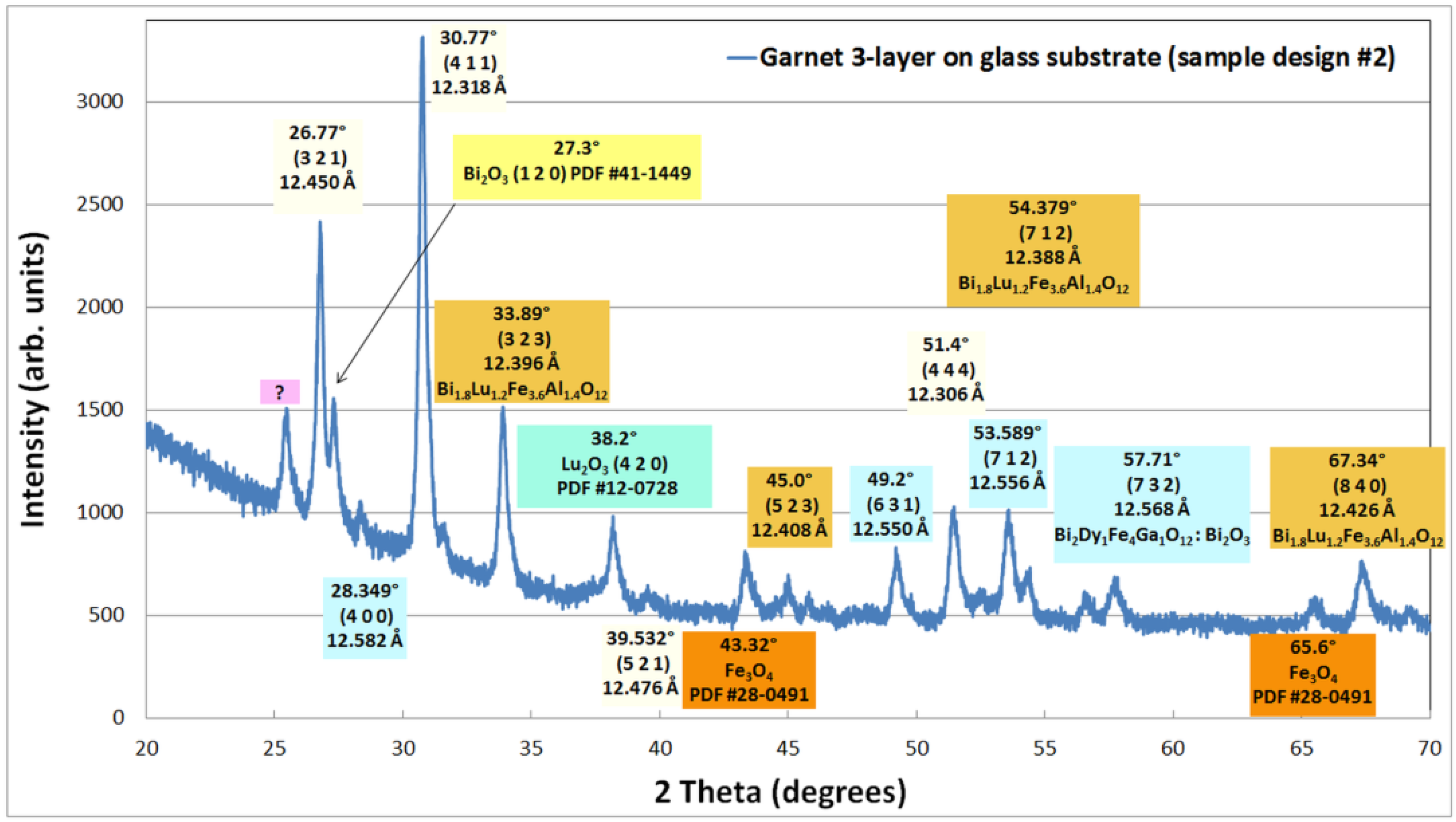
500nm in cm 1
To convert from wavenumber (cm-1) to nanometers, use the following equation: 1/cm-1 x 107 = nanometers.
Or to convert from nanometers to wavenumber, you simply reverse the equation so that wavenumber (cm-1) = 107 / wavelength in nanometers.
Or use the tool below.29 jan. 2024
How do you find wavelength in cm 1?
IF you are talking about ABSOLUTE wavelength (i.e. the wavelength of CO2 laser is 10.6 microns) then the conversion goes as follows:
1Wavelength in µm = 10,000/cm-12Wavenumbers in cm-1= 10,000/µm.3) Wavenumbers in cm-1= 10,000,000/nm.4d( Wavelength in µm) = (10,000 * d(cm-1)/(cm-1)2)
What is cm inverse?
Reciprocal length or inverse length is a quantity or measurement used in several branches of science and mathematics, defined as the reciprocal of length.
Common units used for this measurement include the reciprocal metre or inverse metre (symbol: m−1), the reciprocal centimetre or inverse centimetre (symbol: cm−1).
|
Seasonal Variations and Long-term Trend of Mineral Dust Aerosols
23 nov. 2020 Table 1. Dates CALIPSO dust ratios (%) |
|
Seasonal Variations and Long?Term Trend of Dust Particle Number
5 déc. 2019 (with NDd > 500nm > ~1 cm. ?3. ) to the CALIPSO classified aerosol data. Thirteen of these top 1% dust events occurred within the period ... |
|
Tissue Optics
2 févr. 2020 4µs = scattering coefficient [cm?1] g = anisotropy of scatter. ... where µs(500nm) = 44 cm?1 |
|
Lida Latifzadeh Masoudipour Ph.D. © 2009 Infrared (IR) Spectroscopy
absorption of IR radiation by a molecule which causes stretching bending and motion of chemical bond. The absorbed wavenumber v. -. = 1 / in cm giving |
|
Advantages of a Fourier Transform Infrared Spectrometer
of infrared uses wavenumbers (cm1) which are linear in energy (cm1 = 1/(nm * 107) |
|
ADAPTIVE OPTICS FACILITY AND MUSE ON THE VLT-UT4
AND MUSE ON THE VLT-UT4. 1. GTC lecture. Fuyan Bian ???. ESO Chile seeing ~ ?/r0 For a good site |
|
Course number: BIP-31306 Principles and Applications of
b) Emission at 15342 cm-1 c) A NMR transition at 300 MHz d) An ESR transition at 3 cm. Question 1.2. Suppose we have blue green light (500 nm) with an |
|
Atomic and Molecular Physics NET-JRF June 2011-Dec 2018
A laser operating at 500 nm is used to excite a molecule. If the Stokes line is observed at. 770 cm-1 the approximate positions of the Stokes and the |
|
1. A laser light beam (the wavelength is 500 nm) hits a screen with
An electron microscope uses 2 keV electrons for imaging. The diameter of the electrostatic lens is 5 cm with a focal length of 50 cm. What is the diffraction |
|
Fundamentals of Raman spectroscopy Vibrational Spectroscopy
13 nov. 2012 Example: A Raman band at 1020 cm-1 and excited with a laser wavelength of 500 nm scatters light at a wavelength of 527 nm. A vibration that ... |
|
Solutions (PDF)
(e) visible spectrum: violet (500 nm) red (800 nm) 408 nm = UV Problem #3 number should be in cm–1, we first change the wavelength into cm: = 5 89 x 10 |
|
Stichpunkte zur Lösung zum Übungsblatt 2 zu 1) λ = 500 nm
zu 1) λ = 500 nm; Umrechnung: λ = 107 / Wellenzahl a) Wellenzahl = 20000 cm-1 (ΔO) b) 1 kJ = 83 5 cm-1, hier: ΔO = 239 5 kJ zu 2) M + L = ML β = [ML] / [M] [L] |
|
Liquid Phase Exfoliation and Dispersion of Two Dimensional Materials
1 10-4 10-2 100 102 104 σ DC ~ M f σ DC (s/m) Graphene Mass Fraction, M f f 500 nm h 800 1200 360 380 400 420 gth (nm) Wavenumber (cm-1) |
|
The Scale of Things – Nanometers and More
1 nanometer (nm) 0 01 μm 10 nm 0 1 μm 100 nm 1 micrometer (μm) 0 01 mm 10 μm 0 1 mm 100 μm 1 millimeter (mm) 1 cm 10 mm 10-2 m 10-3 m |
|
1 Answers to spectroscopy questions 1 Consider the spectrum
A = 0 095 at 350, so a = 0 24 mM-1·cm-1 or 240 M-1·cm-1 a concentration of 74 2 ppm and an absorbance of 0 972 at 500 nm and an absorbance of 0 276 at |
|
1 A laser light beam (the wavelength is 500 nm) hits a screen with
An electron microscope uses 2 keV electrons for imaging The diameter of the electrostatic lens is 5 cm with a focal length of 50 cm What is the diffraction limited |
|
Electronic Supplementary Information - The Royal Society of
absorbance peaks at 1084 and 924 cm-1 due to stretching of the C–O bonds of 500nm Supplementary Material (ESI) for Journal of Materials Chemistry |
|
Infrared (IR) Spectroscopy - IFSC/USP
cm -1 to identify a particular chemical bond and study the molecular structure For example, = 2500 nm, 1 nm = 10 -9 m , 1 cm = 10 -2 m , in cm : 2500 nm x (10 |
|
HW_27pdf
1 PSE6 35 P 012 [317889] The wavelength of red helium-neon laser light in air is n=1 angle of refraction at first surface 1 2 50cm a s angle of incidence at x = 500nm = 500x109 m = 5 x 10-7 m G=1 (c) Does P correspond to a maximum, |
|
WVC (cm) AOD 500 nm 1−SSA 500 nm WVC (cm) SSA 500 nm
WVC (cm) AOD 500 nm 1 8 2 2 2 2 4 2 6 2 8 3 3 2 3 4 3 6 0 04 0 06 0 08 1−SSA 500 nm 0 0 5 1 1 5 2 2 5 3 3 5 4 0 9 0 92 0 94 0 96 WVC (cm) |















![PDF] Nano‐Domain Pinning in Ferroelastic‐Ferroelectrics by PDF] Nano‐Domain Pinning in Ferroelastic‐Ferroelectrics by](https://0.academia-photos.com/attachment_thumbnails/38340340/mini_magick20190225-7539-hk20av.png?1551131693)