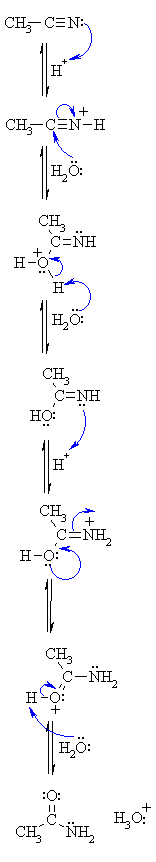
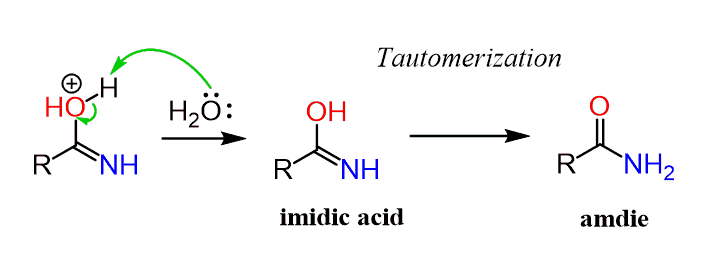
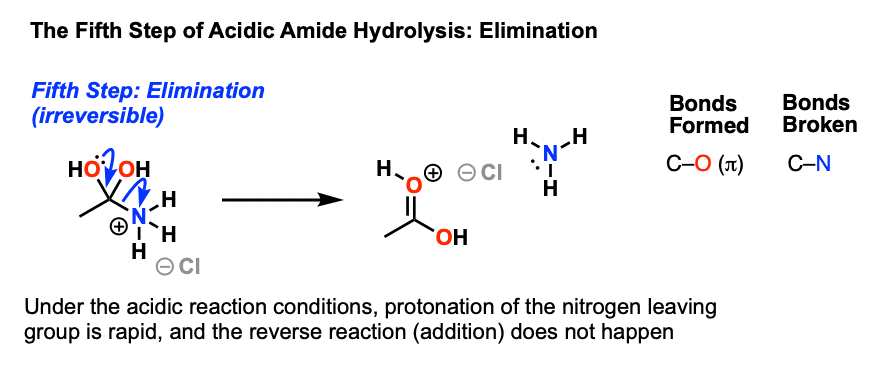
base catalyzed hydrolysis of nitriles
|
Base Catalyzed Hydrolysis of Nitriles
This irreversible reaction produces the salt of a carboxylic acid You may need to perform an acid workup if the product requires further chemical changes |
What is the alkaline hydrolysis of nitrile?
Alkaline hydrolysis of nitriles generally results in the formation of the salt of the carboxylic acid and ammonia, and is conveniently applied to the simple alkyl cyanides and also to nitriles such as α-cyanocarboxylic acids producing dicarboxylic acids which are unstable in hot mineral acid.
How does Dibal H reduce nitriles?
The hydride reagent Diisobutylaluminium hydride, or DIBAL-H, is commonly used to convert nitriles to the aldehyde.
Regarding the proposed mechanism, DIBAL forms a Lewis acid-base adduct with the nitrile by formation of an N-Al bond.
The hydride is then transferred to the carbon of the nitrile.What is base catalysed amide hydrolysis? It is a reaction of the amide with water in a basic medium.
In a basic medium, amide interacts with the water molecule to give a carboxylic acid and the salt of ammonia or amine salt.
What is base-catalyzed hydrolysis?
Base-catalyzed hydrolysis of ester occurs by SN2 pathway and is irreversible, because the end product of base-catalyzed hydrolysis of ester produces alcohol and carboxilate ion (not carboxylic acid), which being resonance stabilized shows very little tendency to react with alcohol.
|
Kinetic study of the base-catalyzed hydrolysis of aminocapronitrile
in methanol/water mixtures. This finding was explained by a single reaction mechanism similar to that for nitrile hydrolysis |
|
N. F. Jain and C. E. Masse Carboxylic acid derivatives can be
Hydrolysis of carboxylic acid esters amides |
|
Note Selective conversion of nitriles to amides by Amberlyst A-26
solution of nitrile and hydrogen peroxide (35%) at room temperature. Nitriles may be hydrolyzed to give either amides or or base' catalyzed hydrolysis |
|
The Acid-Catalyzed and Uncatalyzed Hydrolysis of Nitriles on
One of the best methods to synthesize carboxylic acids is the hydrolysis (acid- or base-catalyzed) of nitriles which occurs in two steps through the |
|
21.7 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
For example a typical reaction of nitriles is addition. Acid-Catalyzed Ester Hydrolysis Because esterification of an acid with an alcohol is a. |
|
Chapter 21: Carboxylic Acid Derivatives
The large boiling points of nitriles acids and The reactivity of acid derivatives can be correlated ... Base Catalyzed Hydrolysis of Nitriles. |
|
Chemoselective hydration of nitriles to amides using hydrated ionic
distinct steps upon treatment with strong acids or bases. especially transition metal catalyzed nitrile hydrolysis reactions have offered mild reaction. |
|
Untitled
Mechanism of the Reaction of Nitriles with Alkaline Hydrogen Peroxide. The alkaline hydrolysis: 0.1-0.4 M NaOH 1-2.5 M. MeCN or 0.1 M PhCN. |
| Selective Hydration of Nitriles to Corresponding Amides in Air with |
|
A mild alkaline hydrolysis of N- and NN-substituted amides and
nitrile to primary amide. From the alkaline hydrolysis of benzonitrile (14) pure benzamide can be isolated after refluxing in NaOH/MeOH/Dioxane for about |
|
The Acid-Catalyzed and Uncatalyzed Hydrolysis of Nitriles on
One of the best methods to synthesize carboxylic acids is the hydrolysis (acid- or base-catalyzed) of nitriles which occurs in two steps through the intermediate amide (eq Thus, the relative rates of nitrile hydrolysis correlate linearly with anticipated nitrile basicity |
|
NF Jain and CE Masse Carboxylic acid - thieme-connectde
Hydrolysis of carboxylic acid esters, amides, and nitriles can be either catalyzed by acid or base Synthesis by nitrile hydrolysis is widely employed because the |
|
Base Catalyzed Hydrolysis of Nitriles - Chemwiscedu
Base Catalyzed Hydrolysis of Nitriles Reaction: Mechanism: This irreversible reaction produces the salt of a carboxylic acid You may need to perform an acid |
|
217 HYDROLYSIS OF CARBOXYLIC ACID DERIVATIVES
Acid-Catalyzed Ester Hydrolysis Because esterification of an acid with an alcohol The hydrolysis of amides in base is analogous to the saponification of esters |