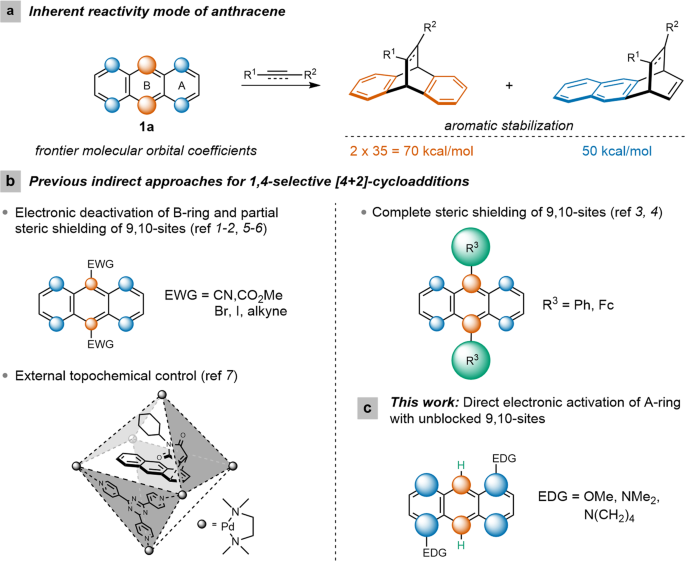
electron withdrawing groups diels alder
|
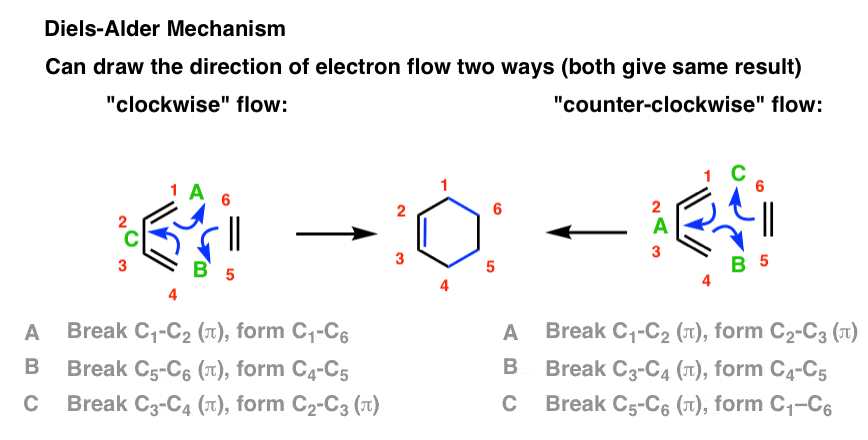
Diels-Alder Reaction
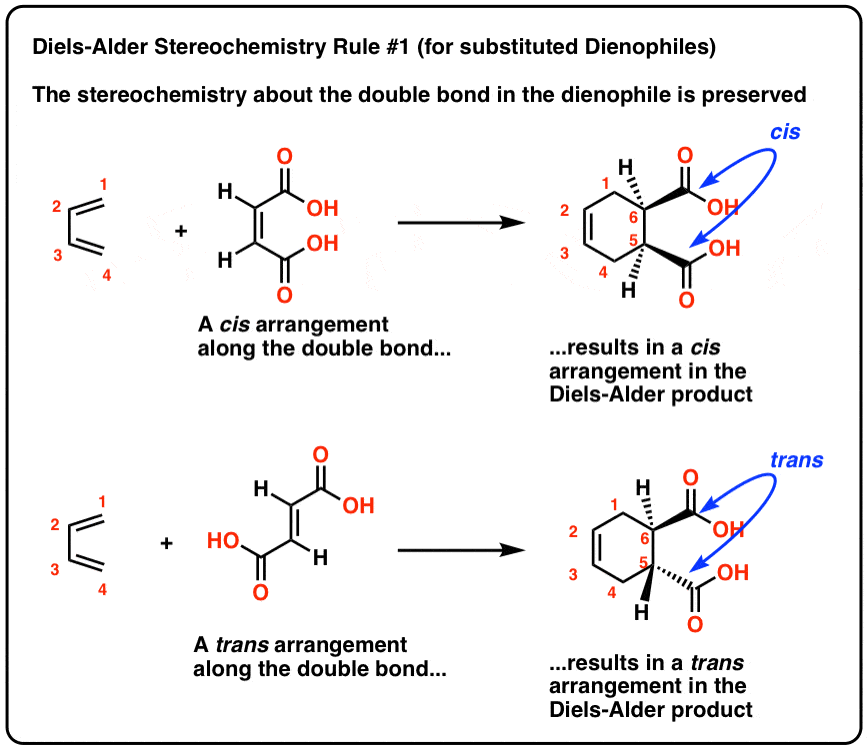
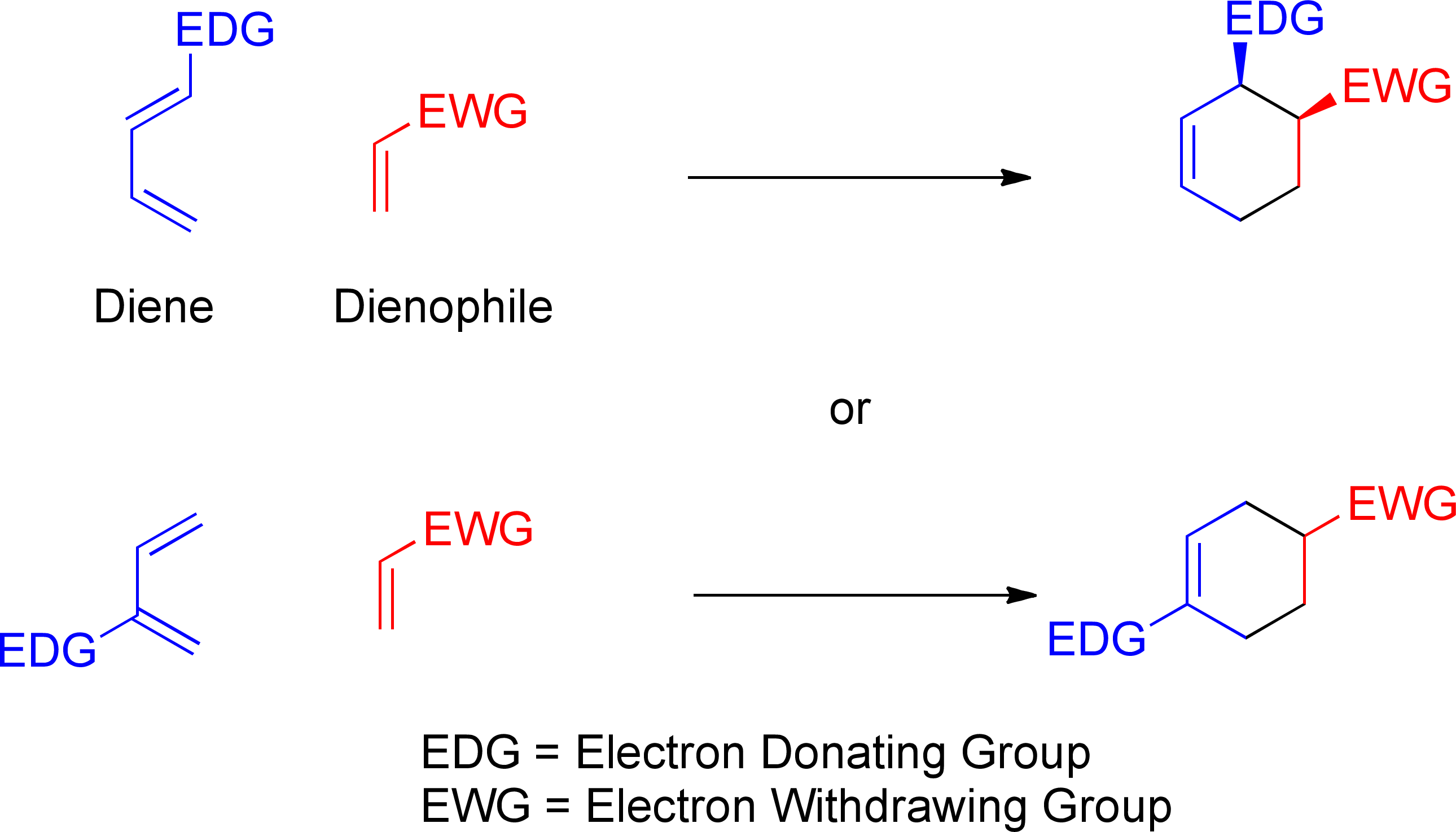
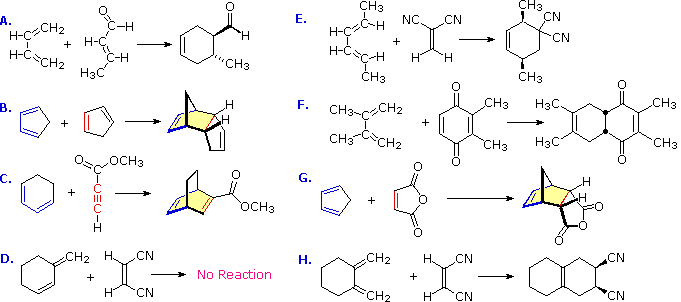
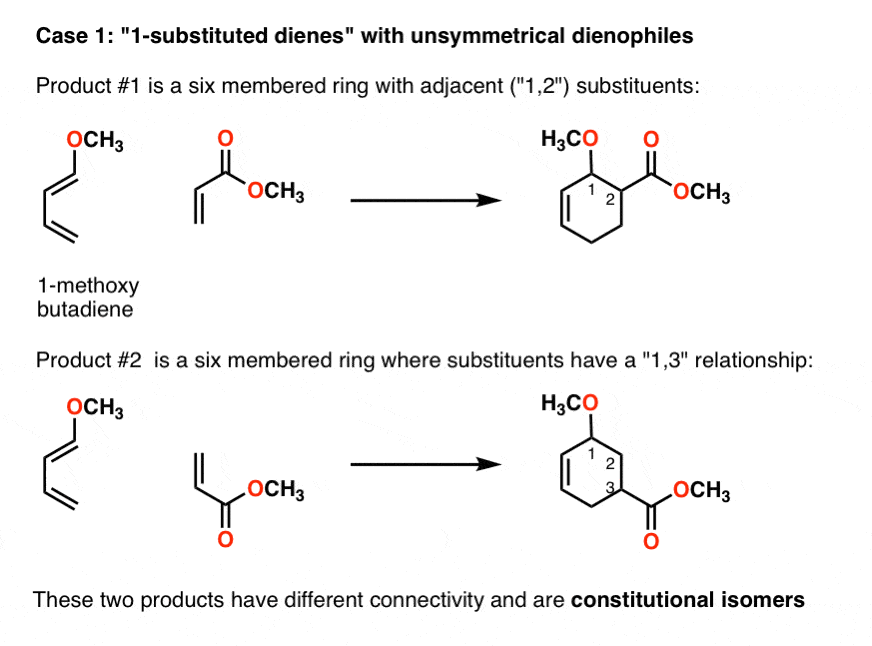
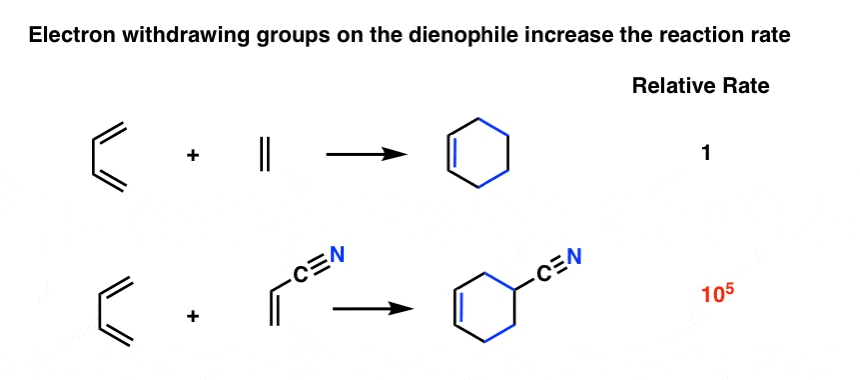
Diels-Alder Reaction Diels-Alder Reaction (a very important reaction) Reaction between a conjugated diene and an alkene (dienophile) to give a cyclohexene Diene Dienophile cyclohexene ‡ The Diels-Alder reaction is favored by electron withdrawing groups on the dienophile and electron donating groups on the diene Good Dienophiles: |
Can alkynes serve as dienophiles in Diels-Alder reactions?
Alkynes can also serve as dienophiles in Diels-Alder reactions: In general, Diels-Alder reactions proceed fastest with electron-donating groups on the diene (eg. alkyl groups).
How does a Diels Alder reaction work?
In general, Diels-Alder reactions proceed fastest with electron-donating groups on the diene (eg. alkyl groups). The Diels-Alder reaction is a single step process, so the diene component must adopt an s-cis conformation in order for the end carbon atoms (#1 & #4) to bond simultaneously to the dienophile.
How do dienes form and interact before dies Alder reactions?
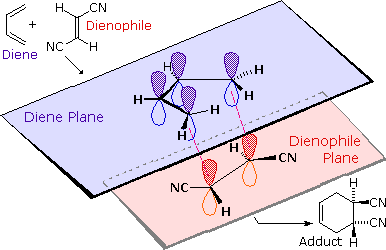
Show more... How do Dienes form and interact prior to dies alder reactions? The Diels-Alder reaction is a cycloaddition of a 4 pi + 2 pi (diene + dienophile) system that forms a more stable product due to the fact that the sigma bonds created are more stable than the pi bonds destroyed.

Diels-Alder reaction Organic chemistry Khan Academy

Diels-Alder: stereochemistry of diene Organic chemistry Khan Academy

Diels-Alder: endo rule Organic chemistry Khan Academy
|
Diels-Alder Reaction (a very important reaction) Reaction between a
The Diels-Alder reaction is favored by electron withdrawing groups on the dienophile and electron donating groups on the diene. Good Dienophiles:. |
|
Vinylboronates P-substituted by an electron withdrawing group
Vinylboronates P-substituted by an electron withdrawing group : synthesis and Diels-Alder reactivity of a new type of electron deficient olefins. |
|
Summary of Diels-Alder Rules: 1.) Diene must be able to adopt an s
Dienophile should have at least one electron withdrawing group on it. Electron withdrawing groups are carbonyls nitriles |
|
Experiment 10. The Diels-Alder Reaction [see: Eges 18.4 pp 742
27 May 2011 Dienophiles: More electron-deficient dienophiles undergo faster Diels-Alder Reactions. X: electron-donating groups such as -OCH3 and -NRR'. |
|
UNIVERSITY OF CALIFORNIA Los Angeles Diels-Alder Reactivities
Diels-Alder Reactivities of Cyclic Dienes and Dienophiles provide syn adducts while electron-donating groups provide anti adducts. Structural. |
|
Article - Development and application of Diels-Alder adducts
16 Feb 2022 group (7c) has stronger electron-withdrawing properties than the sulfonyl fluoride inducing an $10 nm red shift in the absorption spectrum when ... |
|
Experiment 13: The Diels-Alder Reaction of a Conjugated Diene in
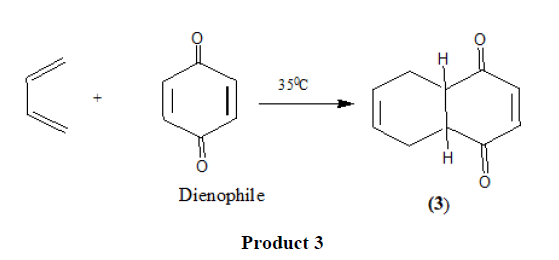
electron withdrawing carbonyl groups. In fact maleic anhydride was one of the original dienophiles that Diels and Alder used in their studies. |
|
Lewis acid catalysis of Diels-Alder reactions
as expected on inductive grounds.8 For alkyl groups the two coefficients are nearly identical in the LU.17·28. Electron-withdrawing groups have exactly the |
|
Lecture 23 Organic Chemistry 1
6 Apr 2010 Reactivity of Diels-Alder. 4. Electron donating groups (EDG) increase the reactivity of the diene = faster reaction. Electron withdrawing ... |
|
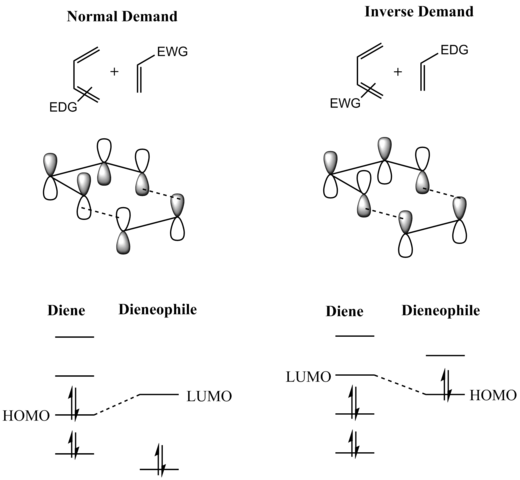
Electron Deficient Dienes I. Normal and Inverse Electron Demand
a diene moiety bearing electron withdrawing groups at the l and 3 positions and its completely regiosdective inverse electron demand Diels-Alder. |
|
Diels-Alder Reaction (a very important reaction) Reaction between a
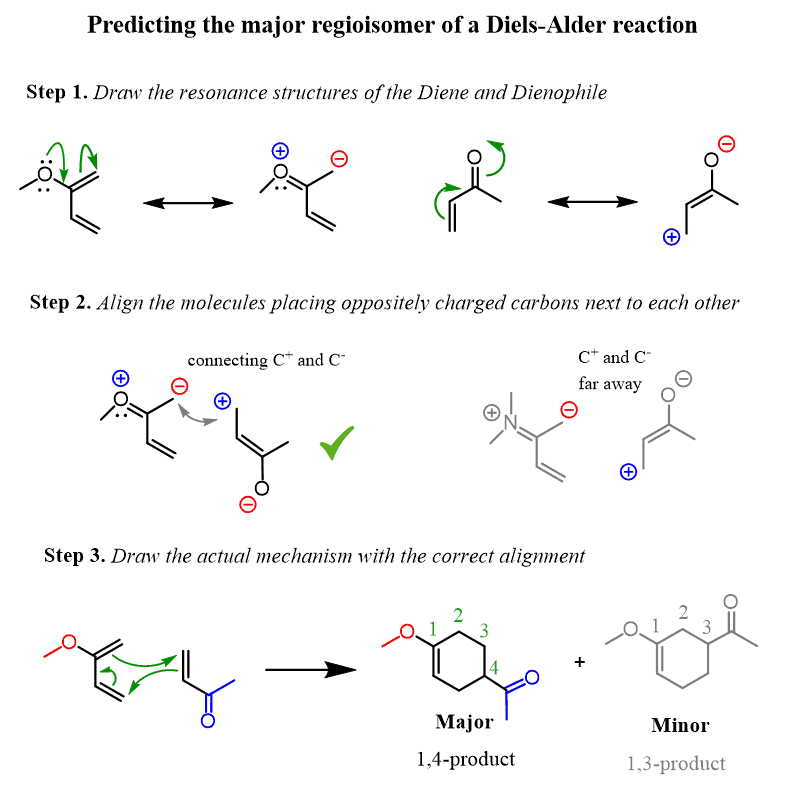
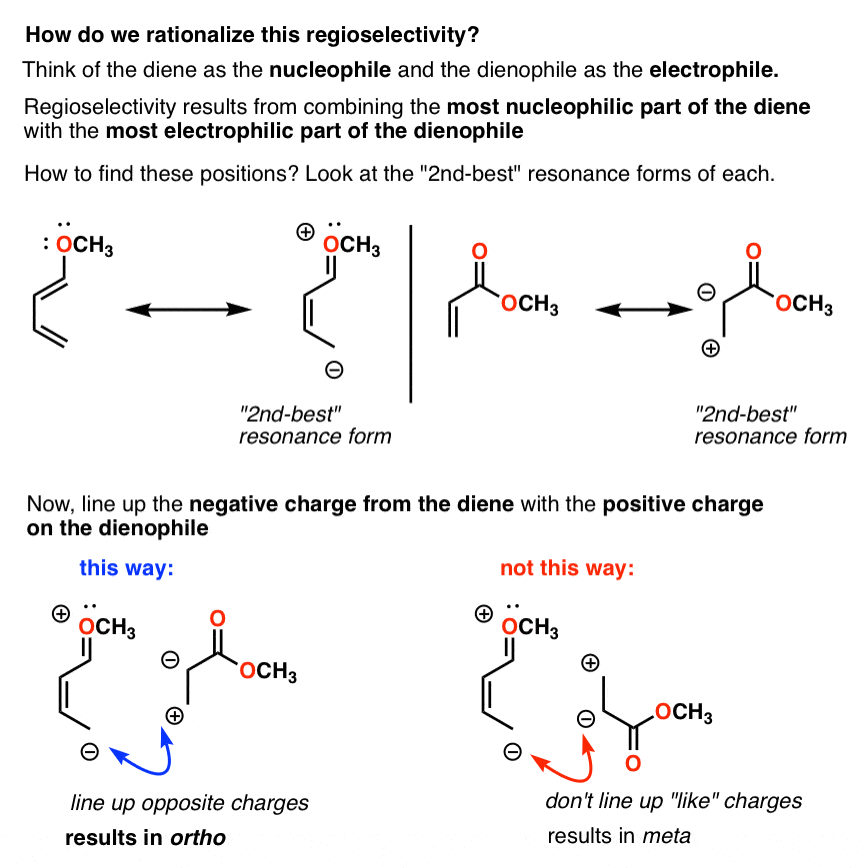
Typical electron donating groups on the diene are ethers, amines and sulfide; all have a non- bonding pair of electrons to donate The regiochemistry of this Diels-Alder reaction is explained by looking at the dipolar resonance structures |
|
Substituent Effects on Simple Diels-Alder Reactions - IJCRCPS
Normal electron demand Diels Alder reactions are promoted by electron donating substituent on diene and electron withdrawing substituent on the dienophile In contrast, inverse electron demand Diels Alder reactions are accelerated by electron withdrawing groups on diene and electron donating ones on dienophile |
|
Diels-Alder Reaction - Dr May Group
EWG (electron withdrawing group) = COR, CO2R, COAr, NO2, etc HOMO LUMO HOMO LUMO or Reviews: Nicolaou et al Angew Chem |
|
Diels–Alder reaction
EDG = electron-donating group; EWG = electron-withdrawing group Example 16 OMe CO2Et Me3SiO OMe CO2Et H Me3SiO AcO OAc hydroquinone |
|
Experiment 10 The Diels-Alder Reaction [see: Eges 184, pp 742
27 mai 2011 · Dienophiles: More electron-deficient dienophiles undergo faster Diels-Alder Reactions X: electron-donating groups such as -OCH3 and -NRR' |
|
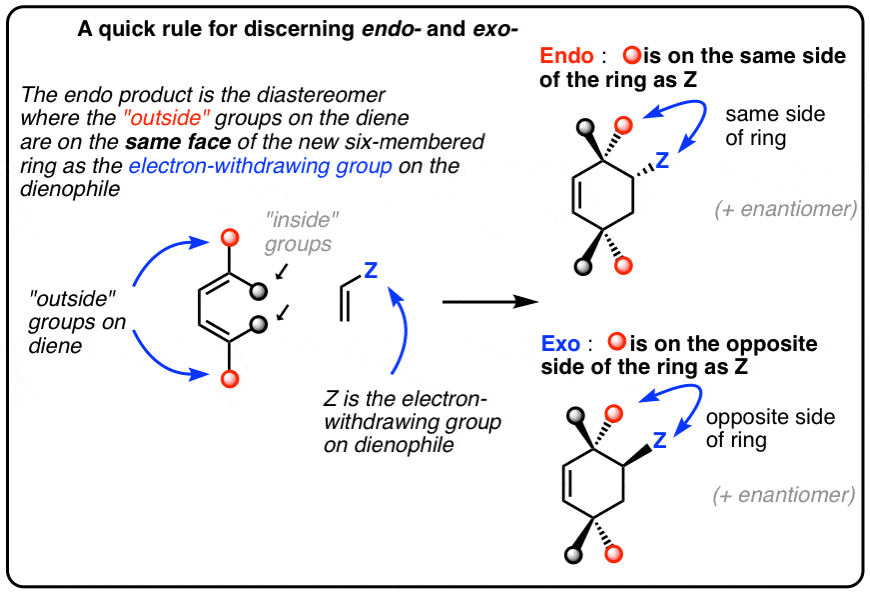
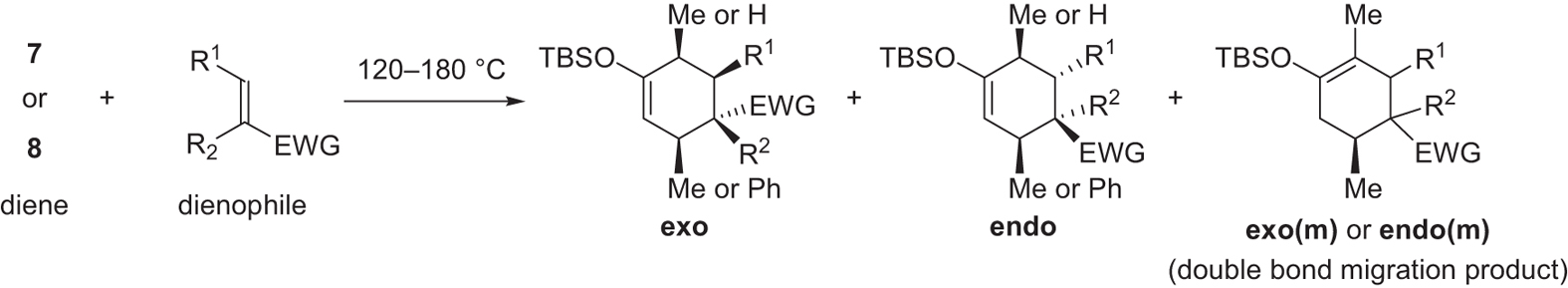
Unconventional exo selectivity in thermal normal-electron - Nature
12 oct 2016 · Figure 1 General stereoselective prediction for normal-electron-demand Diels– Alder reactions EWG = electron-withdrawing group, EDG = |
|
THE VIRTUAL KINETICS OF A DIELS-ALDER CYCLOADDITION
The Diels-Alder reaction, a [4+2] cycloaddition of a conjugated diene and an alkene as hydroxy and alkoxy) on the diene, and/or electron-withdrawing groups |
|
Lecture 23 Organic Chemistry 1 - UIC Chemistry - University of
6 avr 2010 · Reactivity of Diels-Alder 4 Electron donating groups (EDG) increase the reactivity of the diene = faster reaction Electron withdrawing groups |