ethanoic acid and methanol reaction
What is the difference between methanol and ethanol?
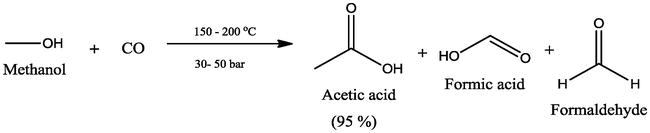
Just like ethanol, the first step changes the alcohol to the aldehyde, and the second step changes the aldehyde to the carboxylic acid. From methanol though, formaldehyde and formic acid are produced instead of the harmless acetic acid (as in the case of ethanol).
Can methanol react with 1-methoxyethanol?
Thus methanol can react with 1-methoxyethanol to form the acetal, 1,1-dimethoxyethane, and water: The reactions of alcohols with aldehydes and ketones are related to the reactions of alcohols with acids (esterification) discussed in the preceding section.
Can methyloxonium ion reduce methanol reactivity?
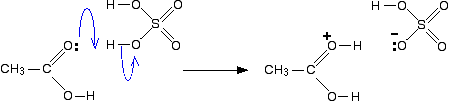
If too much acid is present, then too much of the alcohol is converted to the oxonium salt: Clearly, formation of the methyloxonium ion can operate only to reduce the nucleophilic reactivity of methanol toward the carbonyl carbon of the carboxylic acid. Another practical limitation of esterification reactions is steric hindrance.
What is the reaction of ethanoic acid with alcohol?
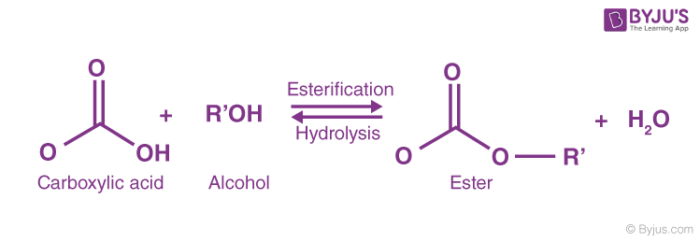
The Reaction of Ethanoic Acid with Alcohols to make Esters. concentrated sulfuric acid, to form esters. Concentrated sulfuric acid is a catalyst for this reaction. methanol + ethanoic acid methyl ethanoate + water. The s tructure of methyl ethanoate. ethanol + ethanoic acid ethyl ethanoate + water. The s tructure of ethyl ethanoate.

Methanol to Ethanoic acid Conversion of Organic Chemistry

Properties of ethanoic acid Carbon and its compounds Class 10 Chemistry Khan Academy

Methanol Explained Alcohol Science
|
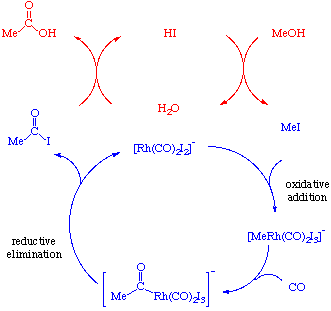
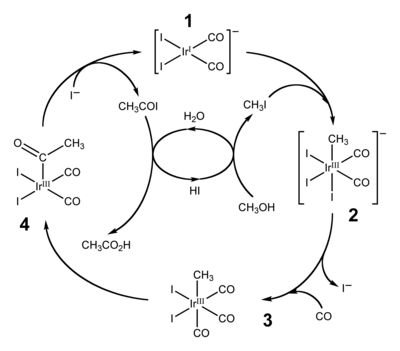
Recent advances in the methanol carbonylation reaction into acetic
21 déc. 2021 Today acetic acid (ethanoic acid) is at 85 % produced by methanol carbonylation. Its production is significantly growing since 2015 |
|
Chapter 5 Carboxylic Acids and Esters
Learn the major chemical reaction of carboxylic acids and esters and learn how to Ethanoic acid/acetic acid is the main ingredient in vinegar. |
|
Ester Synthesis Lab (Student Handout)
Esters are produced by the reaction between alcohols and carboxylic acids. alcohol and carboxylic acid you will use in the synthesis reaction. |
|
AS Chemistry Paper 2 2018 Model Answer - Science Skool!
25 mai 2018 The student dissolved 784 mg of impure citric acid in water to prepare 250 ... When ethanoic acid reacts with sodium hydroxide the enthalpy ... |
|
???? ?????? ??????? ????? ???????? ??????? ???????? ??
1.1- Write the equation of this reaction. 1.2- Why is the mixture heated? 1.3- If a mixture of ethanoic acid and methanol is heated in the absence of |
|
Untitled
10 juin 2016 Ethene reacts with steam in the presence of an acid catalyst to form ... Methanol was placed in a spirit burner and the mass of the spirit ... |
|
H432/01 Periodic table elements and physical chemistry - OCR
1 juin 2020 A chemist proposed producing methanol at 525 K using equilibrium 1. ... 20 A student investigates the reaction between ethanoic acid CH. |
|
Untitled
13 juin 2017 Calculate the concentration of the ethanoic acid in the buffer ... The equation shows the final stage in the production of methanol. |
|
FET PHYSICAL SCIENCES TERM 1 & 2 2019 TRAINING HANDOUT
reaction because the two molecules of alcohol and carboxylic acid link up by Odour of ester ethanol ethanoic acid. (1) methanol salicylic acid. (1) ... |
|
4970364461
(d) Explain why chlorine does not react with aqueous sodium fluoride. (d) Ethanoic acid reacts with methanol to form an ester. |
|
Worksheet 1
Esters are produced by the reaction between alcohols and carboxylic acids alcohol and carboxylic acid you will use in the synthesis reaction Provide space |
|
THE CHEMISTRY OF SMELL
17 avr 2015 · Acetic acid and methanol react in a condensation reaction producing an ester and water |
|
Esters
What alcohol and carboxylic acid could be used to form the following esters? Write equations for the following reactions using structural formula Propan-1-ol |
|
Lesson 10 Chemical reactions with alcohols carboxylic acids and
Esters An ester is synthesised from a carboxylic acid and an alcohol using an acid catalyst (H2SO4) The reaction that forms an ester is referred to as an |
|
By- step reaction pathway for formation of an ester from ac
4 8 Reaction Pathway for Esterification A somewhat simplified step- ester from a carboxylic acid and an alcohol is shown below Kirt Michael, Organic Chem |
|
Synthesis of acetic acid via methanol hydrocarboxylation with CO 2
11 mai 2016 · such as alcohols, urea, carbonates, polymers and carboxylic acids Figure 1 Synthesis of acetic acid by reaction of methanol with CO2 and |
|
Elements, Mixtures and Compounds - St Andrews & St Brides High
Have a go at drawing the full structural formula of this ester Making Esters Esters are made by a condensation reaction between carboxylic acid and alcohol in |
|
FET PHYSICAL SCIENCES TERM 1 & 2 2019 TRAINING HANDOUT
The reaction between an alcohol and a carboxylic acid to form an ester is known as esterification It is an example of a condensation reaction because the two |
|
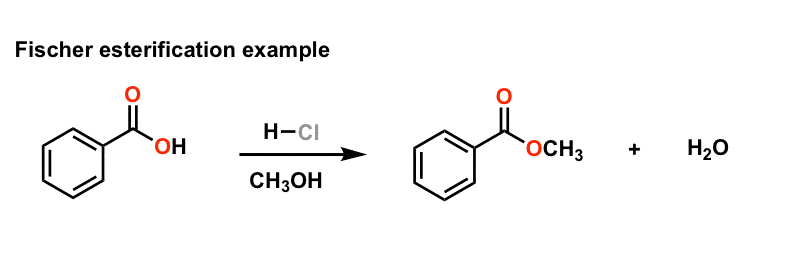
Fischer Esterification
carboxylic acid is treated with an alcohol in the presence of a mineral inorganic ethanol used in the reaction mix, producing HCl, which then will initiate the |

















:max_bytes(150000):strip_icc()/45855026674_f85153017e_b-5c49cb0ac9e77c0001da1c9f.jpg)
