identify the various types of solutions. describe the characteristics of each type of solution
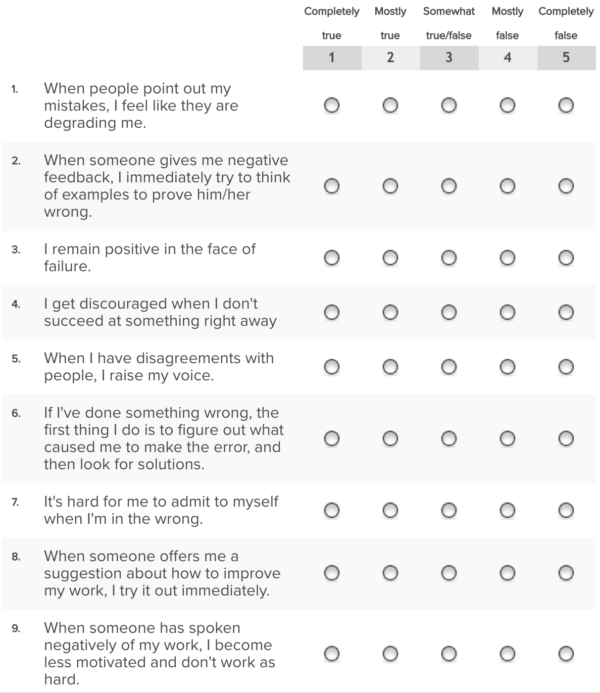
|
Characteristics of Solutions: Solutions are homogeneous mixtures
A solution can exist as a solid liquid or gas depending on the state of the solvent. Types of Solutions and Examples. Type of Solution. Example. Solvent. |
|
4.1 Preparing policy briefs 4.2 More reporting formats 4.3 Writing
identify two types of policy briefs (advocacy and objective);. describe the characteristics of a policy brief; What are the various options? |
|
IASC FrAmework on Durable SolutionS for internally DiSplaceD
? what Is a durable solutIon For Internally ? what crIterIa deterMIne to what extent a durable solutIon ... different types of durable solutions. |
|
Common European Framework of Reference for Languages
experimenting with alternative forms of expression in different languages or dialects the identification of text types and forms |
|
ICH Topic Q 2 (R1) Validation of Analytical Procedures: Text and
TYPES OF ANALYTICAL PROCEDURES TO BE VALIDATED Different validation characteristics are required for a quantitative test than for ... Type of analytical. |
|
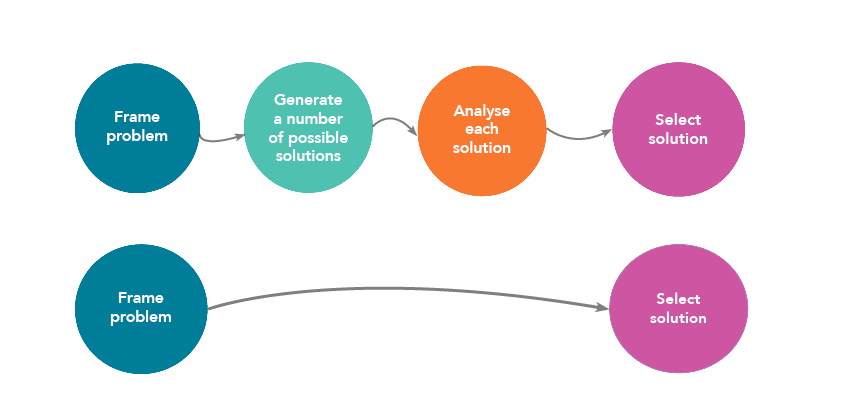
Linear Programming
mathematical function of the decision variables that converts a solution into Each example has a name describing the type of prob-. |
|
Chemical Mixtures
One of the main aspects of chemistry is combining different substances. Sometimes specific types of mixtures including solutions alloys |
|
MEASURING STUDENT KNOWLEDGE AND SKILLS
How OECD/PISA is different from other international assessments . identification of a set of key characteristics that should be taken into account when ... |
|
Academic Standards for Science and Technology
Jan 5 2002 students' abilities to identify problems and determine solutions. ... Identify and describe different types ... impacts of the solution. |
|
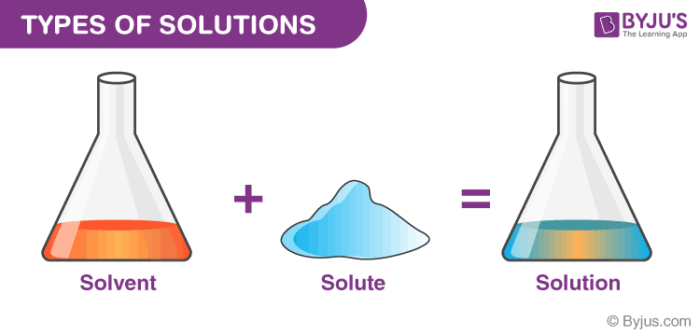
TYPES OF SOLUTIONS
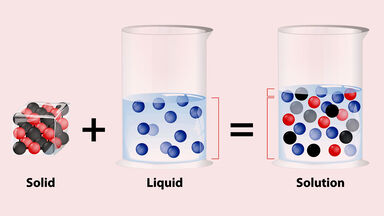
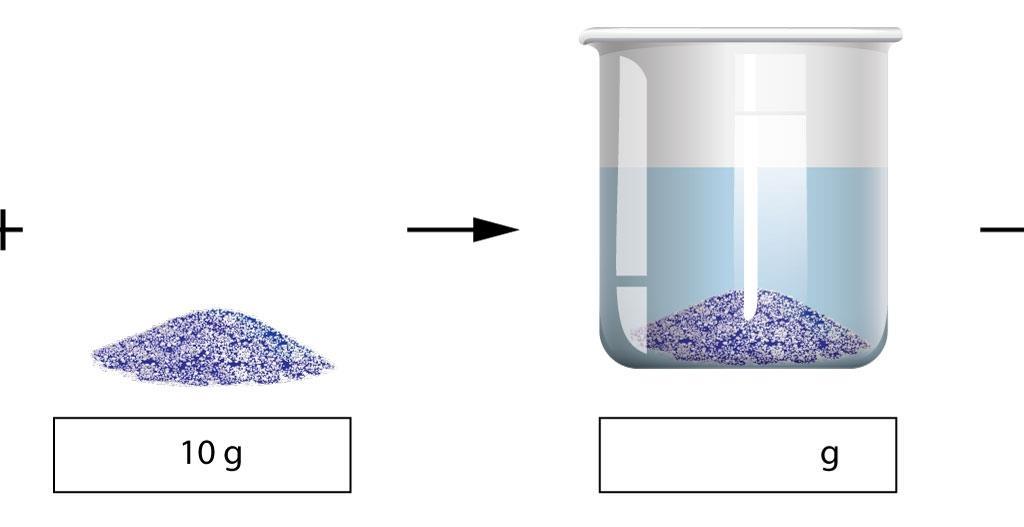
TYPES OF SOLUTIONS A solution is a homogeneous mixture of two substances: a solute and a solvent Solute: substance being dissolved; present in lesser amount Solvent: substance doing the dissolving; present in larger amount Solutes and solvents may be of any form of matter: solid liquid or gas Some Examples of Solutions |
|
TYPES OF SOLUTIONS
describe the relationship between the concentration of ouabain and the Na /K ATPase ++ activity both in normal human kidney (NHK) cells AND in PKD cells Accept one of the following: • Increasing concentrations of ouabain result in decreasing ATPase activity (in both types of cells) |
|
Type of Solution Example Solvent Solute - Wylie
Types of Solutions 1 When a solvent has dissolved all the solute it can at a particular temperature the solution is said to be saturated 2 Unsaturated solutions have dissolved some solute but can dissolve more 3 When a solution is heated and saturated then if it is allowed to cool gently it can become supersaturated |
|
Chapter 9 Solutions Practice Problems Section 91 Solutions
Goal: Identify the solute and solvent in a solution; describe the formation of a solution Summary: A solution forms when a solute dissolves in a solvent The particles of the solute are evenly dispersed throughout the solvent The solute and solvent may be a solid liquid or gas |
|
21 Solutes and Solvents
are the parts of the solution that dissolve Solutions are generally made by dissolving one or more solutes in a solvent Liquid Solutions You are probably most familiar with solutions that are liquids ! ese all have liquid solvents In food preparation the solvents are usually liquids like water or vegetable oil In iced tea water is the |
What are the two types of solutions?
- TYPES OF SOLUTIONS. ? A solution is a homogeneous mixture of two substances: a solute and a solvent. ? Solute: substance being dissolved; present in lesser amount. ? Solvent: substance doing the dissolving; present in larger amount. ? Solutes and solvents may be of any form of matter: solid, liquid or gas.
What are the characteristics of solutions?
- Characteristics of Solutions: Solutions are homogeneous mixtures containing two or more substances called the solute and solvent. The solute is the substance that dissolves.
What is the difference between a solution and a dissolving medium?
- Solutions are homogeneous mixtures containing two or more substances called the solute and solvent. The solute is the substance that dissolves. The solvent is the dissolving medium.
Is a solution a solid or liquid?
- A solution can exist as a solid, liquid or gas depending on the state of the solvent. Remember that a substance that dissolves in a solvent is said to be soluble in that solvent. A substance that does not dissolve in a solvent is insoluble.
|
Characteristics of Solutions: Solutions are homogeneous - Wylie ISD
Solutions are homogeneous mixtures containing two or more substances called Types of Solutions and Examples Solvent particles surround solute particles to form a solution in a process called Amount of solute: All tables and figures showing solubility MUST indicate the showing the solubility of several different |
|
14 Mixtures and Solutions
3 Identify the various types of solutions Describe the characteristics of each type of solution All solutions are homogeneous mixtures containing two or more |
|
TYPES OF SOLUTIONS
A solution is a homogeneous mixture of two substances: a solute and a solvent Identify the predominant particles in each of the following solutions and write the Many factors such as type of solute, type of solvent and temperature affect the What is the mass (m/m) of a solution prepared by dissolving 30 0 g of NaOH |
|
Suspensions and solutionspdf - Madison County Schools
Identify each mixture as a solution, colloid, or suspension Explain httA LLLLLLLLLLL The particles of ice cream are dispersed in many tiny bubbles of air Ice cream also Write the letter of the correct answer on the line at the left 1 Which of 10 Co laid A characteristic property of a(n) solution is that it can scatter light |
|
A solution is a type of mixture
BD Chapter 4: Solutions 111 VOCABULARY solution p 111 solute p 112 solvent p 112 other types of mixtures that its ingredients cannot be identified as different substances A What is the difference between a solute and a solvent? |
|
Activity 3 Solutions, Suspensions, and Colloids
One way to get different types of materials is to just mix them together Lots of different things can happen when materials are mixed Each kind of mixture has its own characteristics Be prepared to discuss your responses Determine why certain kinds of In a solution, the particles that dissolve are so tiny they can 't |
|
Laboratory
: Mixtures & Solutions - RIC
Heterogeneous mixtures consists of visibly different substances a pizza, the composition of sand, and the grainy appearance of wood indicate that Each component will have its own characteristic balance of You will determine the ANSWER: (b) Water, H2O, is a chemical compound made of the elements hydrogen |
|
Solubility and dissolution - Pharmaceutical Press
is a solution? • What is the difference between solubility and dissolution? Solubility and dissolution are different concepts, but are related Solubility is the of solute that the pure solvent can hold in solution, at specified environmental conditions Within 'solid inliquid solutions,' there are two major types of dissolution |
|
Mysterious Mixtures Experiment - Dow Corporate
In liquid form, homogeneous mixtures are generally In this experiment, different solutions are made by Discuss mixtures and the two types of Define and identify solutions and the parts of a solution • Understand separation processes |
|
Classification of Matter
What words would you use to describe different kinds of matter? What You'll Learn solutions, colloids, and might recognize are helium, aluminum, water, and salt What are solution: a homogeneous mixture of particles so small that |