alkalinity measurement
|
Total Alkalinity Measurement in Natural Waters
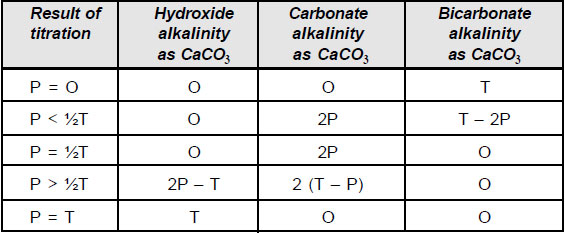
Alkalinity is measured commonly by titration using either a burette or the drop count technique A sample is titrated with an acid solution |
|
Alkalinity
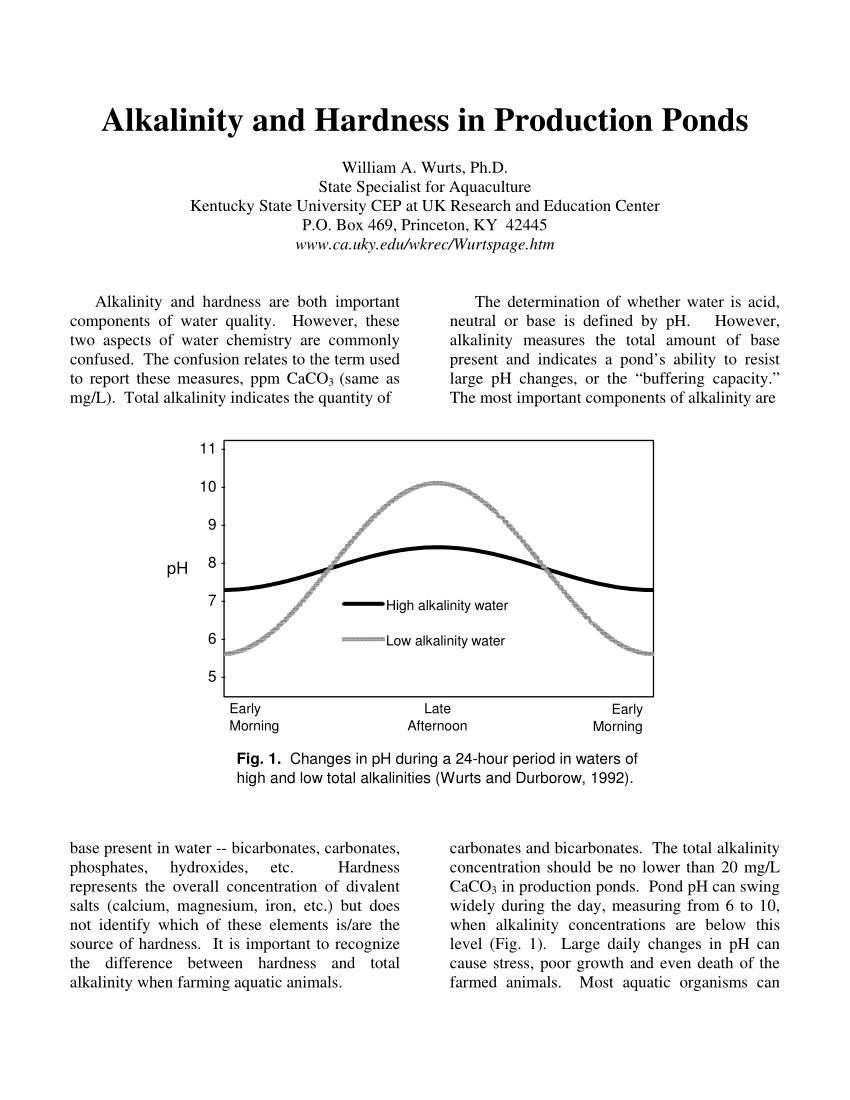
Alkalinity is a chemical measurement of a water's ability to neutralize acids Alkalinity is also a measure of a water's buffering capacity or its ability |
What is the unit of alkalinity?
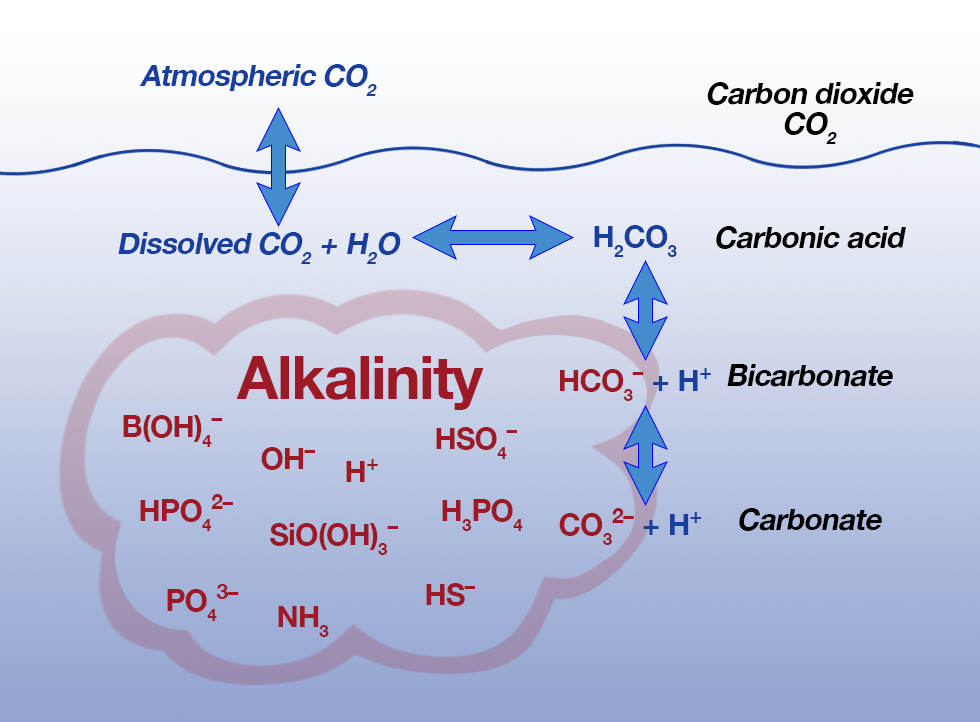
Alkalinity is a measure of the presence of bicarbonate, carbonate or hydroxide constituents.
Concentrations less than 100 ppm are desirable for domestic water supplies.
The recommended range for drinking water is 30 to 400 ppm.How do you calculate alkalinity?
The unit of measurement for alkalinity is mg/L CaC03.
What is the significance of alkalinity? Alkalinity is the measure of the resistance of water to the lowering of pH when acids are added to the water.
Acid additions generally come from rain or snow, though soil sources are also important in some areas.
|
Technical Bulletin 517 – Total Alkalinity Measurement in Natural
Measurement in Natural Waters. Alkalinity measurement is important in the control of water and wastewater treatment. In a sample alkalinity is defined as |
|
Alkalinity Alkalinity is a chemical measurement of a waters ability to
Alkalinity is also a measure of a water's buffering capacity or its ability to resist changes in pH upon the addition of acids or bases. Alkalinity of natural |
|
Field Measurement of Alkalinity and pH
Field Measurement of. Alkalinity and pH. By IVAN BARNES. GEOCHEMISTRY OF WATER. GEOLOGICAL SURVEY WATER-SUPPLY PAPER 1535-H. Method for determinations of pH |
|
Alkalinity Measurement by Titration to a Designated Endpoint pH
Jul 1 2017 This SOP describes the procedure for measuring alkalinity by titration to a specified endpoint (potentiometric method). This method is based on ... |
|
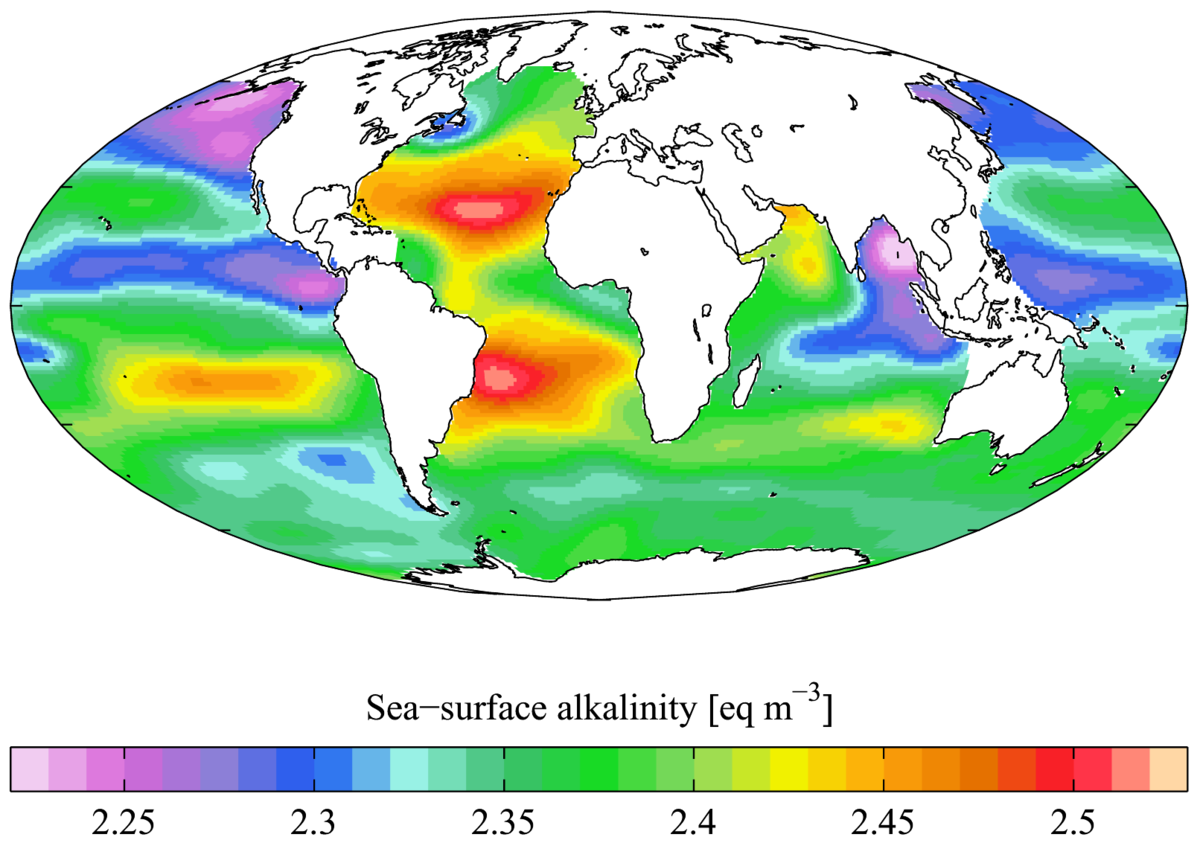
Interpreting measurements of total alkalinity in marine and estuarine
Jul 29 2020 Total alkalinity. Dissolved organic matter. Titration method. Marine carbonate system. Marine CO2 system. Seawater. Measurement ... |
|
Contribution of non-carbonate anions to total alkalinity and
Oct 31 2011 TAlk has also been used |
|
Chapter 11: pH and Alkalinity Voluntary Estuary Monitoring Manual
The pH measurements are given on a scale of 0.0 to 14.0 (Figure 11-1). Pure water has a pH of 7.0 and is neutral; water measuring under 7.0 is acidic; and that. |
|
Alkalinity
Why measure alkalinity? Alkalinity measurement is important in many environmental and industrial applications such as: Water quality – The alkalinity level |
|
Guide to Best Practices for Ocean CO2 Measurements
Dec 13 2007 SOP 3b Determination of total alkalinity in sea water using an open-cell titration. SOP 4 Determination of p(CO2) in air that is in ... |
|
SOP for the Measurement of Hardness and Alkalinity in Streams
WATER QUALITY PROGRAMS DIVISION. Standard Operating Procedure for the Measurement of. Hardness and Alkalinity in Streams. Revised and Adopted November 2005. |
|
Total Alkalinity Measurement in Natural Waters - Thermo Fisher
Alkalinity is measured commonly by titration, using either a burette or the drop count technique A sample is titrated with an acid solution, which neutralizes the alkaline species present The endpoint is determined by observing a color change or by titrating to a pH value of 4 5, using a pH electrode as an indicator |
|
Field Measurement of Alkalinity and pH
The following steps were found necessary for accurate pH and alkalinity determinations: 1 Measure water temperature with the thermometer in shade 2 Set |
|
PH Alkalinity Hardness pH Alkalinity Hardness - Wisconsin DNR
Thus, pH is the measurement of hydrogen ion (H+) activity • Pure water dissociates to yield equivalent concentrations of hydrogen [H+] and hydroxide [ OH-] ions: |
|
Laboratory Experiment 2: Alkalinity and Acidity
Alkalinity is measured by titrating a sample with acid A titration curve of a bicarbonate containing water is presented in Figure 2 Alkalinity is significant in many uses and treatments of natural waters and wastewaters |
|
Determination of Alkalinity by Titration (Short Method) 1 Scope and
1 1 Alkalinity determination by titration is used when analysis by an auto analyzer is not possible due to the color or turbidity of a given sample MMSD routinely |
|
70 EXPERIMENT ON DETERMINATION OF ALKALINITY OF WATER
The alkalinity of water can be determined by titrating the water sample with Sulphuric acid of known values of pH, volume and concentrations Based on |
|
2320-Alkalinitypdf
Continue adding titrant and measure PH until pH 9 is reached Construct the titration curve by plotting observed pH values versus cumulative milliliters titrant |