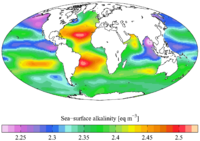
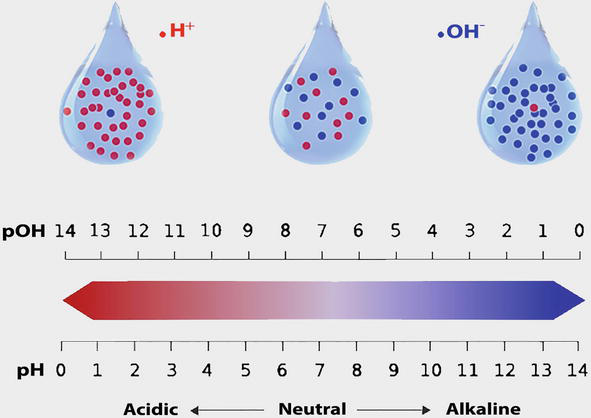
importance of alkalinity in water
|
The Importance Of Alkalinity In Coagulation (Inorganic Coagulant
Why the Alkalinity is important in coagulation and pH control: The chemical reaction between the coagulant and the water can consume bicarbonates. If. |
|
Water Quality Notes: Alkalinity and Hardness1
The objectives of this document are to provide readers with 1) an overview of alkalinity and hardness. 2) an understanding of the importance of alkalinity and. |
|
Chapter 11: pH and Alkalinity Voluntary Estuary Monitoring Manual
Since the pH of water is critical to the survival of most aquatic plants and animals monitoring pH values is an important part of nearly every water quality. |
|
Using this fact sheet Interpreting Drinking Water Quality Results
important characteristics of your well water water or boiling water for 5 minutes before using for drinking or cooking ... alkalinity in water. The. |
|
Importance of Hydroxide Ion Conductivity Measurement for Alkaline
26 mai 2022 †. These authors equally contributed to this work. Abstract: Alkaline water electrolysis (AWE) refers to a representative water electrolysis ... |
|
Alkalinity Alkalinity is a chemical measurement of a waters ability to
Nonetheless highly acidic and alkaline waters are considered unpalatable. Knowledge of these parameters may be important because: (1) The alkalinity of a |
|
Alkalinity and Hardness in Production Ponds
www.ca.uky.edu/wkrec/Wurtspage.htm. Alkalinity and hardness are both important components of water quality. However these two aspects of water chemistry |
|
IR 322 The importance of hardness and alkalinity in assessing the
The effect of water hardness (Ca and Mg) varied depending on increase in alkalinity (carbonate) had no effect on Cu toxicity to H. viridissima while it. |
|
{Paper Title}
Alkalinity is important for the coagulation process and in maintaining a stable final water quality that minimises corrosion of the supply pipework. |
|
PREDICTING LAKE ALKALINITY AND DEPTH FOR CLASSIFYING
23 déc. 2016 most important variable for predicting water depth was the range of slope within 50m ... importance of lake alkalinity mean depth and size. |
|
Alkalinity - Kennesaw State University
Alkalinity is important to aquatic organisms because it protects them against rapid changes in pH Alkalinity is especially important in areas where acid rain is a problem Sources One source of alkalinity is calcium carbonate (CaCO3) which is dissolved in water flowing through geology that has limestone and/or marble |
|
Alkalinity - Thermo Fisher Scientific
Alkalinity is important for: the water softening process for corrosion control for disinfection coagulation and the oxidation process Drinking water plants report source water alkalinity along with TOC to demonstrate compliance with the Disinfectants and Disinfection Byproducts Rules (DBPR) Sampling and Storage for Alkalinity Samples |
|
Water Quality Notes: Alkalinity and Hardness
sample Water with low levels of alkalinity (less than 150 mg/L) is more likely to be corrosive High alkalinity water (greater than 150 mg/L) may contribute to scaling If total hardness is half or less the alkalinity result it may indicate that your water has passed through a water softener |
|
An Approach towards the Determination of Alkalinity in Water
The alkalinity of water is determined by titrating the water sample with Standard acid solution (HCl) Alkalinity of water is attributed to the presence ofOH?CO?²? and HCO??ions The titration is performed volumetrically using phenolphthalein and methyl orange indicators |
|
Standard Operating Procedure for - CCAL
1 2 The alkalinity of water is its acid-neutralizing capacity comprised of the total of all titratable bases Alkalinity is a measure of the sum of properties of water and can be interpreted in terms of specific substances only when the chemical composition of the sample is known |
|
Searches related to importance of alkalinity in water filetype:pdf
Alkalinity is important for fish and aquatic life because it protects or buffers against pH changes (keeps the pH fairly constant) and makes water less vulnerable to acid rain The main sources of natural alkalinity are rocks which contain carbonate bicarbonate and hydroxide compounds |
What is the significance of determining alkalinity in water?
- Significance Alkalinity Alkalinity is important because it buffers the pH of water within the system. Without this buffering capacity, small additions of acids or bases would result in significant changes of pH, which could be deleterious for aquatic life. Alkalinity also influences the distribution of some organ-isms within aquatic systems.
What does alkalinity in water mean?
- What is Alkalinity of Water? Alkalinity of water means acid neutralization capacity of water. When you add acid in water (adding H + ions) water absorbs H + ions without showing significant change in pH. Mainly, it is due to carbonate, bicarbonate & hydroxide ion present in water or the mixture of two ions present in water.
Is alkaline water really better than ordinary water?
- While we had trouble finding any sort of proof that alkaline water is healthier than regular water, we also couldn’t find any proof that it is detrimental to your health either. Although some have brought up the possibility that drinking alkaline water might disrupt the acidity of the stomach which could disrupt digestion.
|
Alkalinity
Carbonates and hydroxide may be significant when algal activity is high and in certain industrial water and wastewater, such as boiler water Alkalinity is significant in the treatment processes for potable water and wastewater The alkalinity acts as a pH buffer in coagulation and lime-soda softening of water |
|
PH and Alkalinity - US EPA
Testing water samples for total alkalinity measures the capacity of the water to neutralize acids This test is important in determining the estuary's ability to |
|
PH Alkalinity Hardness pH Alkalinity Hardness - Wisconsin DNR
Alkalinity Hardness Importance of General Chemistry Relationships in Water Treatment George Bowman Inorganics Manager State Laboratory of Hygiene |
|
The Role of Alkalinity in Aerobic Wastewater Treatment Plants
16 jan 2015 · Alkalinity can be defined as the ability of a water to neutralize acid or to absorb hydrogen ions In order to maintain these optimal pH conditions for biological activity there must be sufficient alkalinity present in the wastewater to neutralize acids generated by the active biomass during waste treatment |
|
Alkalinity 101
The alkalinity of water is a measure of its capacity to neutralize Why is alkalinity important to Wastewater Alkalinity is lost in an activated sludge process |
|
PH in Drinking-water - WHO World Health Organization
Originally published in Guidelines for drinking-water quality, 2nd ed Vol 2 Health The pH is of major importance in determining the corrosivity of water Rose P Alkaline pH and health: a review prepared for the Water Research Centre |
|
The alkalinity of a solution may be defined as the capacity for solutes
aliphatic acid anions to titratable alkalinity in water from certain oil fields The sensitivity toward pH clhange through this effect The definition of alkalinity, or |
|
PH and Alkalinity - College of the Environment and Life Sciences
Why is pH important? Aquatic organisms need the pH of their water body to be within a certain range for optimal growth and survival Although |