electrochemistry cell potential problems
What are the practice problems in electrochemistry?
The following practice problems are to assist in your mastery of the topic of Electrochemistry. From the following observations, estimate the E ∘ from the half reaction M + (aq) + e − → M(s): The metal M reacts with H 2SO 4(aq); but not with HI(aq); M displaces Au + (aq), but not Fe3 + (aq).
What is a standard electrode potential?
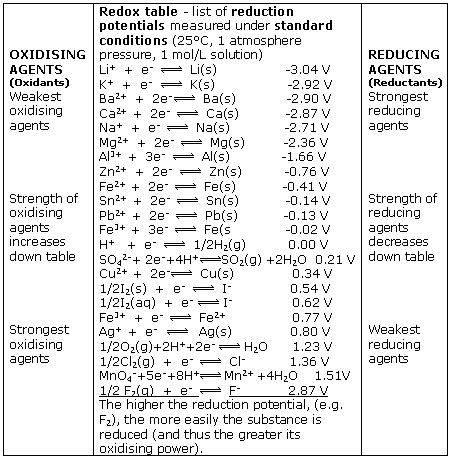
When the half-cell X is under standard-state conditions, its potential is the standard electrode potential, E°X. Since the definition of cell potential requires the half-cells function as cathodes, these potentials are sometimes called standard reduction potentials.
How do you calculate electrode potential for a half-cell x?
The electrode potential (EX) for a half-cell X is defined as the potential measured for a cell comprised of X acting as cathode and the SHE acting as anode: Ecell = EX −ESHE ESHE = 0V(defined) Ecell = EX E cell = E X − E SHE E SHE = 0 V (defined) E cell = E X
How can a cell potential (voltage) be predicted?
The cell potential (voltage) for an electrochemical cell can be predicted from half-reactions and its operating conditions ( chemical nature of materials, temperature, gas partial pressures, and concentrations).
|
Test4 ch19 Electrochemistry Practice Problems.pdf
p2 K Values and Voltage p11. Spontaneous Voltaic Electrochemical Cells p4 Nonstandard Concentrations and Cell Potential p11. Cell Potentials p5 Electrolysis. |
|
Untitled
Ch.17- Electrochemistry: Practice Problems I 2) What voltage will be produced by this electrochemical cell? a) +2.97V. Reduction Potential Eº. |
|
Electrochemical Cells Worksheet
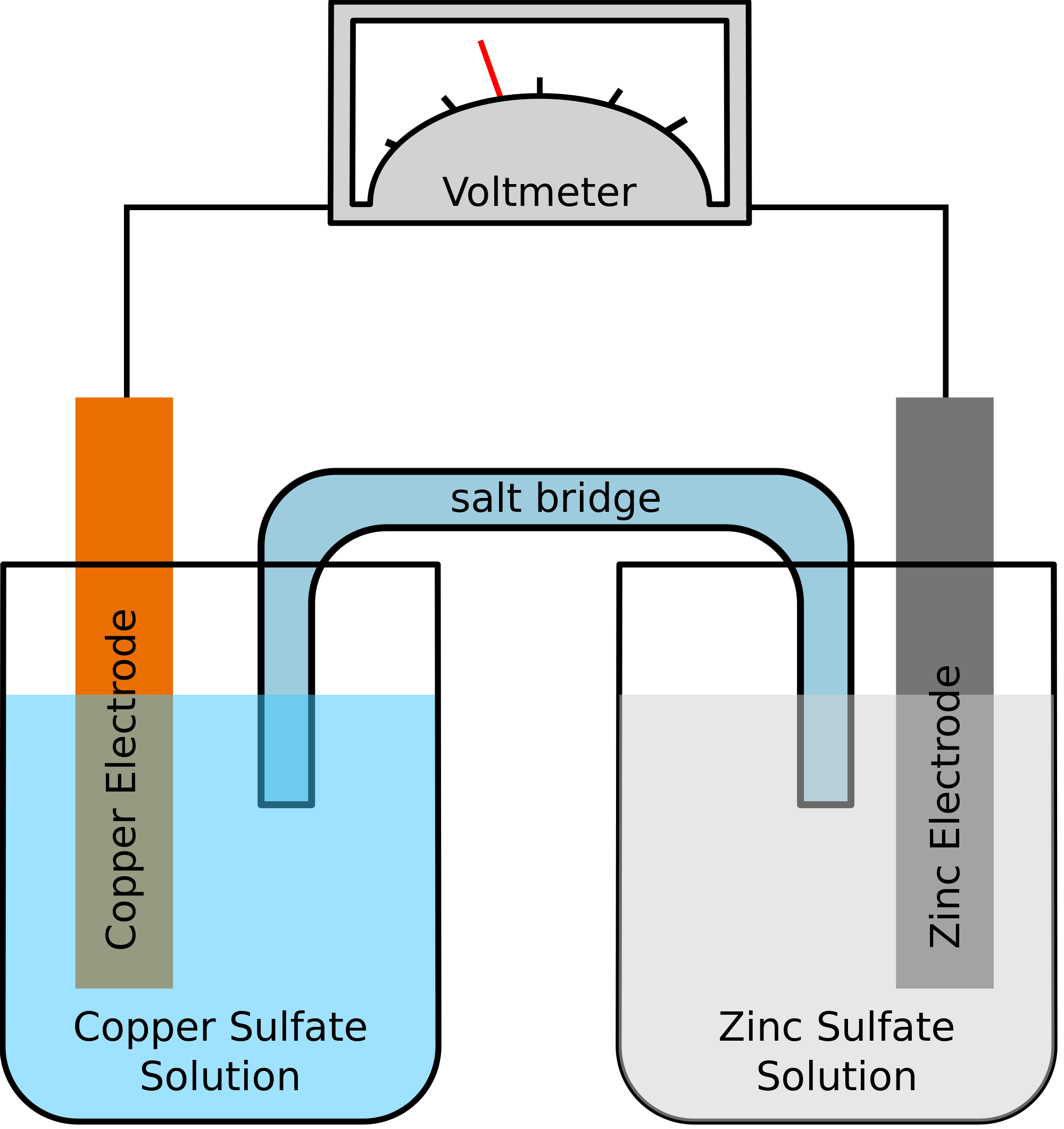
Which is anode and which is the cathode? 3. Calculate the standard cell potential produced by a galvanic cell consisting of a sodium electrode in contact with a |
|
Ch 11 Practice Problems
Consider the galvanic cell shown below (the contents of each half-cell are written beneath each compartment). The standard reduction potentials are as |
|
Test4 ch19 Electrochemistry Practice-answers-Marked
Extra Practice Problems Spontaneous Voltaic Electrochemical Cells p4 Nonstandard Concentrations and Cell Potential p11. Cell Potentials p5 Electrolysis. |
|
Bookmark File PDF Ap Chemistry Electrochemistry Answers
8 days ago Electrochemistry Practice Problems -. Basic Introduction AP Chemistry. Electrochemistry: Cell Potentials. Introduction to Galvanic Cells ... |
|
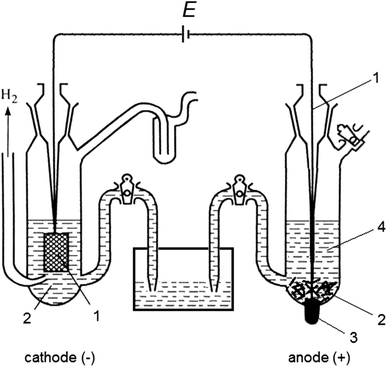
Electrochemical cells
Surface area of the metal will change the electrode potential. This article Redox: reducing difficulties |
|
A.P. Chemistry Practice Test - Ch. 17: Electochemistry MULTIPLE
20 Feb 2018 13) The purpose of the salt bridge in an electrochemical cell is to ______ ... 19) The standard cell potential (Eecell) for the voltaic cell ... |
|
Download File PDF Ap Chemistry Electrochemistry Answers
1 Sept 2022 Problems - Basic. Introduction AP Chemistry. Electrochemistry: Cell. Potentials Introduction to. Galvanic Cells &. Voltaic Cells AP ... |
|
A Practical Beginners Guide to Cyclic Voltammetry
3 Nov 2017 The Nernst equation relates the potential of an electrochemical. Figure 1. (A) Homogeneous and (B) heterogeneous reduction of Fc+ to Fc. The ... |
|
Test4 ch19 Electrochemistry Practice Problems
p2 K Values and Voltage p11 Spontaneous Voltaic Electrochemical Cells p4 Nonstandard Concentrations and Cell Potential p11 Cell Potentials p5 Electrolysis |
|
Ch 17 Electrochemistry Practice Problems I - keypdf
2) What voltage will be produced by this electrochemical cell? a) +2 97V Reduction Potential, Eº (6) +1 55V Sete Cathde Aarole pb2+ + 2€“ -> P6 -0 13 V |
|
Ch 11 Practice Problems - UCSB CLAS
Consider the galvanic cell shown below (the contents of each half-cell are written beneath each compartment) The standard reduction potentials are as follows: |
|
Solutions for Electrochemistry Problem Set
24 avr 2001 · Notice that in this reaction the cell potential is positive, this electrochemical cell is spontaneous (the reactions are going the way they want to) |
|
2 Electrochemistry
One of common problems, that will become be the solution to the problem: The standard cell potential E0 of a galvanic cell is the cell potential measured |
|
Electrochemistry Problems
is the cathode and its reduction potential is 0 951 V (a) Write the half-reaction that occurs at the anode (b) If E° is 0 609 V, what is the potential for the oxidation |
|
Chapter 18: Electrochemistry
Key topics: Galvanic cells Cell Notation (shorthand for describing a galvanic cell): Standard Standard cell potential : The cell potential under standard state |
|
1 10 Fundamentals This chapter introduces the electrochemical cell
Consider the electrochemical cell shown below where the electrolyte is stagnant It consists of zinc In galvanic cells, the half cell reactions occur spontaneously and a potential difference is automatically One major problem for students in |
|
Electrochemistry example problems
25 avr 2017 · Problem 1: At 25 , the standard reduction potential of Zn2 is E o 0 763 V A galvanic cell voltage is positive is made by coupling a standard |
|
Electrochemistry - MT Educare
Electrochemical Series 9 CELL POTENTIAL OR EMF OF A CELL The difference between the electrode potentials of two half cells is called cell potential |