alkalinity of water sample measures the
What does alkalinity of water indicates?
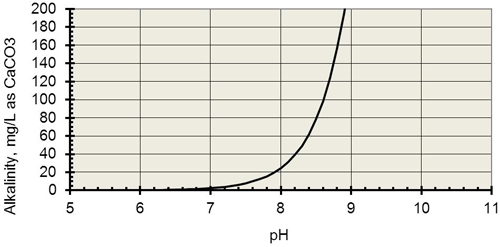
Alkalinity is a measure of water's ability to neutralize acids or resist changes that cause acidity, maintaining a stable pH.
Titration is used to measure the alkalinity of a water sample, and water with a pH of 8 or higher is considered alkaline.
Soils and geology have the biggest influence on water pH and alkalinity.14 sept. 2022Why test drinking water for alkalinity?
Monitoring alkalinity in drinking water is important for both taste and for maintaining effective pH levels required for disinfection.
Moderate alkalinity is desirable in most water supplies.
Strong alkaline water has an unpleasant “soda” taste.What is total alkalinity a measure of?
Total alkalinity is a measurement of the concentration of all alkaline substances dissolved in the water that can both attract and release Hydrogen ions (H+).
This interference with Hydrogen is why alkalinity buffers against change in pH.Water alkalinity and pH are not the same.
Water pH measures the amount of hydrogen (acid ions) in the water, whereas water alkalinity is a measure of the carbonate and bicarbonate levels in water.
Think of carbonates and bicarbonates as dissolved limestone.
|
Chapter 11: pH and Alkalinity Voluntary Estuary Monitoring Manual
Testing water samples for total alkalinity measures the capacity of the water to neutralize acids. This test is important in determining the estuary's |
|
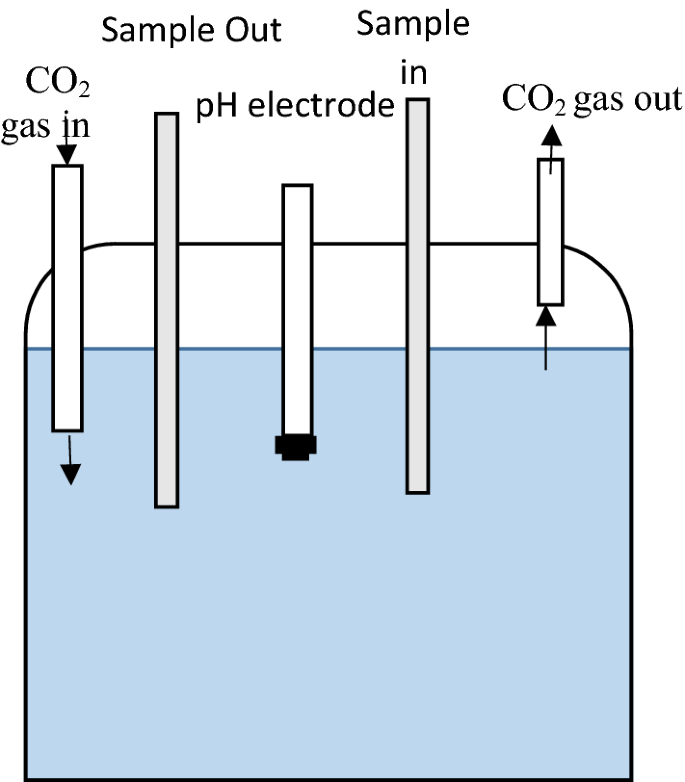
Measuring pH of Pure Water and Other Low Conductivity Waters
Purified water that has very few ionic species is said to be low in alkalinity ionic strength |
|
Using this fact sheet Interpreting Drinking Water Quality Results
There are no health concerns related to alkalinity. The value should be roughly. 75% to 100% of the total hardness value in an unsoftened sample. |
|
Background information on water quality measurements using in situ
Water quality sampling using in situ water quality instruments The pH measures the acidity or alkalinity of water with a pH of 1 being strongly acidic |
|
7.0 EXPERIMENT ON DETERMINATION OF ALKALINITY OF WATER
For the calculation of Phenolphthalein Alkalinity. • The Sulphuric acid is taken in the Burette. • For the first titration the volume of water sample taken is |
|
Determination of total alkalinity of tap water (T alkalinity/M alkalinity)
Outline. Alkalinity is determined by titrating a water sample with a strong acid (such as chlorine and sulfuric acid) and expressed by the calcium carbonate |
|
Alkalinity
Drinking water plants measure alkalinity at different stages of the treatment process: for example on the raw water |
|
Technical Bulletin 517 – Total Alkalinity Measurement in Natural
Alkalinity measurement is important in the control of water and wastewater treatment. In a sample alkalinity is defined as the quantitative ability to |
|
PH and Alkalinity
Figure 1: pH scale and examples of solutions at particular pH's. Why is pH important? Aquatic organisms need the pH of their water body to be within a certain |
|
Measuring pH of Non-Aqueous and Mixed Samples
aqueous (water-based) samples there are many pH applications that call for pH measurements and titrations in non-aqueous samples or in mixed aqueous/non-. |
|
70 EXPERIMENT ON DETERMINATION OF ALKALINITY OF WATER
Alkalinity is primarily a way of measuring the acid neutralizing capacity of water In other words, its ability to maintain a relatively constant pH The possibility to maintain constant pH is due to the hydroxyl, carbonate and bicarbonate ions present in water |
|
Laboratory Experiment 2: Alkalinity and Acidity
Collect 50 mL water sample, add 3 drops of phenolphthalein indicator, titrate the 50 mL sample with 0 02N sulfuric acid to pH 8 3 and estimate phenolphthalein |
|
Total Alkalinity Measurement in Natural Waters - Thermo Fisher
Alkalinity is measured commonly by titration, using either a burette or the drop count technique A sample is titrated with an acid solution, which neutralizes the |
|
PH Alkalinity Hardness pH Alkalinity Hardness - Wisconsin DNR
pH describes a water's relative acidity, provides no Alkalinity is a total measure of the substances in water Calcium and magnesium ions in the sample are |
|
Alkalinity
Alkalinity is a measure of the capacity of water to neutralize acids following end points are recommended for determining total alkalinity in water samples of |
|
Determining Strong and Total Alkalinity in Water - Mediray
Alkalinity is a measure of a water sample's ability to resist a pH change when reacted with an acid; and is an important measure of water quality If a body of |
|
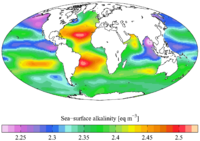
HELCOM Guidelines for sampling and determination of total alkalinity
Although not listed among HELCOM Core Indicators, total alkalinity should be account when determining the state of the marine CO2 system based on AT measurements Water samples are collected from discrete depths, and analysed |