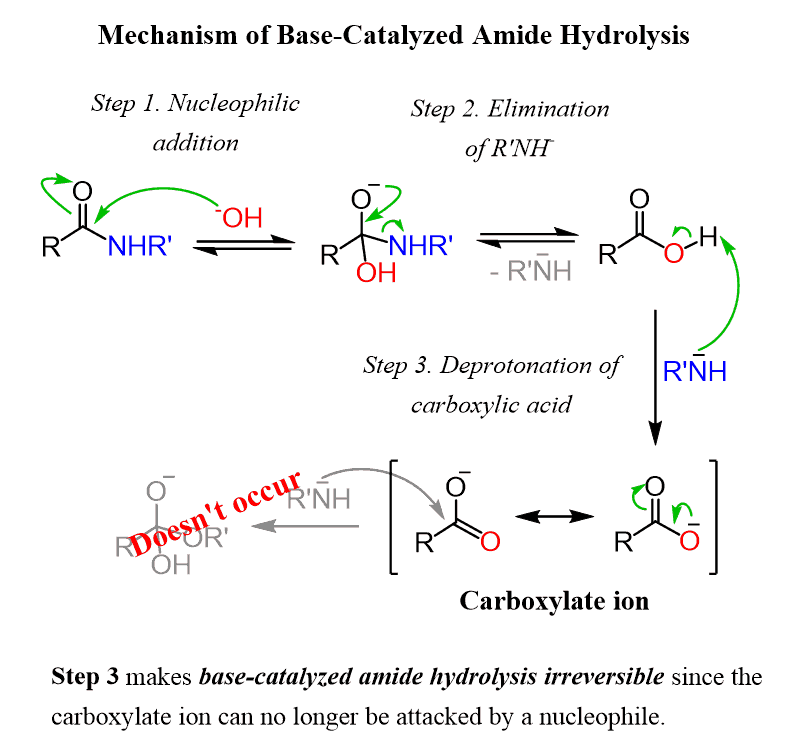
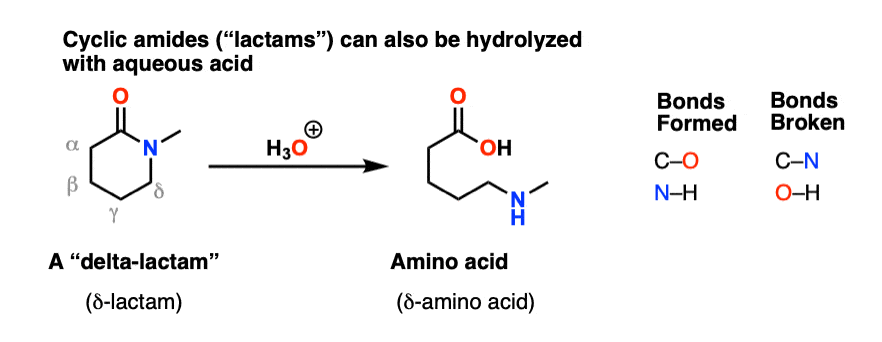
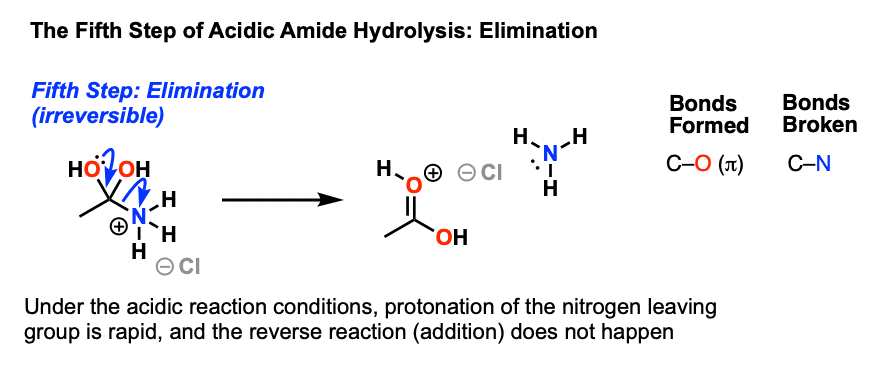
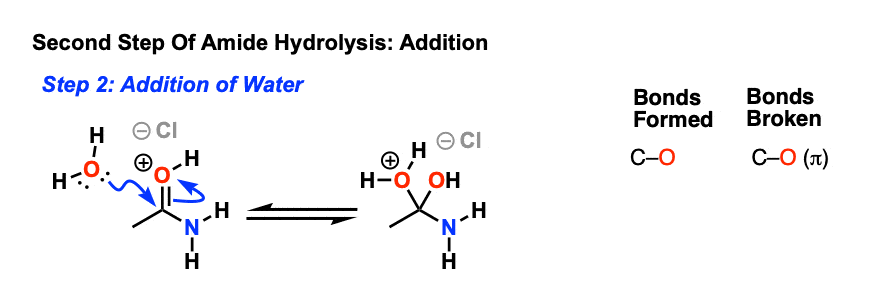
acid catalyzed hydrolysis of amides
|
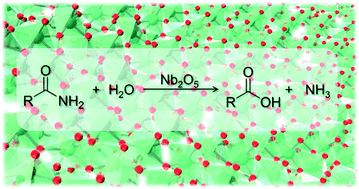
Hydrolysis of Amides to Carboxylic Acids Catalyzed by Nb2O5
25 déc. 2020 KEYWORDS: Nb2O5 Catalyst Amide hydrolysis |
|
Reversible formation of intermediates during hydronium-catalyzed
acid-catalyzed hydrolysis of amides when conducted at low. [H30+] 7. Herein we report that two typical amides |
|
Transition state activity coefficients in the acid-catalyzed hydrolysis
This is interpreted in terms of an AoT2 mechanism of amide hydrolysis acid-catalyzed hydrolysis of amides there are. |
|
Catalyzed hydrolysis of amide and peptide bonds in proteins 1
and acidity but also on the acid used. When hydrolyzed at 65° C by certain strong monobasic acids of high molecular weight the amide and the peptide. |
|
Catalyzed Hydrolysis of Amides
and Trypsin-Catalyzed Hydrolysis of Amides of the amide bond catalyzed by trypsin and a-chymotrypsin. ... pf -guanidine benzoic acid [ 161. |
|
Observation of large amounts of carbonyl-18O exchange concurrent
demonstrates that the acid-catalyzed hydrolysis of amides is observed during H30+-catalyzed amide hydrolysis depends upon the C-N/C-O cleavage ratio and. |
|
Mild hydrolysis or alcoholysis of amides. Ti(IV) catalyzed conversion
aqueous hydrochloric acid converts this class of compounds into the corresponding carboxylic acids or esters. Thus heating the carboxamide in a solution of the |
|
Mechanism of the. alpha.-chymotrypsin-catalyzed hydrolysis of
enzyme-substrate complex and accepted notation for amino acids e.g. |
|
Copper Complex Catalyzed Hydrolysis of Amides
enzyme for hydrolyzing esters or amides |
|
Theoretical Study of Base-Catalyzed Amide Hydrolysis: Gas-and
Even though acids can promote amide hydrolysis base catalysis is generally more efficient.1 In a number of studies on aliphatic amides |
|
On the hydrolysis mechanisms of amides and peptides
15 mai 2018 · There are large pH regions for which the hydrolysis is dom- inated by acid catalysis (0 < pH < 5, slope = −1) or base catalysis (6 < pH < 10, |
|
Lecture 6: Hydrolysis Reactions of Esters and Amides
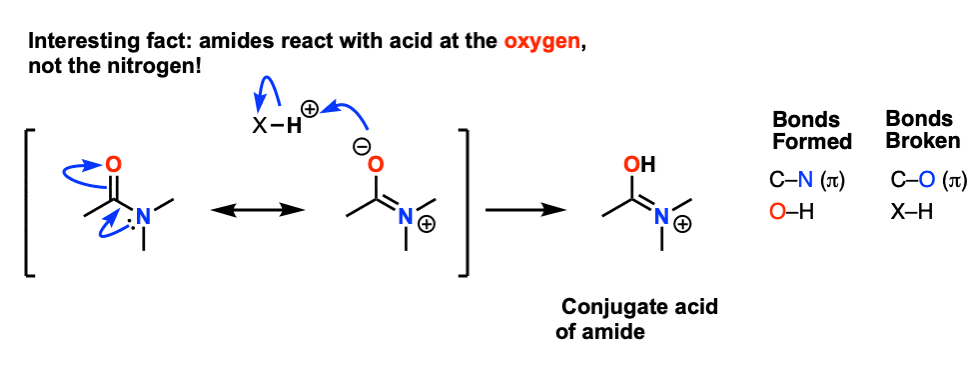
Acid-catalysed hydrolysis reactions are reversible The forward reaction is driven over to product by using an excess of water, usually as the solvent Transesterification reactions proceed via similar mechanisms and are also catalysed by either acid or base Protonation of amides occurs on oxygen and not on nitrogen |