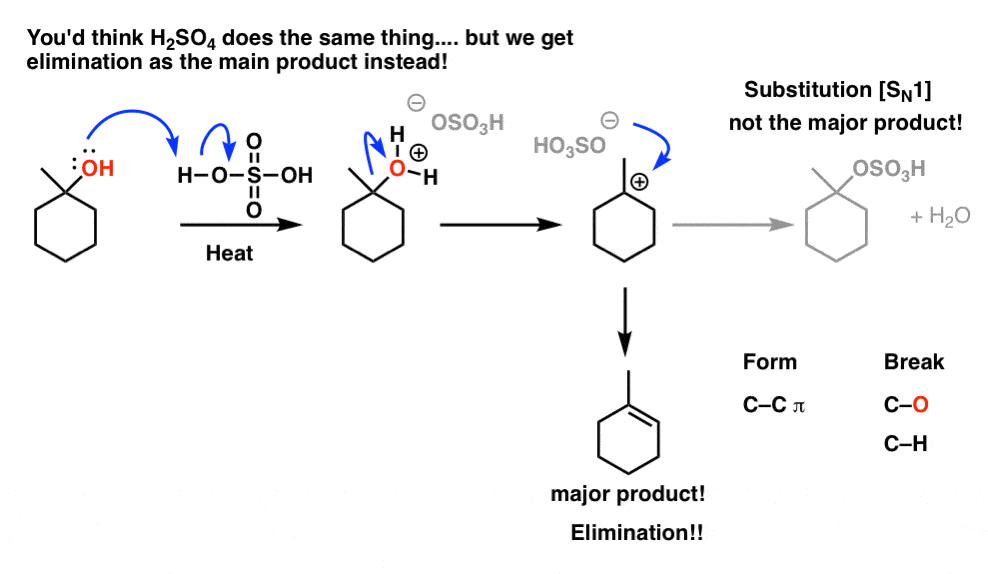
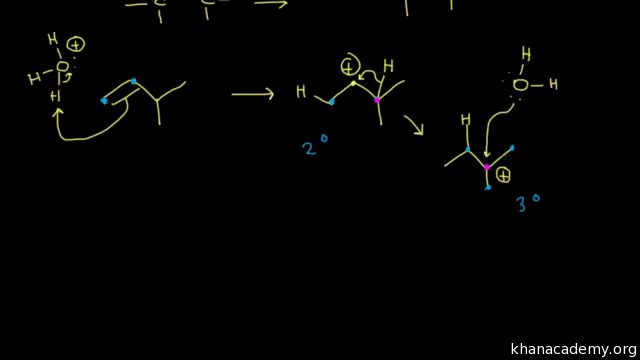
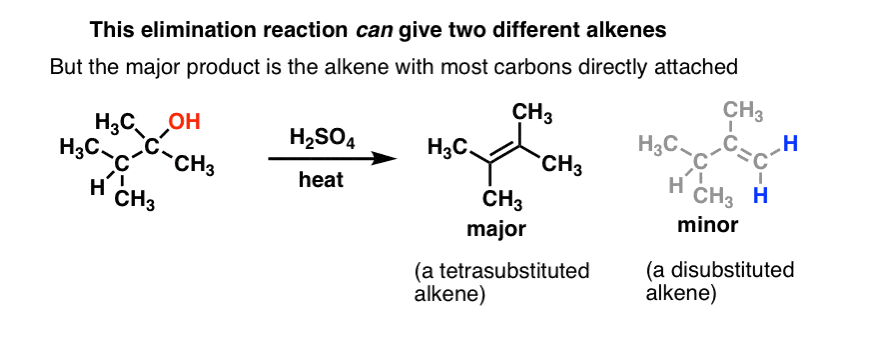
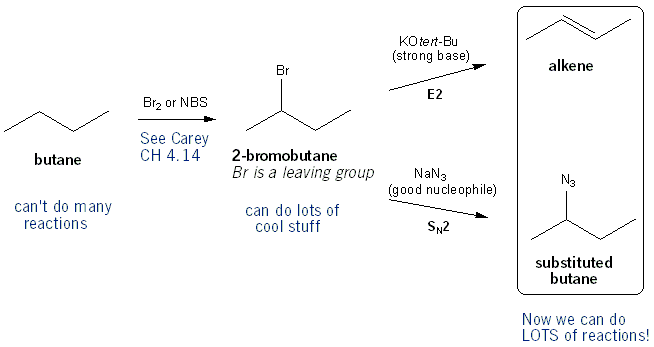
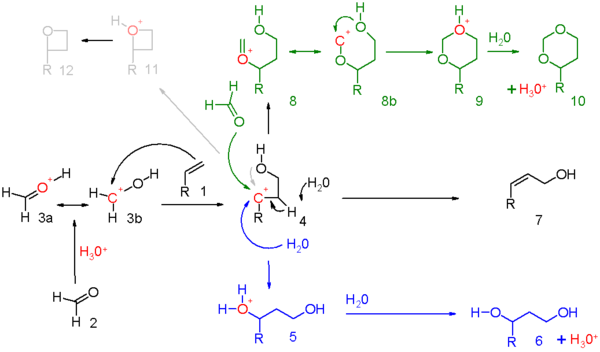
alkene reaction with h2so4 and h2o
Why is H2SO4 added in alkyne hydrolysis reaction?
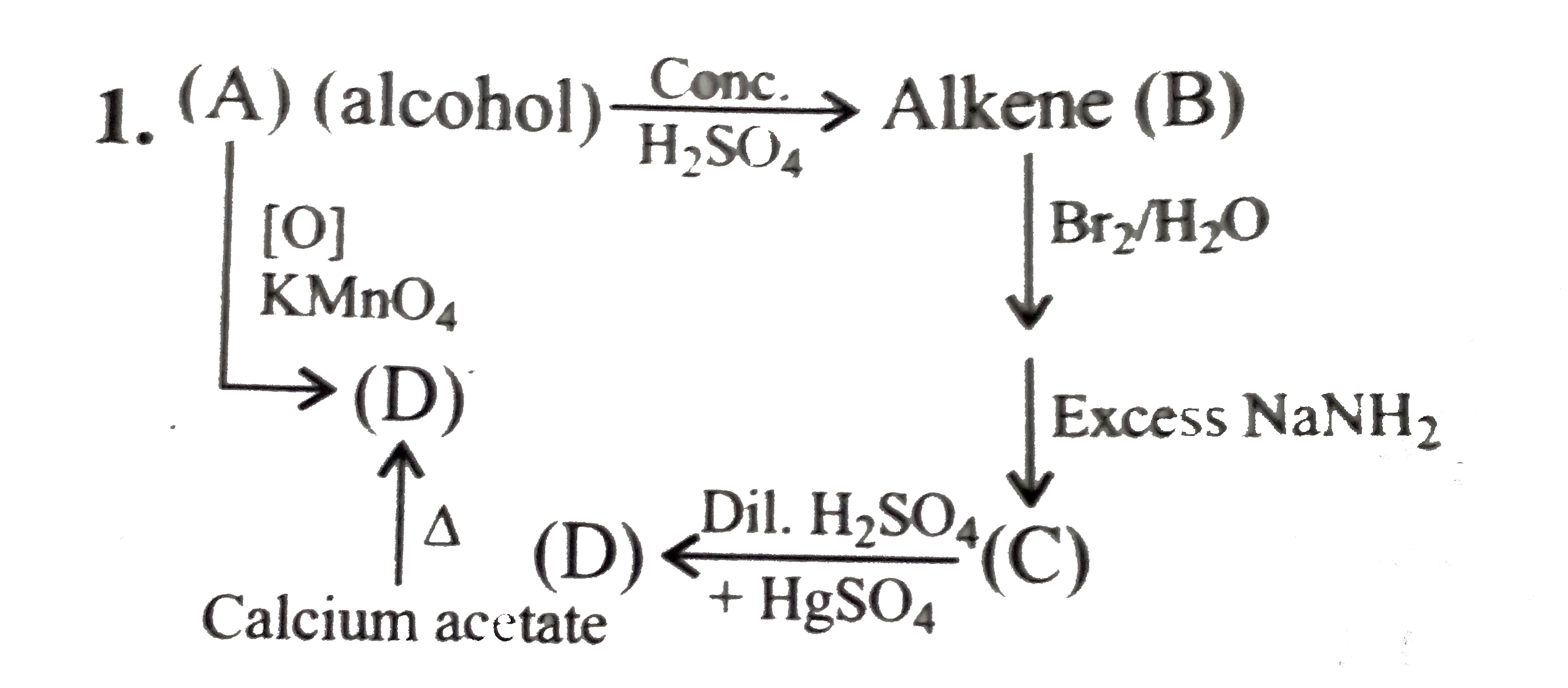
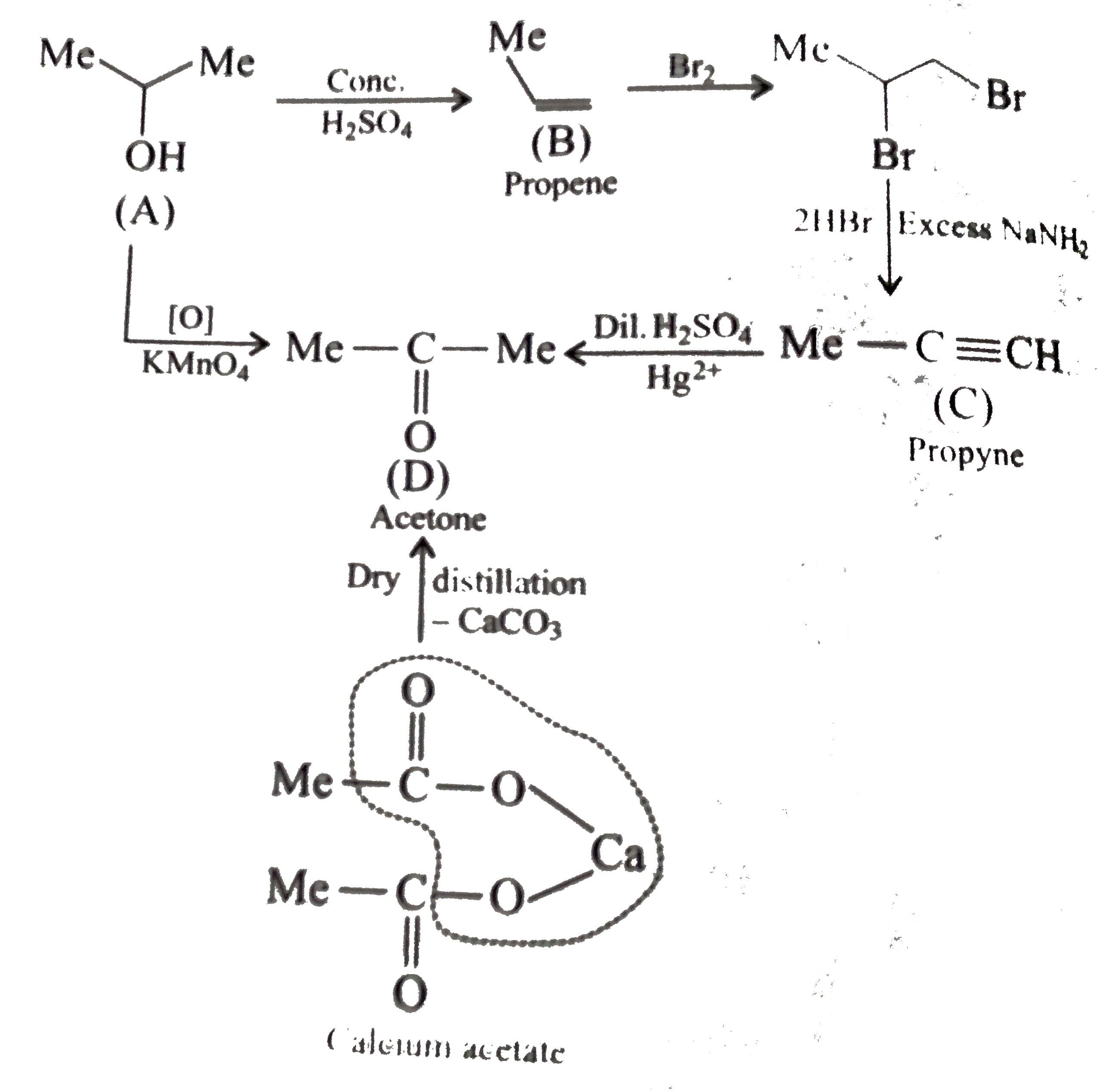
Alkyne hydrolysis is difficult than alkene hydrolysis. Therefore HgSO 4 is added as an extra catalyst dilute with H 2 SO 4 in alkyne hydrolysis. Can I identify alkenes and alkynes from alkene hydrolysis reaction? Yes. You can. Alkenes and alkynes react differently with dilute sulfuric acid. Alkenes are hyrolyzed by dilute sulfuric acid.
How do alkenes and alkynes react with dilute sulfuric acid?
Alkenes and alkynes react differently with dilute sulfuric acid. Alkenes are hyrolyzed by dilute sulfuric acid. But alkynes are not. After addition of sulfuric acid, do some experiments to identify given products (products are given only by alkenes). After hydrolysis of alkene, primary alcohol or secondary alcohol is given.
What is the difference between hydration of alkynes and alkenes?
As with alkenes, hydration (addition of water) of alkynes requires a strong acid, usually sulfuric acid, and is facilitated by the mercuric ion (Hg 2+ ). However, the hydration of alkynes gives ketone products while the hydration of alkenes gives alcohol products. Notice that the addition of oxygen in both reactions follows Markovnikov rule.
What happens when 3-bromo-1-propene reacts with dilute H2SO4?
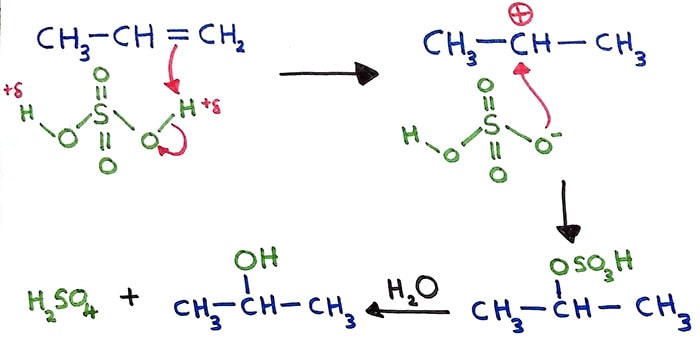
When 3-bromo-1-propene reacts with dilute H 2 SO 4, hydration is occurred and give 3-bromoprop-2-ol. Alkenes can be hydrated and give alcohols as products. Dilute sulfuric acid is used for alkene hydration. A water molecule is added to the carbon atoms which had the double bond.

Alkene + H2SO4 + H2O

Alkene Reactions

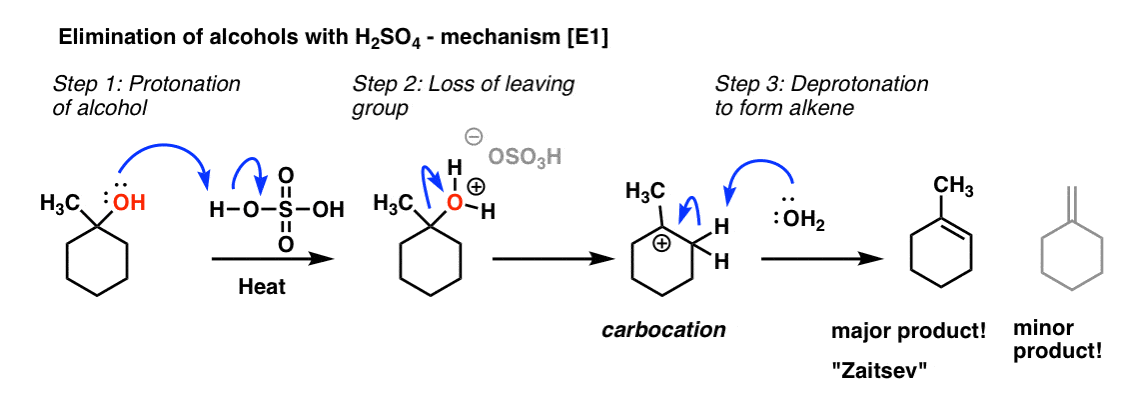
Alcohol Dehydration Reaction Mechanism With H2SO4
|
Chapter 7: Reactions of Alkenes and Alkynes
Reagent: HgSO4/H2SO4/H2O. • You need a Hg catalyst for terminal alkyne hydration. • This reaction adds an OH with Markovnikov regioselectivity to form an enol. |
|
Summary of Alkene Reactions Ch. 8. Memorize Reaction
Alcohols can function in the same way that water does resulting in an ether OR rather than alcohol OH. Page 6. Chem 350 Jasperse Ch. 8 Handouts. 6. 10. |
| PRACTICE EXERCISE – ORGANIC CHEMISTRY I Alkynes |
|
Reactions of Alkenes and Alkynes
H2SO4. Page 22. CHAPTER 5 Reactions of Alkenes and Alkynes. 150. Because hydrogen Yet when treated with H2O H2SO4 |
|
ORGANIC CHEMISTRY I – PRACTICE EXERCISE Alkene reactions
35) Which of the following is the best reaction sequence to accomplish a Markovnikov addition of water to H2SO4. (acid-cat. E1). 1) Hg(OAc)2 / H2O. 2) NaBH4. |
|
Chapter 3 Alcohols Phenols
https://www.angelo.edu/faculty/kboudrea/index_2353/Chapter_03_2SPP.pdf |
|
A hypothesis for growth of fresh atmospheric nuclei
the reaction of alkenes and sulfuric acid–addition of water and addition of J. H. Seinfeld Ternary nucleation of H2SO4 |
|
Pre-Lab: #5 Title: Dehydration of 2-methylcyclohexanol. Introduction
Heading: Alkenes: Preparation Reactions and Properties. Name: Susan W. Mburu H +H2O. (cis / trans) 2-methylcyclohexanol acid. 3-methylcyclohexene water. CH3. |
|
Identifying an Unknown Compound by Solubility Functional Group
Water-insoluble compounds that are insoluble in 5% HCl are tested with concentrated sulfuric acid. (H2SO4). Virtually all organic compounds containing alkene |
|
Reactions of Amines
50. NH2. H3C. 1. NaNO2 HCl. 2. H2O |
|
Reactions of Alkenes and Alkynes
+H2O. H2SO4. 3. CH3. OH. See problems 5.19 5.20 |
|
Summary of Alkene Reactions Ch. 8. Memorize Reaction
Summary of Alkene Reactions Ch. 8. Alkene Synthesis and Reactions. ... -H2O. HSO4. + H2SO4. Protonation. Elimination. Deprotonation. |
|
PRACTICE EXERCISE – ORGANIC CHEMISTRY I Alkynes
19) Provide the structure of the major organic product(s) in the reaction below. C CH. HgSO4. H2SO4 H2O. 20) Provide the structure of the major organic |
|
Practice Set Answer Keys Organic Chemistry I Table of Contents
Test 3 PS3: Test 3 Alkene Reactions Practice H-Cl H2SO4. 102. 2. Hydronium. H3O+ |
|
CHEM 109A CLAS Alkenes and Reactions of Alkenes - KEY 1
trans-3-methyl-pent-2-ene. H. I d. 1-methylcyclopentene. H. Cl. 1-methylcyclopentene. H. Cl. H. Cl-. H. Cl e. 2-butene. H2O. H2SO4. Page 1 of 6 |
|
Chapter 7: Reactions of Alkenes and Alkynes
Reagent: HgSO4/H2SO4/H2O. • You need a Hg catalyst for terminal alkyne hydration. • This reaction adds an OH with Markovnikov regioselectivity to form an enol. |
|
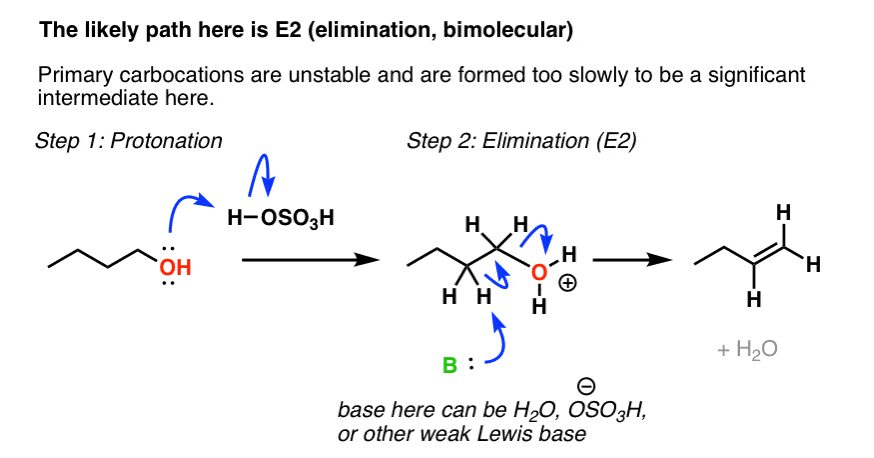
ORGANIC CHEMISTRY I – PRACTICE EXERCISE Elimination
ORGANIC CHEMISTRY I – PRACTICE EXERCISE. Elimination Reactions and Alkene Synthesis or H2O. 4). + H3O. + or H2SO4. 26). 27). 28) Triethylamine. |
|
Chapter 6: Reactions of Alkenes: Addition Reactions 6.1
6.1: Hydrogenation of Alkenes – addition of H-H (H2) to the ?-bond of alkenes to afford + H2O. H2SO4. How is the position of the equilibrium controlled? |
|
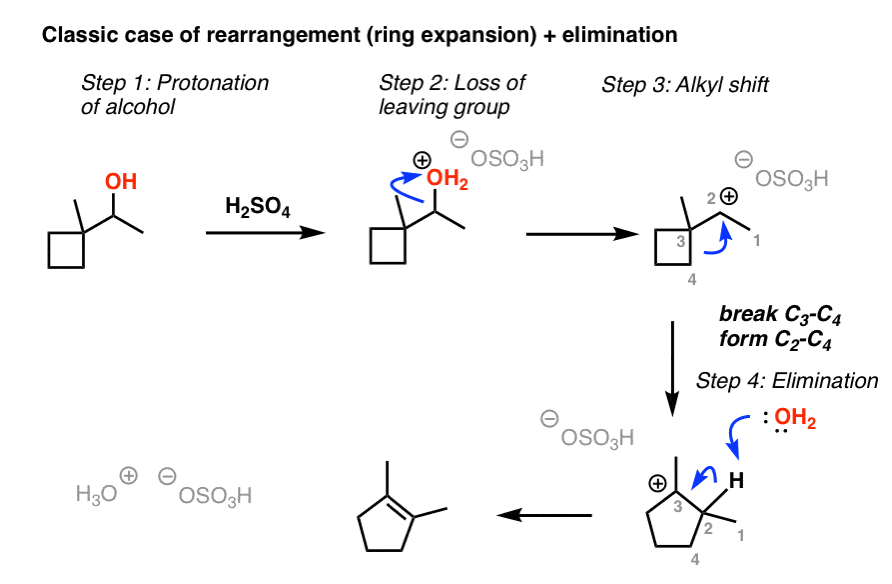
Acid catalyzed reactions you should be able to write arrow-pushing
(-H2O). OH. OH. OTs. H. (-H2O). O. O. O. OH. O. OTs. H. O. O. H2SO4 / H2O (A Lewis or Bronsted acid = E+ = strong the acidity drives the reaction). |
|
Chapter 7: Reactions of Alkenes and Alkynes
Reagent: HgSO4/H2SO4/H2O • You need a Hg catalyst for terminal alkyne hydration This reaction adds an OH with Markovnikov regioselectivity to form an enol The enol product then tautomerizes to form a ketone o (Note: you may also see BH3•THF, B2H6, or even (Sia)2BH drawn out |
|
34 Alkenes - chemrevise
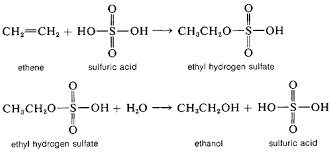
Electrophilic Addition Reactions of Alkenes Definition alkenes undergo addition reactions Addition CH3CH2OSO2OH + H2O → CH3CH2OH + H2SO4 |
|
Summary of Alkene Reactions, Ch 8 Memorize Reaction
+ OH2 -H2O HSO4 + H2SO4 Protonation Elimination Deprotonation OH H2O, H+ OH Yes Problems with Acid-Catalyzed Addition of Water to Alkenes 1 |
|
CHEM 109A CLAS Alkenes and Reactions of Alkenes - UCSB CLAS
d 2-pentene 2-pentanol OH H2O H2SO4 also |
|
Chapter 6: Reactions of Alkenes: Addition Reactions 61
6 1: Hydrogenation of Alkenes – addition of H-H (H2) to the π-bond of alkenes to H3C CH2 + H2O H2SO4 How is the position of the equilibrium controlled? |
|
Reactions of Alkenes and Alkynes
alkenes form polymers by the addition of monomers (Greek: mono, one, and for the product of each alkene hydration reaction: (a) (b) +H2O H2SO4 H2SO4 |
|
Reactions of Alkenes
The alkene reacts with conc sulfuric acid to give an alkyl hydrogen sulfate, The alkyl hydrogen sulfate can be converted to an alcohol by boiling in water |
|
Reactions of Alkenes
The alkene reacts with conc sulfuric acid to give an alkyl hydrogen sulfate, The alkyl hydrogen sulfate can be converted to an alcohol by boiling in water |
|
7-Reactions of Alkenes and Alkynes
Br2/H2O Fall, 2007 9 Hydration of Alkenes ▫ Alkenes react with water to when H3O+ is a reagent, this means aqueous acid (e g HCl or H2SO4 in water) |
|
ALKENES - Knockhardy
the main reaction of alkenes is ELECTROPHILIC ADDITION be converted to ethanol by boiling with water C2H5OSO2OH(aq) + H2O(l) ——> H2SO4(aq) |