is table salt a homogeneous or heterogeneous mixture
|
CLASSIFICATION OF MATTER WORKSHEET HOMOGENEOUS VS
11. rubbing alcohol homogeneous 8. sugar water homogeneous heterogeneous. PURE SUBSTANCES VS. MIXTURES ... 17. table salt Pure substance (C). |
|
Chemical Mixtures
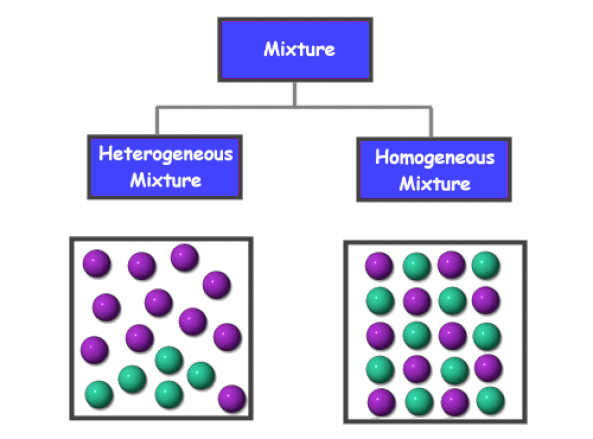
Within the categories of homogeneous and heterogeneous mixtures there are more In this example the water is the solvent and the salt is the solute. |
|
General Chemistry 101 Chapter 1 Review Questions 1) Molecules
E) homogeneous mixtures. C) An example of an amorphous solid is table salt (NaCl). ... 52) Choose the heterogeneous mixture from the list below. |
|
Untitled
13. heterogeneous mixture. Q14. homogeneous mixture ? mixture e. a special name for a homogeneous mixture ... 17. table salt (Nace) compound. Homag.pix. |
|
3.3: Classifying Matter According to Its Composition
Mar 30 2020 Each of the layers is called a phase. Example. Identify each substance as a compound |
|
Lab #2
The separation of the components of a mixture is a problem frequently encountered silicon dioxide (SiO2 sand) |
|
Untitled
Ex. Carbonated water sucrose (table sugar) in water |
|
Chemistry: Classifying Matter Name______________________
Similarly if the material is a mixture |
|
Unit 2A: Matter
mechanical mixtures are heterogeneous while others such as In one test tube |
|
16.1 Classifying Matter
Figure 16.1: Carbonated soft drinks are homogeneous mixtures. Figure 16.2: Chicken noodle soup is a heterogeneous mixture. 16.1 Classifying Matter. |
|
33: Classifying Matter According to Its Composition
30 mar 2020 · Ordinary table salt is called sodium chloride to confuse a homogeneous mixture with a pure substance because they are both uniform |
|
Chemical Mixtures
In a homogenous mixture all the substances are evenly distributed throughout the mixture (salt water air blood) In a heterogeneous mixture the substances are |
|
35: Pure Substances and Mixtures - Chemistry LibreTexts
23 août 2019 · A homogeneous mixture is combination of two or more substances that are so intimately mixed that the mixture behaves as a single substance |
|
Iesc102pdf - NCERT
List the points of differences between homogeneous and heterogeneous mixtures 2 2 What is a Solution? A solution is a homogeneous mixture of two or more |
|
What is iodised table salt ? - Doubtnut
26 avr 2023 · Iodised table salt is homogenous mixture Was this answer helpful? 544 Postée : 26 avr 2023 |
|
Classify table salt as one of the following a element b compound c
Table salt involves atoms of two elements named sodium and chlorine The table salt will be classified as a homogenous mixture only when it is dissolved |
|
Salt water homogeneous or heterogeneous - Squarespace
pdf Saltwater contains dissolved salt so it's a homogeneous mixture as we can't separate salt from it directly But it is also called a heterogeneous mixture |
|
UNIT 3: Pure substances and mixtures 1º ESO - Claseshistoriacom
A dissolution (solution) is a homogeneous mixture composed of two or more substances EXAMPLES OF SOLUTIONS SOLUTE GAS LIQUID SOLID SOLVENT |
|
(b) substance: (c) mixture: (Q2) Give an example of an eleme
Heterogeneous: contains salts water mud decayed plant etc (b) helium gas: element (c) sodium chloride (table salt): compound (d) a bottle |
Is table salt a compound or homogeneous mixture?
Table salt is a compound consisting of equal parts of the elements sodium and chlorine. Salt cannot be separated into its two elements by filtering, distillation, or any other physical process.Why is table salt a homogeneous mixture?
A homogeneous mixture is a mixture in which the composition is uniform throughout the mixture. The salt water described above is homogeneous because the dissolved salt is evenly distributed throughout the entire salt water sample.What type of mixture is table salt?
Common salt is a compound made up of Sodium and chlorine atom. These elements are combined chemically in a fixed ratio to form Common salt. We can not separate these components easily by physical means. So, Common salt is a compound, not a mixture.- The table salt will be classified as a homogenous mixture only when it is dissolved in any appropriate solvent like water. It cannot be classified as a heterogeneous mixture because only a single phase is present.
|
Chapters 1 and 3
Ex Table salt, water (always H 2 O) Heterogeneous mixture: does not blend smoothly throughout and in Homogeneous mixture: has constant composition |
|
Problem Set 1
Freshly brewed coffee and vinegar (a, b) are homogeneous mixtures A slice of white bread is considered to be heterogeneous because its crust is We can distinguish between table salt, sand, and copper by examining their color (salt is |
|
Classification Of Matter - VCC Learning Centre - Vancouver
7) A mixture of oil and water is a homogeneous mixture 8) In a mixture, 14) Homogeneous and heterogeneous mixtures are pure substances 3) table salt |
|
MIXTURES, ELEMENTS AND COMPOUNDS - Junta de Andalucia
Mixture 2 Homogeneous 3 Heterogeneous 4 Solution 5 Element 6 Compound CONTENTS 1 When salt is mixed with water, the salt ______ in the water Salt is ______ in water Make a table of your results substance It will |
|
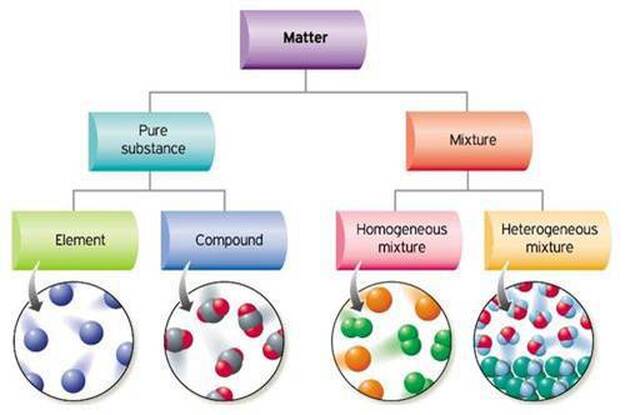
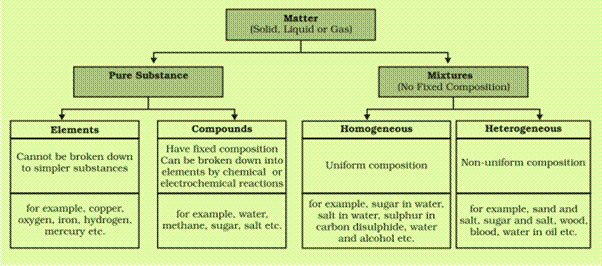
Classification of Matter
Table salt is a compound made Many substances around you are heterogeneous mixtures homogeneous mixture of water, sugar, coloring, and flavoring |
|
Word Pro - exper02
When two solutions (homogeneous mixtures) are brought together there may be a After all the sea is largely a solution of sodium chloride in water and table salt pure NaCl(s) from a heterogeneous mixture sand by a process involving |
|
16 Homogeneous and Heterogeneous Mixture - images
Ordinary table salt is called sodium chloride It is considered a compound, or a pure substance, because it has a uniform and definite composition All samples of |
|
Chemistry: Classifying Matter - Paulding
earn full credit Material Pure Substance or Mixture Element, Compound, Homogeneous, Heterogeneous concrete Mixture Heterogeneous sugar + pure water |
|
Matter Classification Review 1 - Quizpdf
Salt water 3 Tron (Fe) 4 Sugar (Cizitz201) 5 Distilled Water (H2B) 6 Shaving Cream 18 State whether each of the following is a homogeneous or heterogeneous mixture carbon dioxide (cos) Compound 17 table sait (itace) compound |



/definition-of-heterogeneous-mixture-and-examples-605206_final23-ecfa4da6517640429448462eae1f09f7.png)