alkene ir
What is the IR absorbance of a terminal alkene?
Alkynes have characteristic IR absorbance peaks in the range of 2100-2250 cm -1 due to stretching of the carbon-carbon triple bond, and terminal alkenes can be identified by their absorbance at about 3300 cm-1, due to stretching of the bond between the sp-hybridized carbon and the terminal hydrogen.
What are the IR spectra of alkenes?
The C-H wags of alkenes fall in a similar range, between 1000 and 600. To summarize then, the IR spectra of alkenes are characterized by one or more C-H stretching peaks between 3100 and 3000, a possible C=C stretch from 1680 to 1630, and one or more C-H wagging peaks from 1000 to 600.
Which IR bands are not specific to an alkene?
Bands for C–H scissoring (1465) and methyl rock (1378) are marked on this spectrum; in routine IR analysis, these bands are not specific to an alkene and are generally not noted because they are present in almost all organic molecules (and they are in the fingerprint region). The bands at 917 cm -1 and 1044cm -1 are attributed to =C&Mac173;H bends.
Which peaks are characteristic of alkenes?
The spectrum for 1-octene shows two peaks that are characteristic of alkenes: the one at 1642 cm -1 is due to stretching of the carbon-carbon double bond, and the one at 3079 cm-1 is due to stretching of the s bond between the alkene carbons and their attached hydrogens.
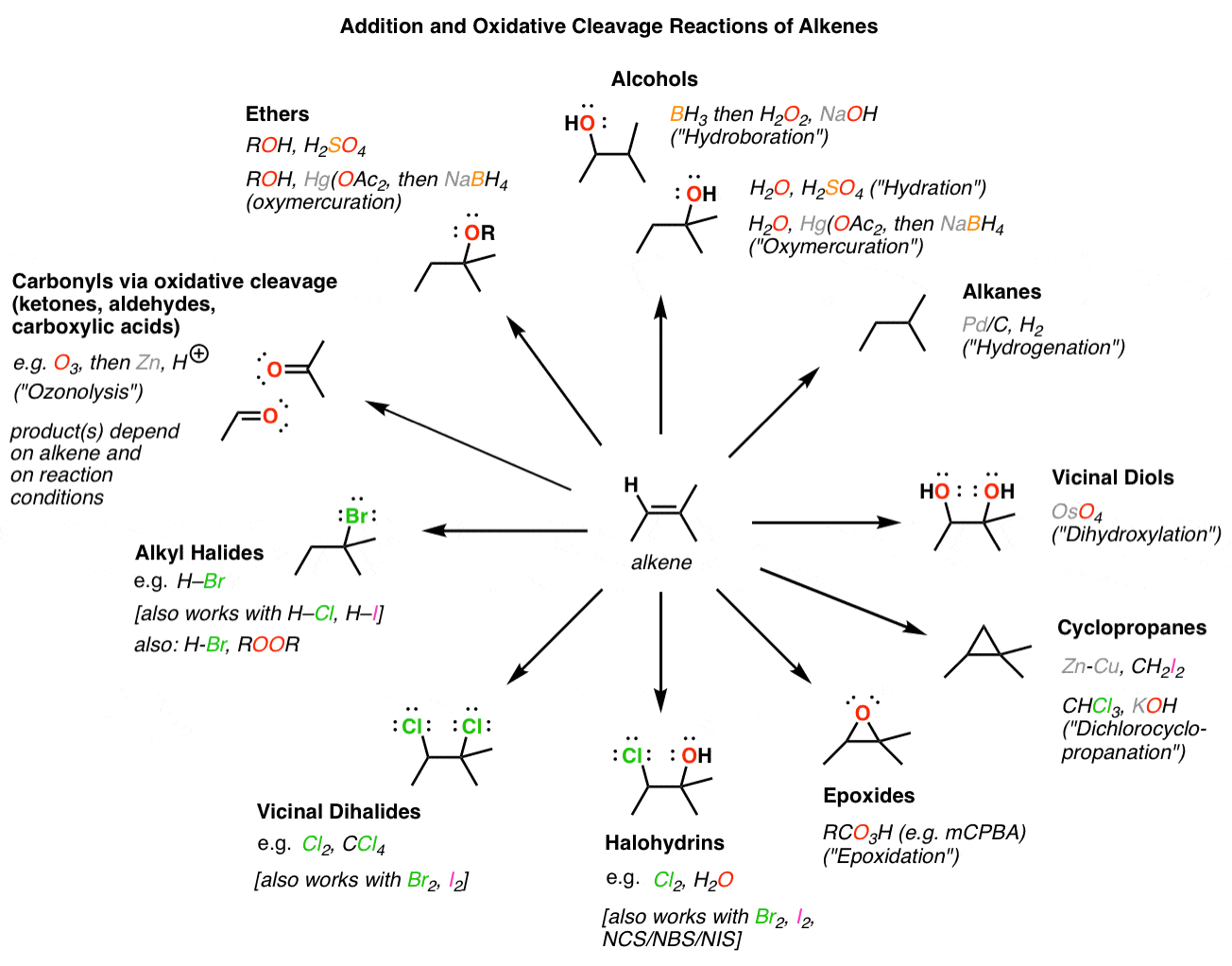
The Chemical Structure of Alkenes
Alkenes contain unsaturated carbons because C=C bonds can have hydrogens added to them via a reaction known as hydrogenationto create saturated carbons (3). Hydrogenated vegetable oil contains unsaturated fats whose C=C double bonds have been hydrogenated to form saturated fats. Structural isomers are molecules that have the same chemical formula b
The IR Spectroscopy of Saturated and Unsaturated Carbons
In a previous column (6), we saw that the saturated carbon containing functional groups methyl (CH3) and methylene (CH2) both have C-H stretches below 3000. Another type of saturated carbon, the methine group, which consists of a carbon atom with three C-C bonds and one C-H bond, also has a C-H stretching peak below 3000 (methine groups will be dis
The IR Spectroscopy of Alkenes
The IR spectrum of a vinyl-containing molecule, 1-octene, is shown in Figure 2. Figure 2:The IR spectrum of 1-octene. Note that in the structure of the molecule shown in Figure 2 there are methyl groups, methylene groups, and a C=C bond. This structure is an example of a mixed molecule and thus it should have C-H stretches above and below 3000. Thi
Distinguishing Alkene Types
We have used Figure 2 so far as a generic example of a double bond spectrum, but it is also the spectrum of a vinyl group. The C=C stretch falls at 1641, and for vinyl groups generally this peak falls from 1660 to 1630. The pair of sharp, intense C-H wags at 993 and 910 are also indicative of vinyl. These peaks generally fall at 990 ± 5 and 910 ± 5
References
B.C. Smith, Spectroscopy31(9), 30–33 (2016).B.C. Smith, Spectroscopy31(3), 34–37 (2016).R. Morrison and R. Boyd, Organic Chemistry, 6th Edition (Prentice Hall, Englewood Cliffs, New Jersey, 1992).B.C. Smith, Spectroscopy31(5), 36–39 (2016).B.C. Smith, Spectroscopy30(1), 16–23 (2015).B.C. Smith, Spectroscopy30(4), 18–23 (2015).B.C. Smith, Spectroscopy31(7), 30–34 (2016). spectroscopyonline.com

IR Spectrum of Alkene: Interpretation

IR spectra practice Spectroscopy Organic chemistry Khan Academy

IR spectra for hydrocarbons Spectroscopy Organic chemistry Khan Academy
|
Infrared Tables (short summary of common absorption frequencies
IR Summary - All numerical values in the tables below are given in wavenumbers cm expansion of alkene & aromatic sp2 C-H bend region (units = cm-1). |
|
12.4 FUNCTIONAL-GROUP INFRARED ABSORPTIONS
absorptions and ACLH bending absorptions. The stretching vibration of the carbon–carbon. Important Infrared Absorptions of Alkenes. Functional group. |
|
Untitled
To see molecules in IR transition must result in a dipole change. IR active -the alkene bond stretching vibrations in alkenes w/o a center of. |
|
Infrared Spectroscopy
Alkanes. 2. Alkenes. 4. Alkynes. 6. Aromatic systems. 9. Alcohols. 12. Ethers. 13. Phenols. 14. Amines. 15. Aromatic amines (anilines). 18. Ketones. |
|
Infrared Spectroscopy
May 15 2013 Most infrared spectra are displayed on a linear frequency ... in routine IR analysis |
|
Predicting cis/trans Structure of Alkene Based on Infrared Spectra
Jul 10 2007 Hence |
|
Ligand Controlled Ir-Catalyzed Regiodivergent Oxyamination of
Jul 16 2019 Regio- and stereoselective alkene oxyamination is attractive ... unactivated alkenes controlled by the Cp ligand on Ir with. |
|
Reactions of an anionic chelate phosphane/borata-alkene ligand
Jun 19 2020 complex underwent a subsequent oxidative addition reaction at the mesityl substituent. Both the resulting Ir(III)hydride complex 15 and the P/ ... |
|
Computational insights into Ir(iii)-catalyzed allylic C–H amination of
May 26 2021 The result suggests that the reaction proceeds through generation of active species |
|
Infrared Tables (short summary of common absorption frequencies
expansion of alkene & aromatic sp2 C-H bend region (units = cm-1) mono mono meta meta expansion of carbonyl (C=O) stretch region (units = cm-1). |
|
Elimination Reactions Alkenes
Chapter 5: Structure and Preparation of Alkenes: Elimination Reactions Alkenes (olefins) are hydrocarbons that contain a carbon-carbon double bond and are |
|
Alkenes & Alkynes - Longdom Publishing SL
27 juil 2020 · Alkenes Alkynes Sudha M Department of Organic Chemistry, Vivekavardhini College, Kothagudem, Telangana, India ABSTRACT Alkenes |
|
Reactions of Alkenes and Alkynes
We discuss this alkene reaction in Chapter 16 5 2 What Is a Reaction Mechanism? A reaction mechanism describes in detail how a chemical reaction occurs It |
|
CHAPTER 3 ALKENES
In contrast, alkane molecules with no double bonds are saturated Ethane ( saturated) Ethene (unsaturated) When we discuss the chemical reactions of alkenes, |
|
Alkene Tutorial
Hydrocarbons (Alkanes, Alkenes, Alkynes and Aromatics) nomenclature to alkene compounds, and alkyl substituents is illustrated in the example below, |
|
Structure and Synthesis of Alkenes
Alkenes (olefins) are hydrocarbons which have carbon–carbon double bonds A double bond is a σ bond and a π bond Double bond B D E = 146 kcal/mol σ |
|
34 Alkenes - chemrevise
Industrially alkenes are converted to alcohols in one step rather than the two in the above sulphuric acid reaction They are reacted with water in the presence of |
|
Module 4 Alkenes and Alkynes Lecture 7 Alkenes - NPTEL
reaction is called the heat of hydrogenation When an alkene is treated with hydrogen in the presence of a platinum catalyst, hydrogen adds to the double bond, |
|
Chapter 8 - Alkenes, Alkynes and Aromatic Compounds
8 1 Alkene and Alkyne Overview 4 8 2 Properties of Alkenes 5 Looking Closer: Environmental Note 6 8 3 Alkynes 10 8 4 Aromatic Compounds: Benzene 12 |








![24 alkene metathesis - [PDF Document] 24 alkene metathesis - [PDF Document]](https://demo.vdocuments.mx/img/378x509/reader018/reader/2020011115/577cd6d11a28ab9e789d5462/r-2.jpg)
![Alkenes PDF - [PDF Document] Alkenes PDF - [PDF Document]](https://demo.pdfslide.net/img/742x1000/reader019/reader/2020031918/5b42e7977f8b9a26268b6ff8/r-2.jpg?t\u003d1612426163)







![Alkane alkene alkyne - [PDF Document] Alkane alkene alkyne - [PDF Document]](https://askb.in/wp-content/uploads/2018/01/Hydrocarbon-Alkene-1.jpg)

![PDF] Alkene (Chemistry) Notes for IIT-JEE Exam Free Download PDF] Alkene (Chemistry) Notes for IIT-JEE Exam Free Download](https://0.academia-photos.com/attachment_thumbnails/36687853/mini_magick20180816-12930-d50m9q.png?1534458760)