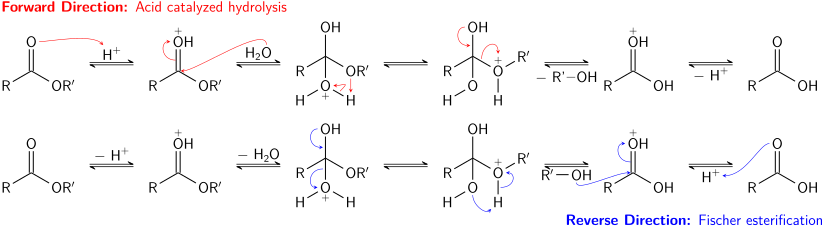
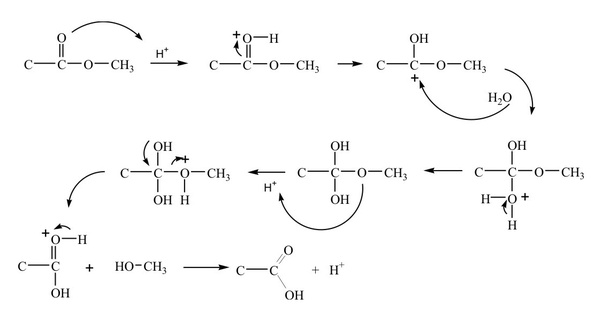
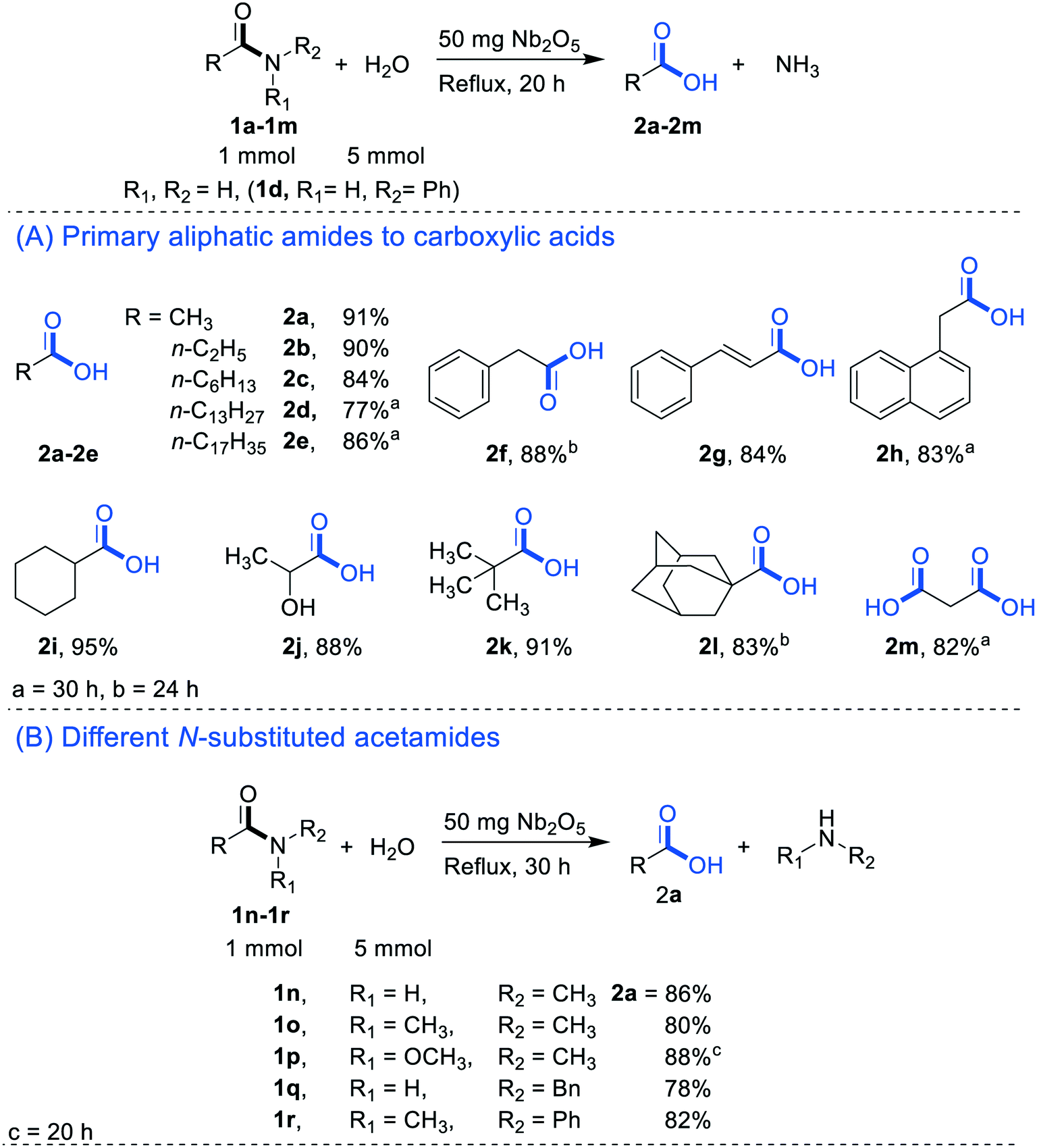
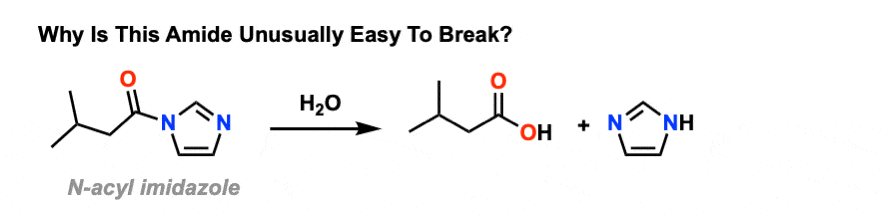
acid catalyzed hydrolysis of amide mechanism
The most general way for obtaining amides involves the activation of the carboxylic function by means the conversion of carboxylic acids into the corresponding acid chlorides [5,6,7,8].
Subsequently this reactive derivative is coupled with the appropriate amine to yield the corresponding amide.
What is the purpose of the acid catalyst in the hydrolysis of an amide?
The acid acts as a catalyst for the reaction between the amide and water.
The alkaline hydrolysis of amides actually involves reaction with hydroxide ions, but the result is similar enough that it is still classed as hydrolysis.
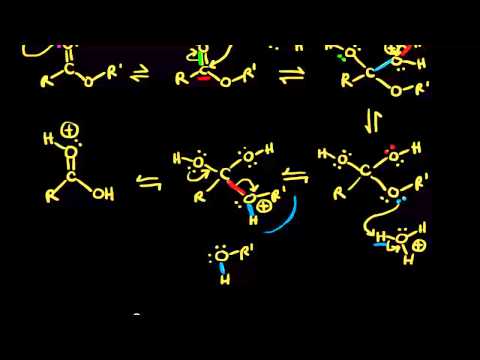
What is the mechanism of acid catalysed hydrolysis?
The nucleophile involved in acid-catalyzed hydrolysis of the ester is H2O.
The water molecule attacks the carbonyl carbon atom by donating the oxygen's lone pair of electrons and forming a bond.
It is a nucleophilic substitution reaction as water act as a nucleophile, by attacking the carbonyl carbon of ester.
What is the mechanism of acid hydrolysis of amides?
Hydrolysis of Amide
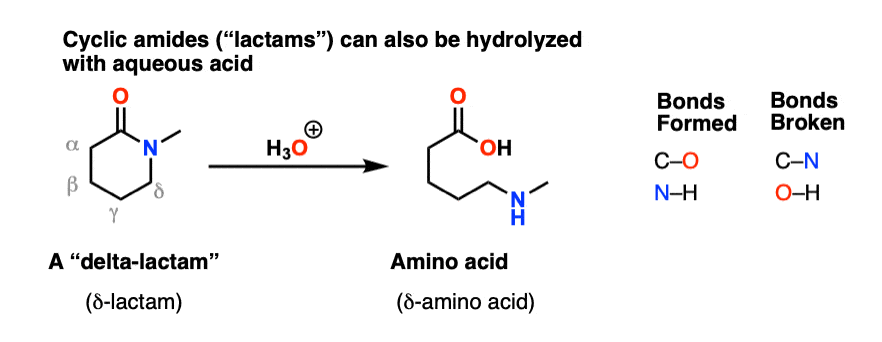
In an acidic medium, amide interacts with the water molecule to give a carboxylic acid and the salt of ammonia or amine salt.
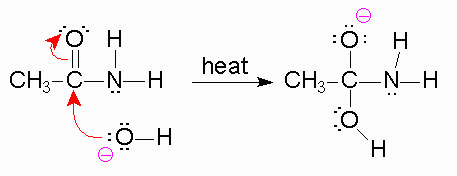
In a basic medium, amide interacts with the water molecule to give a carboxylic acid and the salt of ammonia or amine salt.
|
Catalyzed hydrolysis of amide and peptide bonds in proteins 1
The rates of hydrolysis by dilute acids of both a dissolved protein (egg albumin) and an insoluble protein (wool) are shown to depend not only on the |
|
Catalyzed Hydrolysis of Amides
The applicability of the acylenzyme mechanism to the tryptic hydrolysis of the amide bond has been studied. It was found that trypsin acetylated at the |
|
Observation of large amounts of carbonyl-18O exchange concurrent
A generalized mechanism of acid-catalyzed hydrolysis for the above amides is presented. Introduction. Scheme I. The generally accepted mechanism for |
|
On the hydrolysis mechanisms of amides and peptides
20 avr. 2018 There are large pH regions for which the hydrolysis is dom- inated by acid catalysis (0 < pH < 5 slope = ?1) or base catalysis (6 < pH < 10 |
|
Dilute acid-catalyzed amide hydrolysis: efficiency of the N
Dilute Acid-Catalyzed Amide Hydrolysis: Efficiency major path in acid-catalyzed hydrolysis of amides3 which is ... protonation mechanism. |
|
The kinetics and mechanism of acid catalysed hydrolysis of lactams
schemes which must be considered in amide or. The acid-catalyzed hydrolysis of amides has lactam hydrolysis. Three of these (A-2 types) in-. |
|
Reactivity and mechanism of hydrolysis of phosphonamides
The reactivity of phosphorus amides is discussed in terms of their structure. We found a similar picture in the acid-catalyzed hydrolysis. |
|
Mechanism of the. alpha.-chymotrypsin-catalyzed hydrolysis of
enzyme-substrate complex and accepted notation for amino acids e.g. |
|
Transition state activity coefficients in the acid-catalyzed hydrolysis
This is interpreted in terms of an AoT2 mechanism of amide hydrolysis studies of acid-catalyzed ester hydrolysis (4) and aromatic substitution (5). |
| Carboxylic acid participation in amide hydrolysis. External general |
|
On the hydrolysis mechanisms of amides and peptides
15 mai 2018 · amide, formamide, hydrolysis rate law, mechanism, peptide, square root reaction ied base-catalyzed hydrolysis of three amides, and found |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
The precise mechanism of acid-catalysed hydrolysis of esters depends on the structure of the alkyl group in the ester A mechanism involving cleavage of the acyl−oxygen bond is most common and is observed for all simple esters Protonation of amides occurs on oxygen and not on nitrogen |
|
HYDROLYSIS
mechanisms account for neutral, acid and base hydrolysis Therefore, the SN2 reaction, where the phosphate ester anion is acting as the leaving group |

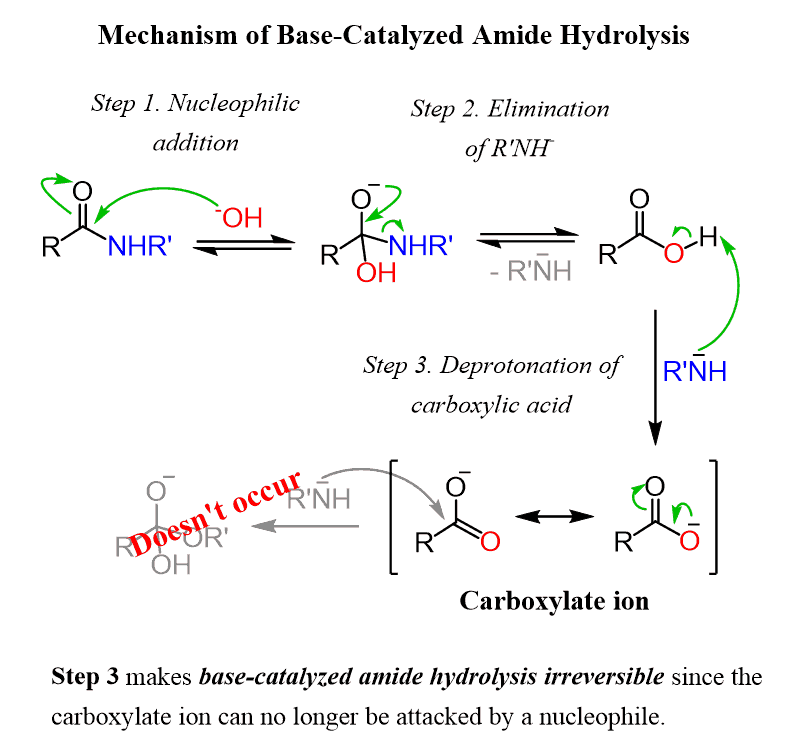
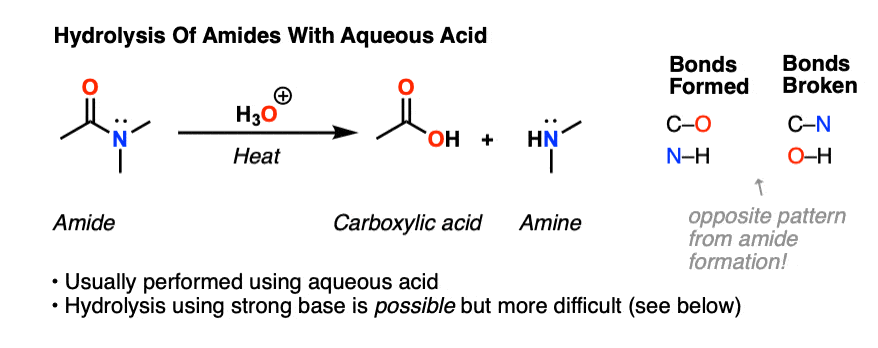


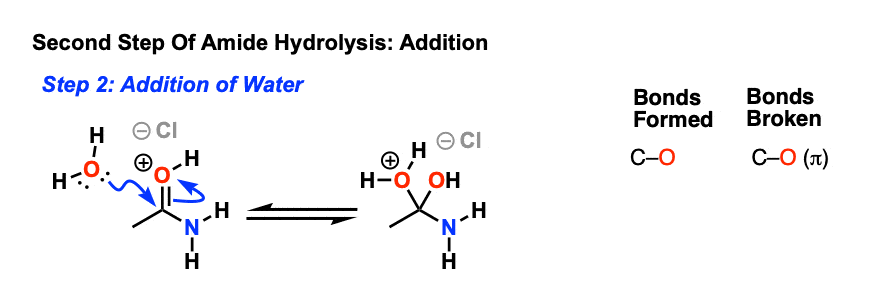


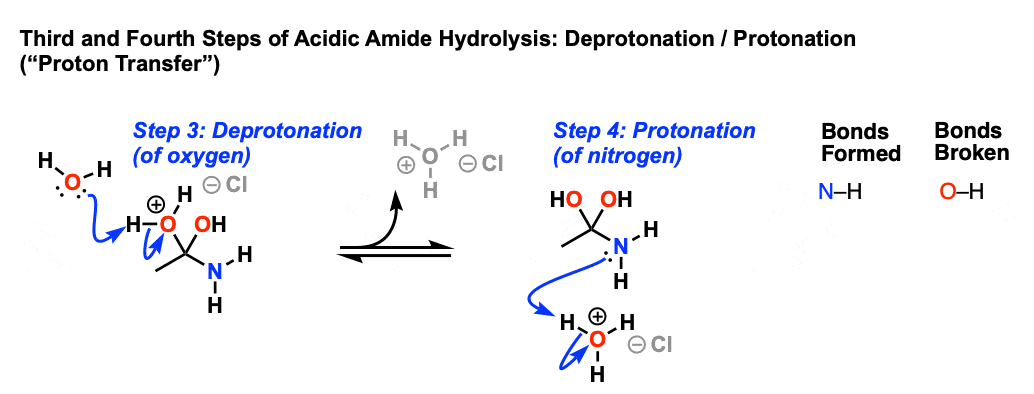


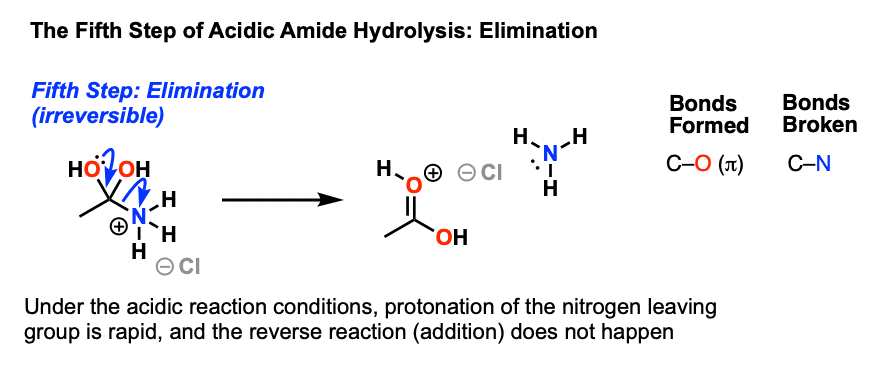
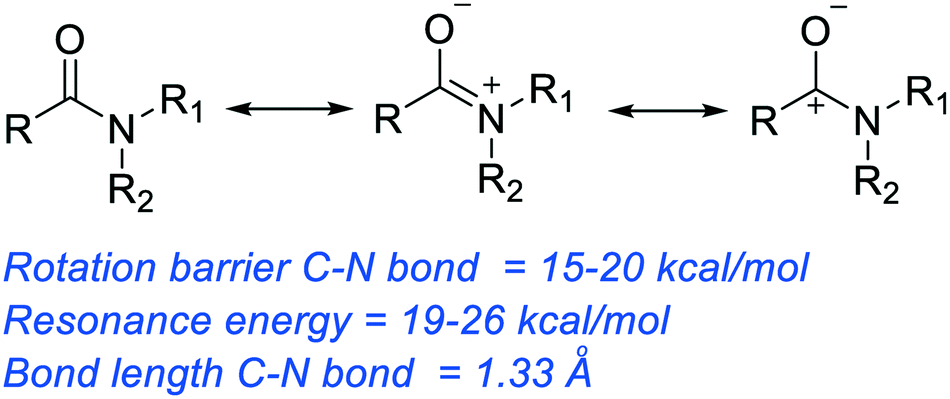






![A New Mechanism for Amide Hydrolysis - [PDF Document] A New Mechanism for Amide Hydrolysis - [PDF Document]](https://i1.rgstatic.net/publication/232944716_The_Mechanisms_of_Acid-Catalyzed_Hydrolysis_of_N-4-Substituted_Arylthio_Phthalimides/links/54293bee0cf238c6ea7d1bfc/largepreview.png)