is iron a pure substance
|
Chemistry: Classifying Matter Name______________________
If the material is a pure substance further classify it as either an element or compound in the right column iron filings (Fe). Pure Substance. Element. |
|
Untitled
Partially molten iron pure. Suss (colloin. 3). Indicate whether each of the following materials is a pure substance |
|
Safety Data Sheet: Iron(III) chloride
Product identifier. Identification of the substance. Iron(III) chloride ?985 % |
|
Pure Element Mixture Suspensions Heterogeneous Mixture
Pure Substance or Mixture. Element or Compound. Homo/Heterogeneous |
|
Untitled
If the material is a pure substance further classify it as either an element or compound in the right column. iron filings (Fe) limestone (CaCO3). |
|
Lesson 10: Iron in Cereal Experiment
Students will use magnets to pull the iron from the cereal mixture. The lesson can be simplified to illustrate the difference between pure substances and |
|
Chapter 1—The Atomic Nature of Matter
A pure solid decomposes on heating yielding a solid and a gas |
|
MIXTURES ELEMENTS AND COMPOUNDS
For example sea water air |
|
Safety Data Sheet: Iron(III) chloride hexahydrate
1.1. Product identifier. Identification of the substance. Iron(III) chloride hexahydrate ? 98% extra pure. Article number. |
|
Lab #2
The basis of the separation is the fact that each component has a different set of physical and chemical properties. The components are pure substances which |
|
Thermodynamics Properties of Pure Substance - Sistemas EEL
substance is can still a pure substance if it is homogeneous like ice and include solid carbon (diamond and graphite) and iron (three solid phases) |
|
MIXTURES ELEMENTS AND COMPOUNDS - Junta de Andalucia
Steel is a homogeneous mixture however it is made from iron and carbon A pure substance is different from a homo- geneous mixture because a pure substance |
|
UNIT 3: Pure substances and mixtures 1º ESO - Claseshistoriacom
To know that a substance is identified according to its characteristic properties like: density and melting or boiling points |
|
Cbiesccs02pdf - CBSE Online
6 sept 2019 · Ice iron hydrochloric acid calcium oxide and mercury are the pure substances 8 Identify the solutions among the following mixtures : (a) |
|
Chapter 1 - UNF
Differentiate among pure substances homogeneous mixtures and heterogeneous Pure Substance Matter that has a fixed composition and distinct properties |
|
Iesc102pdf - NCERT
When a scientist says that something is pure it means that all the constituent particles of that substance are the same in their chemical nature A pure |
|
22: Matter - Chemistry LibreTexts
13 août 2020 · Classify pure substances as elements or compounds Examples of elements include carbon oxygen hydrogen and iron |
|
Matter Anything which has mass and occup solid liquid or gas
Pure substances which are made up elements • They cannot be split up into two or m usual chemical methods • For example Iron gold silver carbo |
|
Chemistry: Classifying Matter
If the material is a pure substance further classify it as either an element or compound in the right column iron filings (Fe) Pure Substance Element |
|
Honors Chemistry
The iron has reacted with oxygen and the original iron metal is changed It now exists as iron oxide a different substance Classify the following properties |
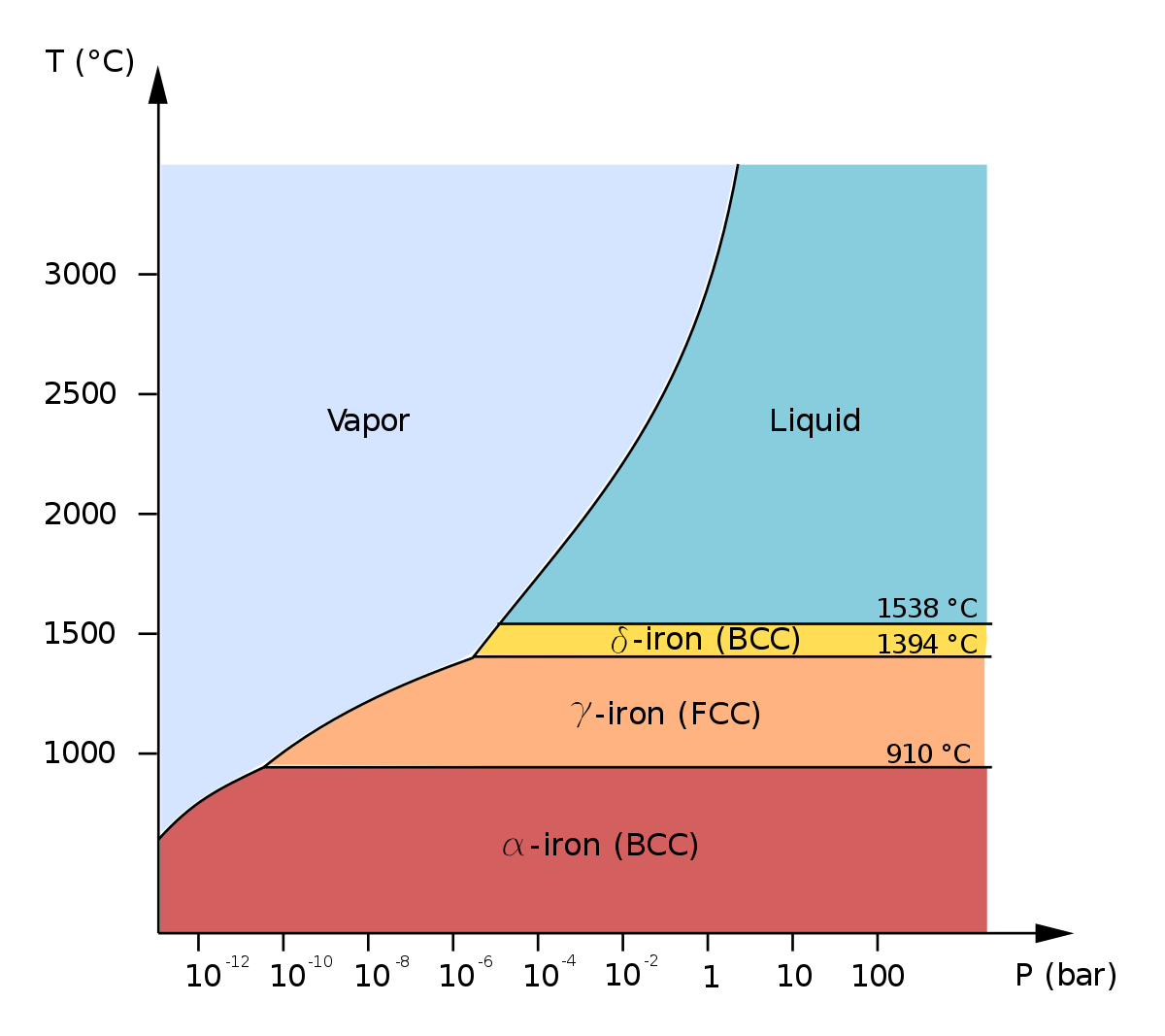
Is an iron a pure substance?
Conclusion: Therefore, iron, hydrochloric acid, and mercury are examples of pure substances.- The iron is an element of atomic number 26. It is represented as Fe. The iron is commonly found to be in solid state. The iron itself is a pure component and not the mixture.
|
Chemistry: Classifying Matter - Paulding
Classify each of the materials below In the center column, state whether the material is a pure substance or a mixture If the material is a pure substance, further |
|
MIXTURES, ELEMENTS AND COMPOUNDS - Junta de Andalucia
Steel is a homogeneous mixture, however it is made from iron and carbon A pure substance is different from a homo- geneous mixture because a pure substance has only one component It has a homogeneous look It has unique properties that characterise it, for example , its density |
|
Pure Element Mixture Suspensions, Heterogeneous Mixture
Pure Substance or Mixture Element or Compound, Homo/Heterogeneous, solution colloid or suspension 1 Iron filings (Fe) Pure Element 2 Orange Juice |
|
Chapter 2 Study Guide Answerspdf
Which of the following is not a pure substance? element compound b molecule d mixture Which of the following is an element? a salt c nylon b ) iron d sand |
|
CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
PURE SUBSTANCES VS MIXTURES Classify the following as pure substances (element or compound) or mixtures 1 sodium Pure substance (E) 11 iron |
|
CLASSIFICATION AND PROPERTIES OF MATTER
A substance is defined as matter which is homogeneous and of which all parts are alike Substances are either elements or compounds Elements are pure |
|
Classification Of Matter - VCC Learning Centre - Vancouver
10) All solutions are mixtures 11) Pure substances are heterogeneous 12) A pure substance is homogeneous but does not have a fixed composition 13) |
|
THERMODYNAMICS PROPERTIES OF PURE SUBSTANCES Pure
A mixture of phases of two or more substance is can still a pure substance if it is homogeneous, like ice and water (solid and liquid) or water and steam (liquid and |
|
Solutions to Supplementary Check for Understanding - CSUN
Alloys are homogeneous mixtures Pure gold is referred to as 24-karat gold c) Propane is a chemical compound so it is a pure substance |