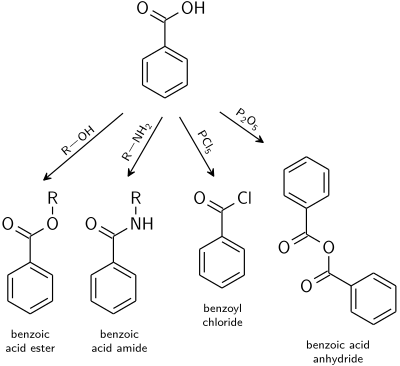
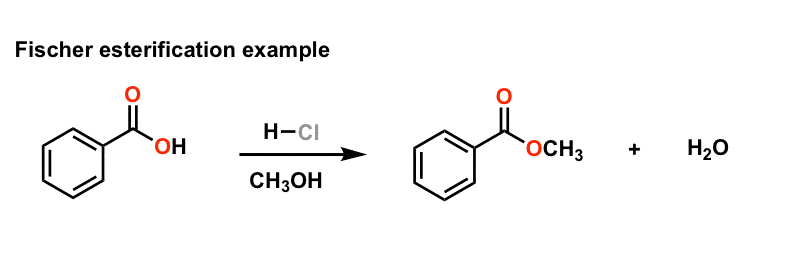
acid catalyzed hydrolysis of ethyl benzoate
What is the hydrolysis product of ethyl benzoate?
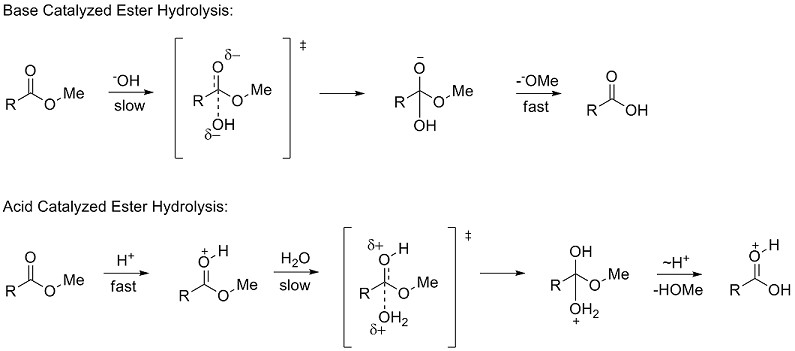
Treatment of an ester with aqueous base results in the hydrolysis of the ester with the formation of the salt of the acid and an alcohol.
For example, hydrolysis of ethyl benzoate yields sodium benzoate and ethyl alcohol.What is the acid hydrolysis of benzoic acid?
Acid hydrolysis occurs when benzoic anhydride is treated with sodium borohydride, followed by a reduction reaction.
As a result, benzoic acid and benzyl alcohol are formed.The base promoted hydrolysis of ethyl benzoate is shown below as an example.
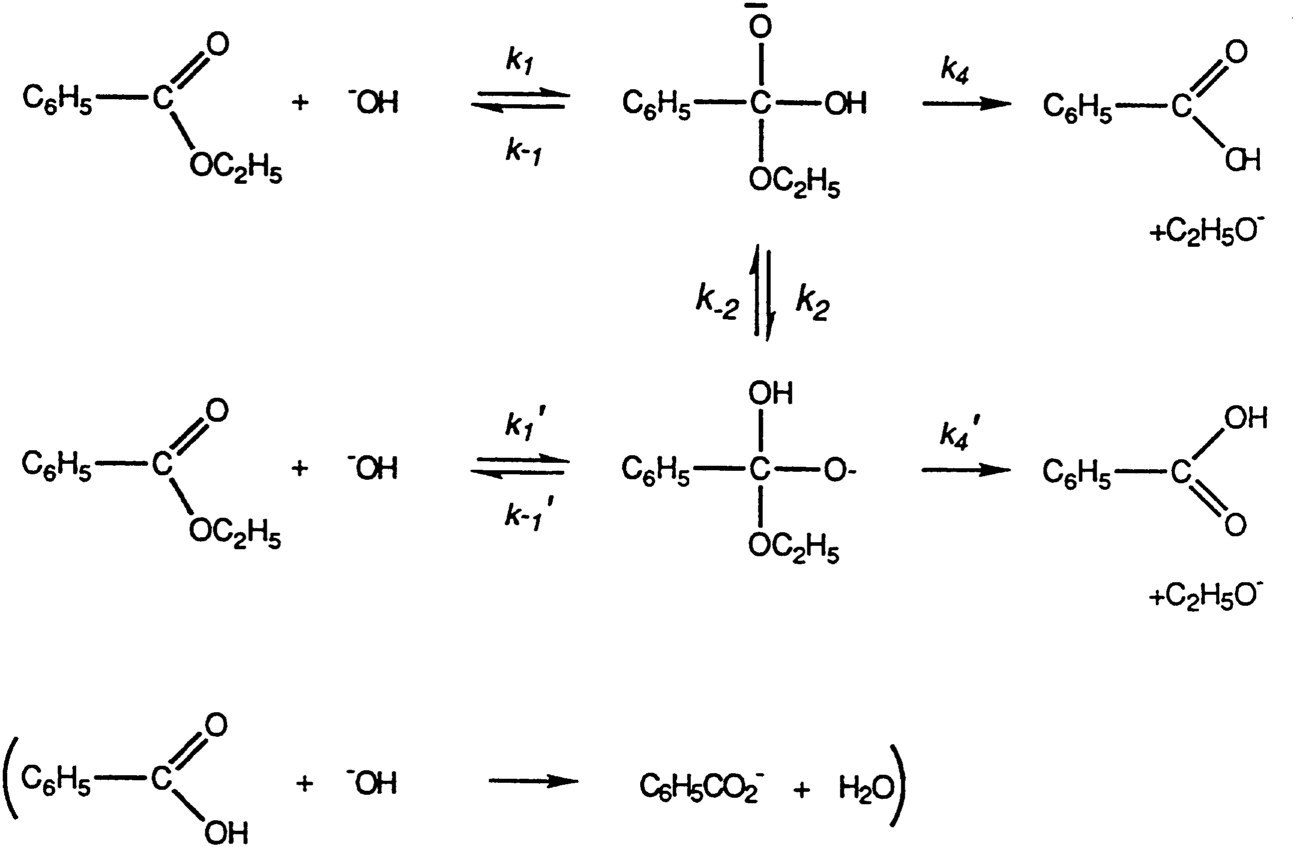
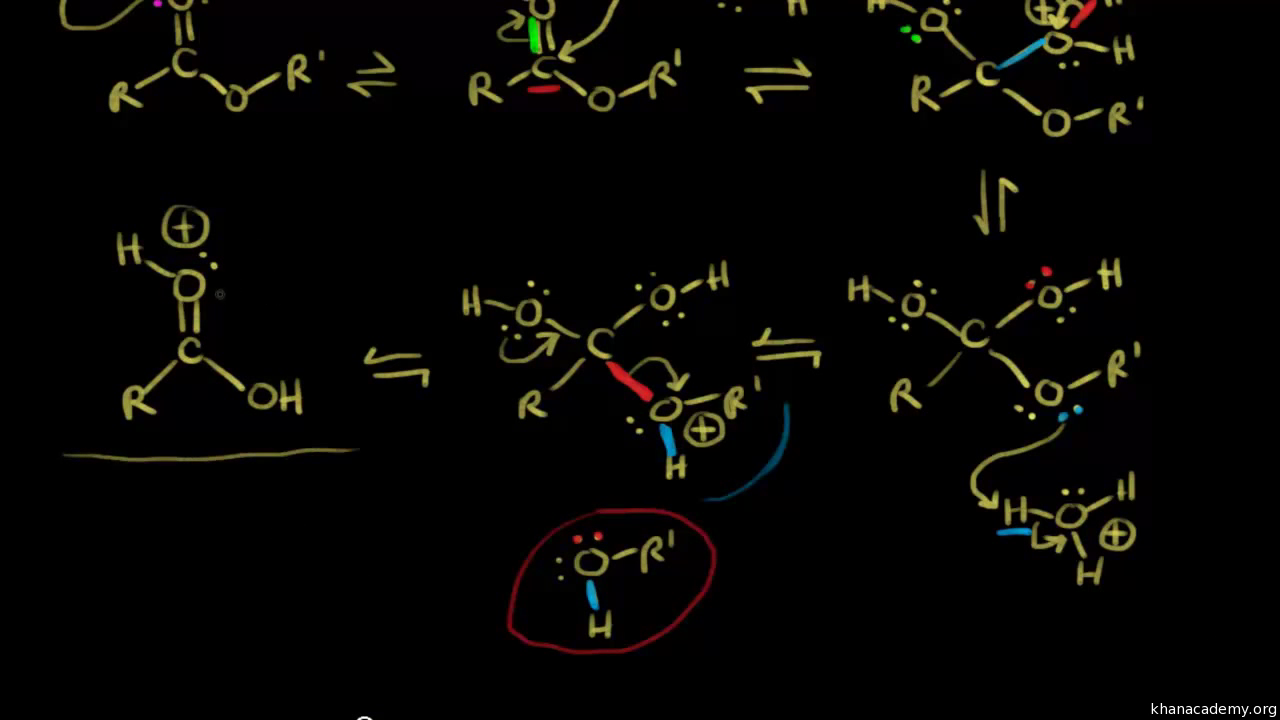
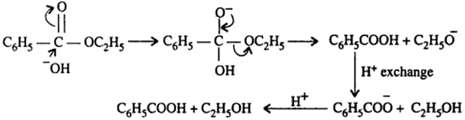
The mechanism for the base promoted hydrolysis reaction begins with the nucleophilic hydroxide reacting with the electrophilic carbonyl carbon atom to form the tetrahedral intermediate.30 mai 2020
What happens when ethyl benzoate reacts with water?
Ethyl benzoate is an ester.
In the presence of H2O and heat, and ester will undergo hydrolysis to produce a carboxylic acid and an alcohol, namely, benzoic acid and ethanol.
FYI - This reaction is the reverse of forming an ester between a carboxylic acid and an alcohol.
|
Benzoic Acid from Ethyl Benzoate by Base Hydrolysis
This process is called base hydrolysis (or saponification) of an ester and is used in this experiment to first obtain sodium benzoate solution, and then benzoic acid |
|
215 HH W11-final-key
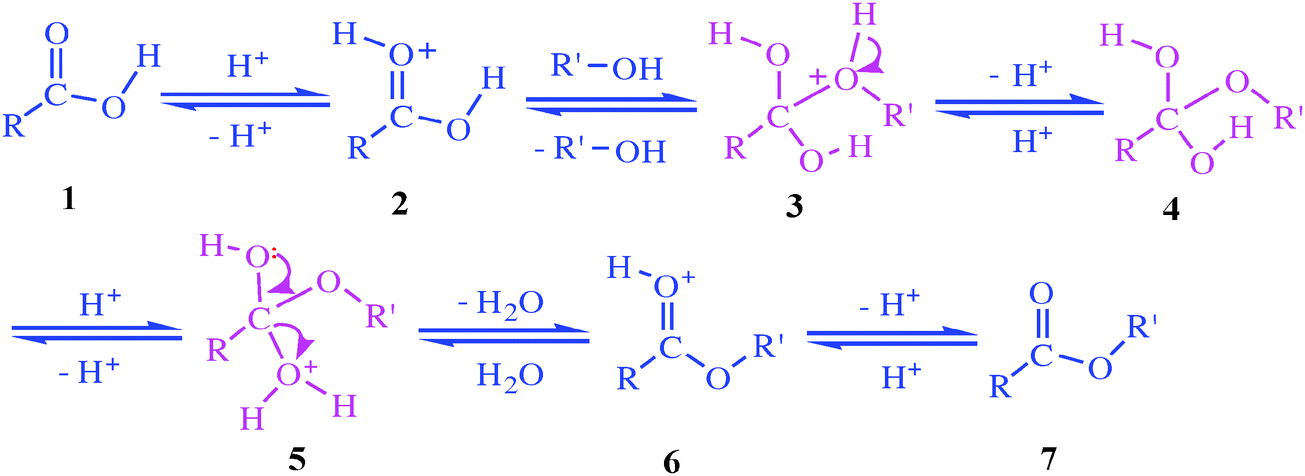
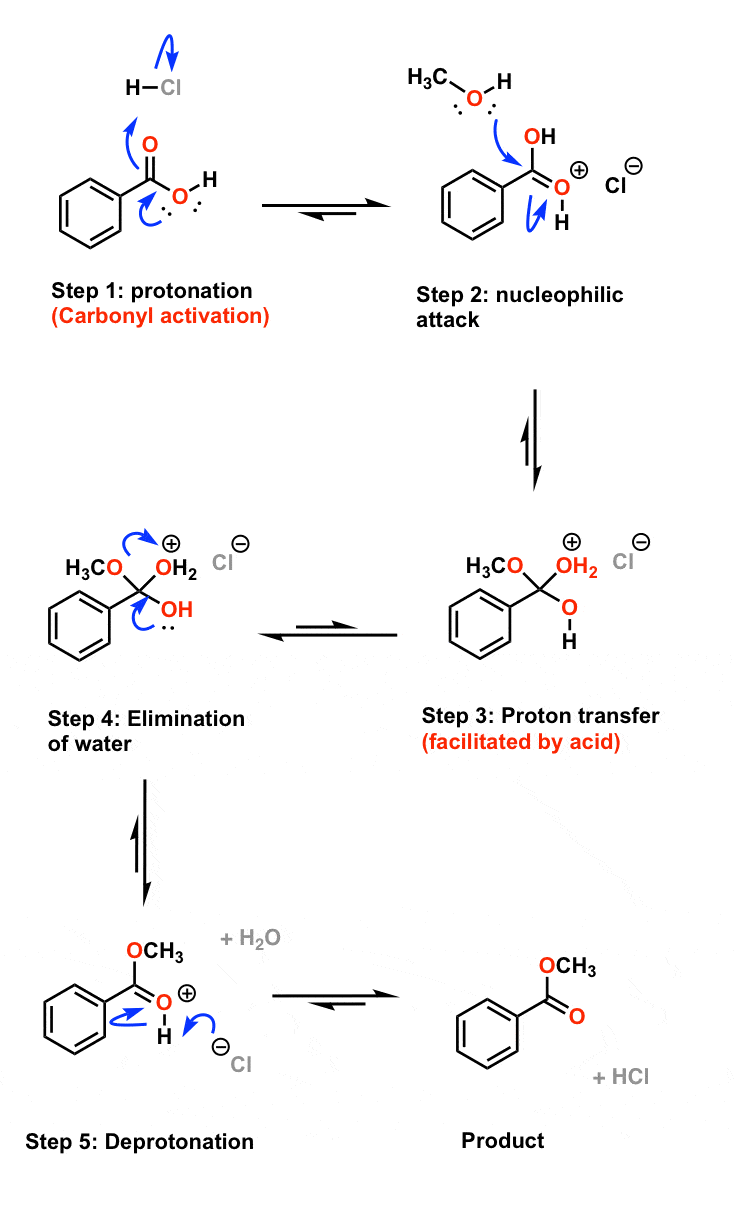
I (44 points) (1) (22 points) Show in the box below a step-by-step, curved-arrow mechanism for the acid- catalyzed hydrolysis of methyl benzoate to benzoic acid |
|
Esterificationpdf
ethyl benzoate 2 The reaction requires an strong acid catalyst (e g , H2S04 or H3PO») and equilibrium is established with a matter of hours The reverse reaction in the above mechanism is known as acid-catalyzed hydrolysis of an ester |
|
Chem 22 Homework set 9 1 Suggest a method for converting
For example, consider the course of the acid-catalyzed hydrolysis of when ethyl benzoate is hydrolyzed in 18O-labelled water (i e 18OH2 instead of the usual |




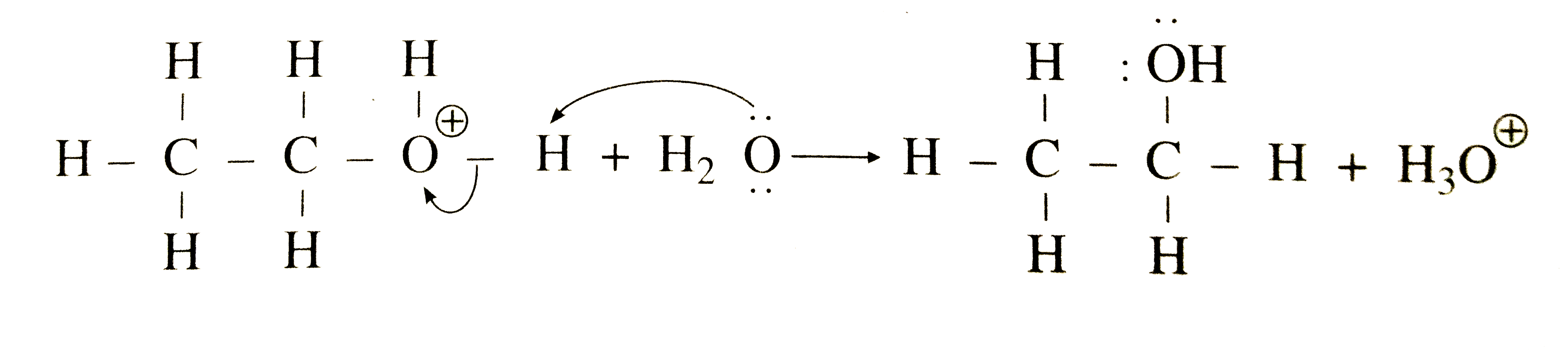








![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://demo.fdocuments.in/img/742x1000/reader019/reader/2020031102/5750a0ef1a28abcf0c8fc478/r-1.jpg?t\u003d1610082576)








![acid base catalysed Ester hydrolysis - [PDF Document] acid base catalysed Ester hydrolysis - [PDF Document]](https://imgv2-1-f.scribdassets.com/img/document/416284607/original/9b74b75b77/1614205044?v\u003d1)