total alkalinity drinking water ppm
|
Commonly Found Substances in Drinking Water
chlorides pH (acidity and alkalinity) |
|
Using this fact sheet Interpreting Drinking Water Quality Results
levels of copper or lead in drinking water are a concern run water for a few minutes before using for Alkalinity and total ... per million (ppm) while. |
|
Interpreting Drinking Water Test Results
pipes or solder and can represent a significant health threat. Tests to Determine Overall Water Quality. ALKALINITY. Measurement needed to determine |
|
IS 10500 (2012): Drinking water
No relaxation. IS 3025 (Part 29). — xxii) Total alkalinity as calcium. 200. 600. IS 3025 (Part 23). — carbonate mg/l |
|
LaMotte Spin Touch Drinking Water.indd
30 avr. 2020 Public Treated Water |
|
2020 Drinking Water Quality Report
Alkalinity: A measure of the water's ability to resist changes in the pH level and a good indicator of overall water quality. Although there is no health risk. |
|
Guidelines for Drinking-water Quality Fourth Edition
reduce the overall risk of disease but may not necessarily render the supply safe. For example chlorine disinfection of drinking-water has limitations |
|
Dutch Drinking Water Statistics 2017
7 août 2017 Gradually more water companies were established and the whole of the Netherlands was connected to the water supply network. The number of. |
|
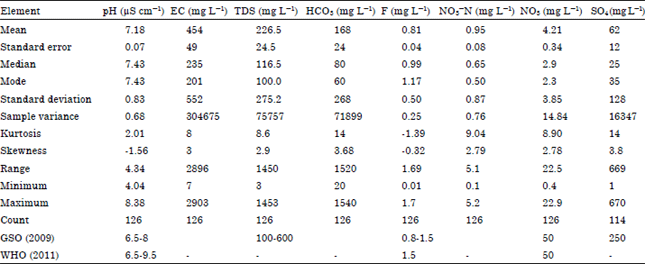
Assessment of Groundwater Quality in Shebna Region Benghazi
2960 ppm total alkalinity 176-400 ppm |
|
Drinking Water Standards
The Detailed Reports above show all regulatory parameters which are monitored in accordance with the. Water Supply (Water Quality)(Amendment) Regulations 2018. |
|
Interpreting Well Water Quality Results
related to alkalinity Alkalinity and total hardness form from the same minerals and are generally close in value when they are both reported as mg/L CaCO 3 Acceptable results: The value should be roughly 75 to 100 of the total hardness value in an unsoftened sample Water with low levels of alkalinity (less than 150 mg/L) is more likely to |
|
Interpreting Drinking Water Quality Results
Alkalinity is a measure of water’s ability to neutralize acids It results primarily from dissolving limestone or dolomite minerals in the aquifer Alkalinity and total hardness are usually nearly equal in concentration (when they are both reported in mg/L CaCO 3 (calcium carbonate) because they form from the same minerals If alkalinity is |
|
Searches related to total alkalinity drinking water ppm PDF
700010 Total alkalinity test kit includes total alkalinity reagent 1 x 475 mL bottle; alkalinity standard/control 1 x 475 mL bottle; and total alkalinity conversion wheel Average of 80 tests per kit 700011 Total alkalinity reagent 4 x 475 mL bottles 700012 Alkalinity standard/control 1 x 475 mL bottle Ordering Information |
What Are The Guidelines For Ph?
Like TDS, pH is given an aesthetic objective in Canada. The Canadian Guidelines for Drinking Water Quality suggest that the pH of drinking water should be between 7.0 and 10.5. The Saskatchewan Drinking Water Standards and Objectives recommend that the pH of drinking water be between 6.5 and 9.0. In the United States, pH is, like TDS, a secondary s...
How Do Water Treatment Facilities Change The Ph of Water?
There are several methods that can increase the pH of water, before disinfection. The pH is commonly increased using sodium carbonate and sodium hydroxide, but a better way of dealing with low pH is to use calcium and magnesium carbonate, which not only will increase pH levels, but will also make the water less corrosive and both calcium and magnes...
What is total alkalinity in water?
What is total alkalinity in water? Total alkalinity is the total concentration of bases and Total Dissolved Solids (TDS) in water expressed as parts per million (ppm) or milligrams per litre (mg/L) of calcium carbonate (CaCO3). High Total Alkalinity decreases the sensitivity of pH. The result is usually high pH and lower chlorine efficiency.
How does alkalinity affect drinking water?
Alkalinity is significant in the treatment of wastewater and drinking water because it will influence treatment processes such as anaerobic digestion. Water may also be unsuitable for use in irrigation if the alkalinity level in the water is higher than the natural level of alkalinity in the soil.
How much alkalinity is in drinking water?
The amount of Alkalinity that should be in our water is 20-200 mg/L for typical drinking water. Alkalinity is basically dissolved minerals in the water that help neutralize the water we drink. Likewise, why do we measure alkalinity in water?
|
Interpreting Drinking Water Test Results - Aquatic Analytics
If your drinking water comes from a private well, you should test your water once million (ppm) alkalinity is much lower than total hardness, test for chloride, |
|
PH Alkalinity Hardness pH Alkalinity Hardness - Wisconsin DNR
Alkalinity is a total measure of the substances in water that have industry), but usually as parts per million (ppm) as calcium drinking water systems |
|
Hardness in Drinking-water - WHO World Health Organization
2 3 Estimated total exposure and relative contribution of drinking-water 2 Depending upon interactions with other factors, such as pH and alkalinity, |
|
Tests for Corrosiveness Alkalinity
What is the standard for alkalinity in drinking water? There is no Total dissolved solids, (TDS), refer to the total amount of inorganic and organic substances, |
|
Total Alkalinity Measurement in Natural Waters - Thermo Fisher
Alkalinity measurement is important in the control of water and wastewater pH as usual, add the total alkalinity reagent and read total alkalinity in ppm as |
|
5 Water quality - Meat & Livestock Australia
quality for cattle drinking water may be too corrosive for boiler feedwater use The total alkalinity of water is always less than its total dissolved solids (TDS) or 10,000 ppm should never be used as water sources for beef cattle The ANZECC |
|
Interpretation of Domestic Water Sample Results - UAEX
HOUSEHOLD WATER: 6 5-8 5 PPM, EC AND TOTAL DISSOLVED SOLIDS, OR DRINKING WATER STANDARD TOTAL ALKALINITY EXPRESSED AS |
|
Whats In My Water? - Texas A&M University
Water with low alkalinity is Acidifying the water will reduce Household method Irrigation: Drinking water standard, 2 0 ppm 2TDS — Total Dissolved Salts |