alkalinity of water definition
What does alkaline mean in water?
Alkaline water has a higher pH level than that of plain tap water.
So proponents say that it can neutralize acid in your bloodstream.
Some say that alkaline water can help prevent disease, such as cancer and heart disease.
However, more research is needed to verify these claims.Alkalinity and pH are closely related and often measured together. pH is a measurement of the concentration of hydrogen ions or the acidity of water.
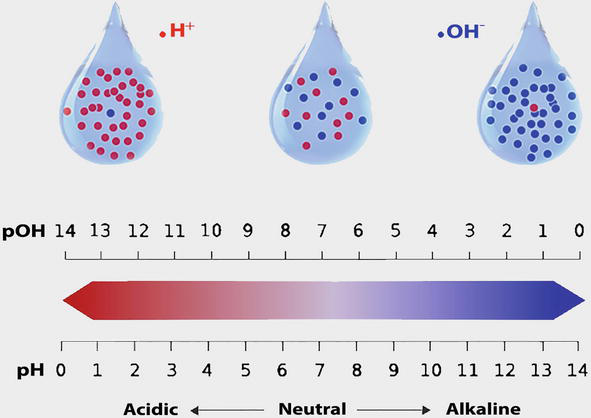
Alkalinity is the ability of the water to neutralize or buffer changes in acidity.
What is the alkalinity of water?
Alkalinity is a measure of water's ability to neutralize acids or resist changes that cause acidity, maintaining a stable pH.
Titration is used to measure the alkalinity of a water sample, and water with a pH of 8 or higher is considered alkaline.
Soils and geology have the biggest influence on water pH and alkalinity.14 sept. 2022
What is the alkalinization of water?
Alkaline water usually has a pH of 8 or 9.
To qualify as alkaline, bottled water must contain alkalinizing minerals like calcium and magnesium carbonate and have negative oxidant reduction potential, meaning it should act as an antioxidant, something like blueberries or fish oil.
|
Alkalinity Alkalinity is a chemical measurement of a waters ability to
Alkalinity is also a measure of a water's buffering capacity or its ability to resist changes in pH upon the addition of acids or bases. |
|
Using this fact sheet Interpreting Drinking Water Quality Results
(Continued on p. 2.) Bacteria - Coliform. 1. Hardness - Total. 2. Alkalinity. 3. Conductivity. |
|
PH Alkalinity of Water
By definition “p” is zero for water which has a pH value of 8.3 or less. The current standard uses HCl 0.1 eq/L as a titrant |
|
The alkalinity of a solution may be defined as the capacity for solutes
226-229). Several different solute species contribute to the alkalinity of water as defined above and titration with acid does not specifically identify them. |
|
Alkalinity
According to Standard Methods for the Examination of Water and Wastewater (SM 2320 Alkalinity) “Alkalinity of a water is its acid-neutralizing capacity. It is |
|
Cross-references ALKALINITY Definition
dissociation of water. Alkalinity then refers to the proton condition with reference to a zero level of protons defined by the species H2CO3 B(OH)3 |
|
Alkalinity and Hardness in Production Ponds
alkalinity when farming aquatic animals. The determination of whether water is acid neutral or base is defined by pH. However |
|
Alkalinity
Alkalinity of water is due primarily to the presence of bicarbonate carbonate |
|
Determination of total alkalinity in sea water
6 août 1997 Definition. The total alkalinity of a sample of sea water is defined as the number of moles of hydrogen ion equivalent to the excess of. |
|
Alkalinity Addition Utilizing Carbon Dioxide & Lime: Inexpensive
result in depositing finished waters. Defining Alkalinity. Alkalinity is defined as the capacity of water to neutralize acid. The most prevalent. |
|
The alkalinity of a solution may be defined as the capacity for solutes
Thus, carbonic acid, HzC03, is converted to HCOa- in the titration to pH 8 3 and is part of the acidity Oxidation of sulfide minerals causes low pH in water draining |
|
PH Alkalinity Hardness pH Alkalinity Hardness - Wisconsin DNR
Two units means 100 times change, • 3 units is 1000 times and so on pH describes a water's relative acidity, provides no measure of how the water will tolerate |
|
PH and Alkalinity - College of the Environment and Life Sciences
water is neutral, whereas the water is acidic if H+ > OH- and basic when OH- > H+ Figure 1: pH scale and examples of solutions at particular pH's Why is pH |
|
PH and Alkalinity - US EPA
Testing water samples for total alkalinity measures the capacity of the water to neutralize acids scale, meaning that for each 1 0 change of pH, acidity or |
|
Total alkalinity - CORE
26 jan 2007 · definition Dickson's expression for TA in natural water samples is given in Section 3 11 The combination of Dickson's expression for alkalinity |
|
Interpreting Drinking Water Test Results - Aquatic Analytics
contaminating the water and means that pathogenic (disease- causing) Alkalinity is a measure of water's ability to neutralize acids, and so is related to pH |
|
PH in Drinking Water - Water Systems Council
pH is measured on a scale that runs from 0 to 14 Seven is neutral, meaning there is a balance between acid and alkalinity A measurement below 7 means acid is |