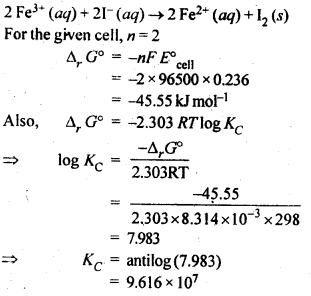electrochemistry problem set answers
|
Solutions for Electrochemistry Problem Set
1 a Calculate the cell potential and free energy Ag s) ( 1+ Ag aq ( 1 0M) 2+ aq ( 1 0M) Cu s) Cu( Anode (oxidation) Ag (s) Ag1+ + 1 Cathode (reduction Cu2+ + 2e- From the table of reduction potentials we can Eanode 0 7996 volt Ecathode 0 3402 volt And then calculate the standard cell potential |

Electrochemistry Practice Problems

Electrolysis & Electroplating Practice Problems

Cell Potential Problems
|
Test4 ch19 Electrochemistry Practice Problems.pdf
Jasperse. Electrochemistry. Extra Practice Problems Spontaneous Voltaic Electrochemical Cells ... The following reaction occurs in basic solution. |
|
ANSWERS TO PROBLEM SET No.2 - ELECTROCHEMISTRY 1
to Be need metals with oxidation potentials greater than 1.97 V (or reduction potentials more negative than -1.97 V). Ba |
|
Solutions for Electrochemistry Problem Set
Ordibehesht 4 1380 AP Notice that in this reaction the cell potential is positive |
|
Untitled
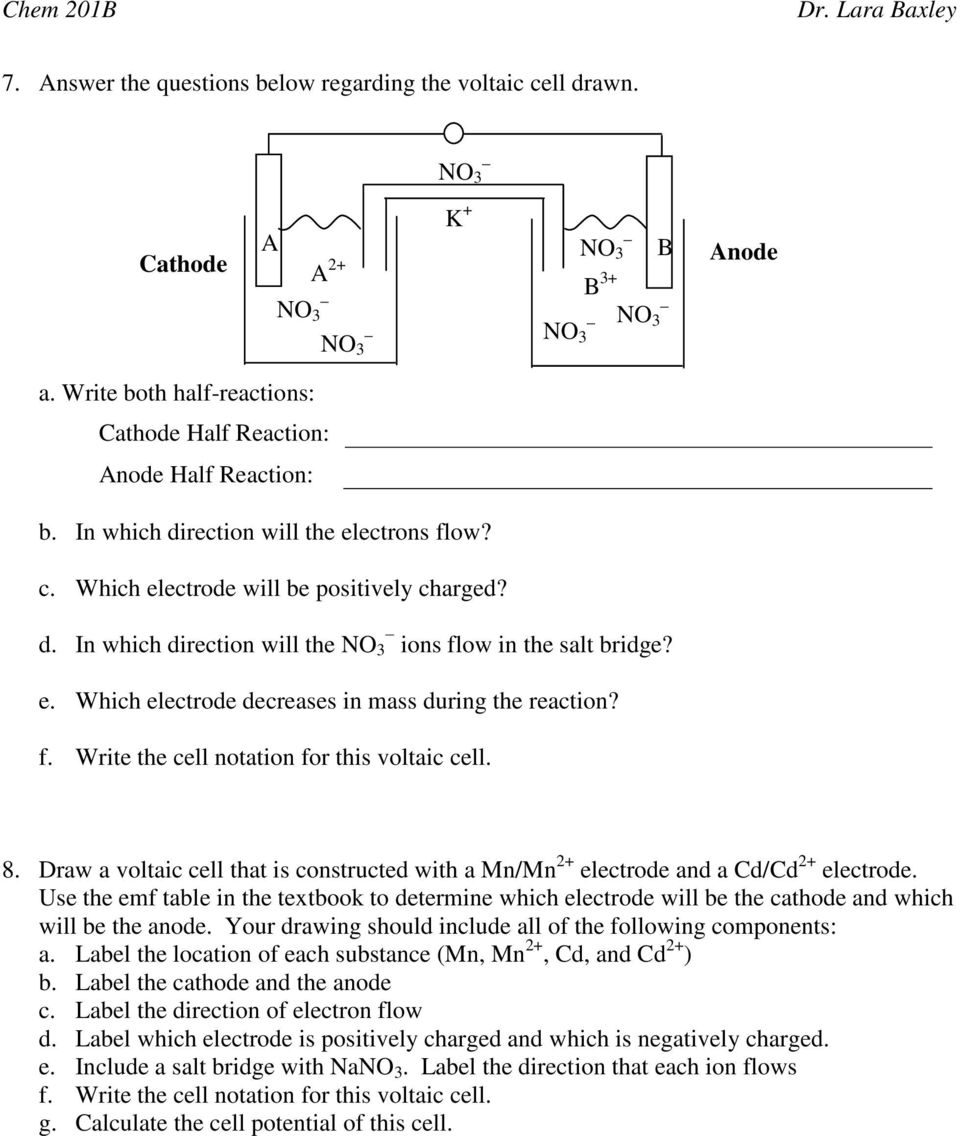
Ch.17- Electrochemistry: Practice Problems I 3) A galvanic cell is set up using the following half-reactions: mtanda. 3+. Al³ +3e ??? AI. |
|
Test4 ch19 Electrochemistry Practice-answers-Marked
Jasperse. Electrochemistry. Extra Practice Problems. Oxidation Numbers p1 Free Energy and Equilibrium p10. Balancing Redox; Electrons Transferred; Oxidizing. |
|
PROBLEM SET No. 2 - ELECTROCHEMISTRY 1. Olmsted 17.25 2
What metal will plate out first if this solution is electrolyzed? (HINT: Use the Nernst equation to calculate for each half- reaction.) 10. A voltaic cell |
|
Chem 447---Electrochemistry---Problem Set # 2 1) Please answer
a) In highly acidic samples the pH measured by glass electrode with a Ag/AgCl reference (with. 0.1M KCl as internal reference electrolyte) is likely to be |
|
Core practical 10: Construct electrochemical cells and measure
Clean the strips of zinc and copper using sandpaper. 2. Set up a zinc half-cell by pouring 50 cm3 of zinc sulfate solution into the 100 cm3 beaker and |
|
Utilization of Open-Ended Problems Model to Analyze Students
Creative Thinking Ability on Electrochemistry Lesson to the correct answer (Hürsen et al. 2014; ... problem set with various correct answers. |
|
Solutions for Electrochemistry Problem Set
24 avr 2001 · Solutions for Electrochemistry Problem Set Calculate the cell potential and free energy available for the following electrochemical systems |
|
ANSWERS TO PROBLEM SET No2 - ELECTROCHEMISTRY 1
to Be, need metals with oxidation potentials greater than 1 97 V (or reduction potentials more negative than -1 97 V) Ba, Ca, Cs, Li, Mg, K and Na all fit this |
|
Chem 447---Electrochemistry---Problem Set &# 2 1) Please answer
Chem 447---Electrochemistry---Problem Set # 2 1) Please answer the following questions True or False---and provide a sentence or two to support your answer |
|
Test4 ch19 Electrochemistry Practice Problems
A voltaic cell is constructed based on the oxidation of zinc metal and the reduction of silver cations Solutions of silver nitrate and zinc nitrate also were used |
|
Ch 17 Electrochemistry Practice Problems I - keypdf
Ch 17 - Electrochemistry: Practice Problems I Habe 3) A galvanic cell is set up using the following half-reactions: solutions and electrodes shown below |
|
Electrochemistry Problems And Solutions
Solved Examples On Electrochemistry - Study Material for Solutions for Electrochemistry Problem Set Constants: F 96484 56 coul mole 1 T (273 15 25 ) K M |
|
Electrochemistry review worksheet answers - Squarespace
21 fév 2021 · 004 Moles of the worksheet - Balancing Redox oxidation reactions using Taurus #'s (*answers) Galvani Cell Worksheet (to complete homework |
|
Electrochemistry Problems And Answers - EduGeneralorg
Read Book Electrochemistry Problems And Answers following electrochemical systems Solutions for Electrochemistry Problem Set Electrochemistry Practice |