method for Determination of Dissociation
Absolute pKa values of two indicator acids 2 |
|
Considerations for the measurement of spectrophotometric pH for
Spectrophotometry using sulfonephthalein pH indicators is the using the same 10 cm pathlength cell configuration described above. pKa′ determination. |
|
SPECTROPHOTOMETRIC DETERMINATION OF THE pKa OF AN
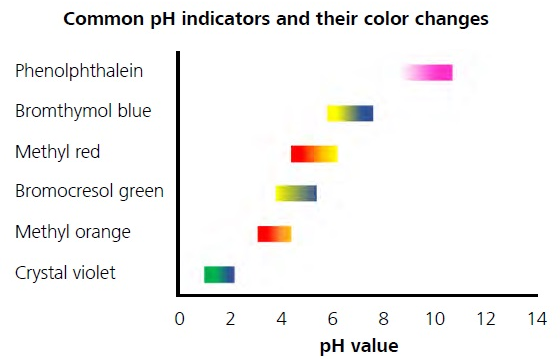
Colorful acid-base indicators are organic weak acids or bases that change color at different pH. In this experiment spectrophotometry is employed to |
|
Spectrophotometric Determination Of The Pka Of Bromothymol Blue
Example absorbance spectra of an acid base indicator in (A) acidic solution (B) basic solution |
|
EXPT. 5 DETERMINATION OF pKa OF AN INDICATOR USING
INDICATOR USING. SPECTROPHOTOMETRY. Structure. 5.1 Introduction. Objectives. 5.2 Principle. 5.3 Spectrophotometric Determination of pKa Value of Indicator. |
|
Experiment # 11: Spectroscopic determination of indicator pKa
This time however |
|
Spectrophotometric Determination of the pKa Isosbestic Point and
7 Dec 2014 The pKa and the isosbestic point of the universal pH indicator Carlo Ebra 1-11 (catalog number. 45712) were determined using UV-Vis ... |
|
Physical Biochemistry Lab - pKa of a dye: UV-VIS Spectroscopy
Using UV-VIS spectroscopy absorbance of different pH solutions around pH 4 By using UV-VIS spectroscopy |
|
Spectrophotometric Determination of pKa of Phenol Red
In this experiment the pKa of phenol red (an acid-base indicator) is determined through a combination of wet chemistry and spectrophotometric analysis. |
|
Chem 151L. Indicators and Simple Acid-Base Calculations
This expression provides an alternative way to determine the pKa of an indicator by plotting pH (y axis) as a function of log([In-]/[HIn]). As shown in Figure 2 |
|
What Can Be Measured in a Colorful Equilibrium Reaction
range of 1.4 bracketing the expected pKa of the indicator. Measuring [H3O+]. For each solution the [H3O+] will be determined by measuring the pH using a pH |
|
The pKa of methyl red will be determined by measuring
The acid form of the indicator which we will designate as HMR |
|
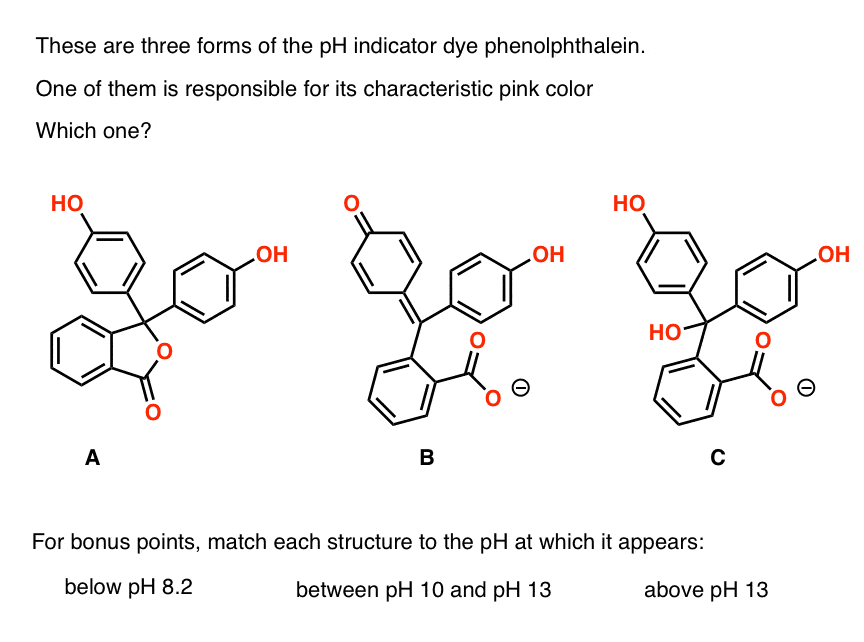
Determination of the pKa Value of Phenolphthalein by Means of
While there have been several experimental articles published in education papers applying spectroscopy to determine the pKa of acid-base indicators [1011] |
|
Spectrophotometric Determination Of The Pka Of - Chem Lab
1 Prepare a plot of absorbance (y axis) as a function of pH (x axis) for the 12 solutions at each of the two wavelengths Connect the points with a smooth line and determine a value of the pKa of the indicator from the inflection point in each plot 2 |
|
Experiment &# 11: Spectroscopic determination of indicator pKa - ULM
the pH of each buffer mixture using the original Henderson-Hasselbalch equation : pH = pKa + log ([salt] / [acid]) The pKa in this equation is for the dissociation of |
|
A Simplified Method for Finding the pKa of an Acid–Base Indicator
3 mar 1999 · chemistry laboratory courses is the spectrophotometric determination of the pKa of using eight common indicators are reported here For the simple pKa – pH (4) The pKa of an indicator can be determined by either of two |
|
EXPT 5 DETERMINATION OF pKa OF AN INDICATOR USING
A typical spectrophotometric determination of the pKa value of the indicator consists of the following steps A solution of a known concentration of the indicator is |
|
PKa of Methyl Red - Colby College
This technique is not restricted to indicators, and can be used with any substance whose In this experiment we will determine this equilibrium constant, pKa In this experiment you will use an Ocean Optics diode array spectrophotometer |
|
Spectrophotometric Determination of the pKa, Isosbestic Point and
7 déc 2014 · The pKa and the isosbestic point of the universal pH indicator Carlo Ebra 1-11 ( catalog number 45712) were determined using UV-Vis |
|
SPECTROPHOTOMETRIC DETERMINATION OF THE pKa OF AN
The solutions contain the same total concentration of indicator, [In-] + [HIn], but the ratios vary with pH Figure 4 shows a typical plot of absorbance vs pH at the |
|
Pka of Indicators
below represents a solution of the indicator, as the pH increases from pKa -1 For each solution the [H3O+] will be determined by measuring the pH using a spectrophotometer can be used to measure how much of a substance is present |
|
Physical Chemistry - LD Didactic
Acid-base indicators, e g bromothymol blue, are weak acids or the concentrations of both forms are determined using the absorbance maxima, and from this the pKa value of bromothy- mol blue is 1 Compact spectrometer USB, complete |
|
Chemistry 321: Quantitative Analysis Lab Webnote
experiment, the pKa of phenol red (an acid-base indicator) is determined through a combination of wet chemistry and spectrophotometric analysis In lecture, we |