electrochemical cell notes pdf
|
10 Fundamentals
1 0 Fundamentals This chapter introduces the electrochemical cell its components basic definitions and the processes that take place during electrolysis The difference between thermodynamics and kinetics is explained through the concepts of reduction potential and overpotentials 1 1 The electrochemical cell |
|
Chapter 5
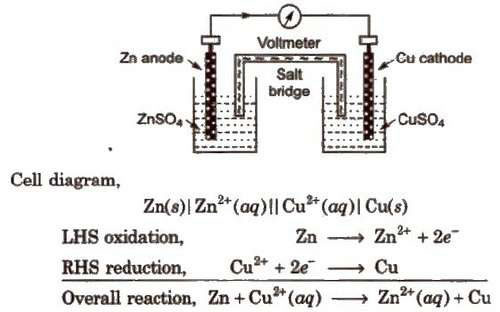
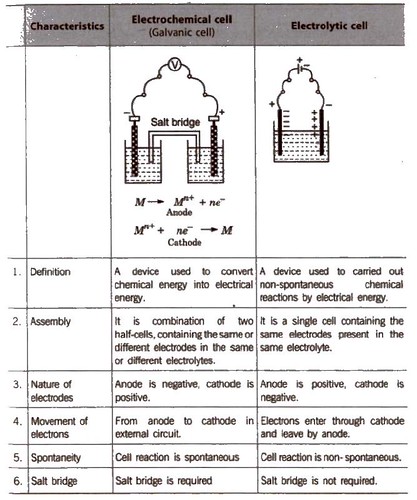
Electrochemical Cell & Galvanic Cell: Electrochemical cell is a system or arrangement in which two electrodes are fitted in the same electrolyte or in two different electrolytes which are joined by a salt bridge Electrochemical cell is of two types (a) Electrolytic Cell (b) Galvanic Cell or Voltaic Cell |
|
1 Introduction to Electrochemical Cells
The electrolyte is an ion conducting material which can be in the form of an aqueous molten or solid solution while the separator is a membrane that physically pre- vents a direct contact between the two electrodes and allows ions but not electrons to pass through; it therefore ensures electrical insulation for charge neutralization in both |
|
Chapter 21: ELECTROCHEMISTRY TYING IT ALL TOGETHER
Electrochemistry is physical of electron movement in In looking for a context for all of chemistry centers around bonds Chemical bonds are formed around nuclei Electrochemistry delivery or measure of a source delivered or produced and the known Note that there will be and acid/base chemistry |
|
CHEM-1100 Chapter 19 Lecture Notes
Electrochemical Cells An electrochemical or voltaic or galvanic cell is an experimental apparatus for generating an electric current with a spontaneous redox reaction Practical applications include the many kinds of batteries which generate electric current for practical purposes Electrochemical cells have various |
What is an electrochemical cell?
A device used to convert the chemical energy produced in a red-ox reaction into electrical energy is known as electrochemical cell. The value of reduction potential of an electrode may be increased by increasing the concentration of metal ions in contact with metal electrode or increasing the temperature.
What are the three types of electrochemical chemistry?
in an electrochemical cell containing the analyte. The three main categories are potentiometry (the difference in electrode potentials is measured), coulometry (the cell's current is measured over time), and voltammetry (the cell's current is measured while actively altering the cell's potential).
Can electrochemical cells be thermodynamics?
Electrochemical cells can be thermodynamics. Electrolytic work cell: must be done for negative.) 5. Tying Electrochemistry to In electrochemistry, the quantity potential energy of the system. or E = 6 V battery. We can electrochemistry to the thermodynamic through the equation: 6.
What are electrochemical processes?
The electrochemical processes are the cathodic reduction of hydrogen ions (protons) at the negative electrode (2H+ 2e− + 2e−).
|
Electrochemical Cell / Galvanic Cell / Voltaic cell L 1 Functions of
Chemical reaction occurs in the cell. Oxidation at one electrode and the electrons given out are consumed for reduction at other electrode. The electrode at |
|
Chapter 5
Galvanic Cell: The device used to convert the chemical energy produced on a red-ox reaction into electrical energy is called an electrochemical cell or |
|
Detailed Notes - Topic 1.11. Electrode Potentials and Cells - AQA
Electrochemical cells use ?redox reactions?as the ?electron transfer?between products creates a flow of electrons. This flow of charged particles is an |
|
Dr.Pusalkar Notes ELECTROCHEMISTRY S.Y.B.Sc
Q.2) Define the following terms: i) Cell ii) Electrochemical cell iii)Anode iv) Cathode v) Oxidation vi) Reduction vii) Redox reaction viii) oxidizing agent. |
|
Electrochemistry Electrochemistry
It now functions as an electrolytic cell a device for using electrical energy to carry non-spontaneous chemical reactions. Both types of cells are quite |
|
Electrochemistry
Electrochemistry is the study of reactions in which charged particles (ions or electrons) cross the interface Figure 2: A simple electrochemical cell. |
|
Corrosion Basics.pdf
Corrosion cell. Corrosion occurs due to the formation of electrochemical cells. In order for the corrosion reaction to occur five things are necessary. |
|
Physical Science - Electrochemistry
4.1 Electrochemical reactions. 5. 4.2 Electrochemistry - a special case of redox reactions. 8. 4.2.1 Constructing an electrochemical cell. |
|
Chapter 18
Oxidation/Reduction Reactions in Electrochemical Cells An electrochemical cell consists of two conductors called electrodes each. |
|
General Chemistry II Chapter 19 Lecture Notes Electrochemistry
An electrochemical or voltaic or galvanic cell is an experimental apparatus for generating an electric current with a spontaneous redox reaction. Practical |
|
Electrochemistry Notes - LMS
Electrochemistry Notes Description of Cells Galvanic Cells: anode (+) cathode (-) Anode (+) is always written first Oxidation at the anode Reduction at the |
|
Electrochemical Cell & Galvanic Cell - VSSUT
Electrochemical cells EMF Measurement of EMF Relation between EMF free energy change of cell reaction Electrode potential with reference to |
|
Electrochemistry Ch 19 Notes: - Moorpark College
These cells use spontaneous redox reactions to produce electric current, electrons flow from one terminal (anode) to another (cathode) when connected by an |
|
PHYSICAL SCIENCE-Electrochemistrypdfpdf
4 1 Electrochemical reactions 5 4 2 Electrochemistry - a special case of redox reactions 8 4 2 1 Constructing an electrochemical cell 8 4 3 Examples of cells |
|
AP CHEMISTRY NOTES 12-1 ELECTROCHEMISTRY - Azle ISD
Galvanic (Voltaic) Cells – these cells are spontaneous and react with no outside In an electrochemical cell, oxidation and reduction occur simultaneously |
|
Introduction to Electrochemical Cells 1 - Wiley-VCH
Scientifically batteries are referred to as electrochemical or galvanic cells, due to to polarization ( http://www msm cam ac uk/Teaching/mat1b/courseB/BH pdf ) In summary, it can be stated that in order to maximize power density, it is impor- |
|
7 ELECTROCHEMISTRY - Mahesh Tutorials Science
In Daniell Cell, Zn acts as the anode and Cu acts as the cathode 5 ELECTROLYTIC CELL These electrodes are dipped in and electrolytic solution containing |
|
Electrochemical cells
electrode compartment If two different electrolytes are used: Electrolytic cell: electrochemical cell in which a non-spontaneous reaction is driven by an external |
|
JF Chemistry 1101 2011 Basic Electrochemistry L 16-18 - School of
An electrochemical cell is formed by coupling together individual oxidation and reduction processes in a specific http://www nature com/nature/journal/v414/ n6861/ pdf /414338a0 pdf A summary diagram for the stoichiometry of electrolysis |
|
General Chemistry II Chapter 19 Lecture Notes Electrochemistry
An electrochemical or voltaic or galvanic cell is an experimental apparatus for generating an electric current with a spontaneous redox reaction Practical |